Intracellular trafficking is an essential and conserved eukaryotic process. Rab GTPases are a family of proteins that regulate and provide specificity for discrete membrane trafficking steps by harnessing a nucleotide-bound cycle. Global proteomic screens have revealed many Rab GTPases as phosphoproteins, but the effects of this modification are not well understood. Using the Saccharomyces cerevisiae Rab GTPase Sec4p as a model, we have found that phosphorylation negatively regulates Sec4p function by disrupting the interaction with the exocyst complex via Sec15p. We demonstrate that phosphorylation of Sec4p is a cell cycle–dependent process associated with cytokinesis. Through a genomic kinase screen, we have also identified the polo-like kinase Cdc5p as a positive regulator of Sec4p phosphorylation. Sec4p spatially and temporally localizes with Cdc5p exclusively when Sec4p phosphorylation levels peak during the cell cycle, indicating Sec4p is a direct Cdc5p substrate. Our data suggest the physiological relevance of Sec4p phosphorylation is to facilitate the coordination of membrane-trafficking events during cytokinesis.
Introduction
Membrane trafficking is a conserved and essential eukaryotic process in which vesicle-enclosed cargo is transported from one cellular destination to another. Vesicles are loaded with specific materials for their predetermined destination; therefore, trafficking must be tightly regulated to prevent deleterious effects, such as diseases including, but not limited to, Alzheimer’s disease, cystic fibrosis, I-cell disease, Griscelli syndrome, and Huntington’s disease (Ben-Yoseph et al., 1987; Ménasché et al., 2000; Heda et al., 2001; Gauthier et al., 2004; Uemura et al., 2004; Howell et al., 2006). Members of the Ras superfamily of small GTPases, Rab GTPases (Rabs), help to coordinate vesicular transport throughout the cell, with at least one Rab controlling each discrete membrane-trafficking step.
Rabs perform their function of regulating membrane trafficking by acting as molecular switches, able to cycle between two different nucleotide-bound conformations (GTP/GDP). Rabs exchange GDP for GTP with the help of guanine-nucleotide exchange factors, usually in association with a distinct membrane location where they engage with effector proteins. Rabs hydrolyze GTP to GDP through the help of GTPase-activating proteins. Rab proteins can cycle on and off of membranes through a dual geranylgeranyl modification at the C terminus (Rossi et al., 1991; Alory and Balch, 2000; Barr and Lambright, 2010) and are recycled from the membrane to a cytosolic pool of protein through interaction with guanine-nucleotide dissociation inhibitor proteins (Ullrich et al., 1993; Pan et al., 2006).
Phosphorylation is a known modification of Rabs, with 55 of the ∼60 human Rabs containing at least one phosphorylated residue (Hornbeck et al., 2015); however, very little is known about the kinases and cellular pathways involved in regulating phosphorylation and how these modifications may affect function. Previous work using Saccharomyces cerevisiae Rab Sec4p as a model found that Sec4p is a multisite phosphoprotein with four serine phosphorylation sites situated within two stretches close to the N and C termini, 5TVpSASpSGNGK15 and 196EGNIpSINpSGSGNS209 (Ficarro et al., 2002), and a negative regulatory role for Sec4p modification (Heger et al., 2011). In addition, global kinase screens have revealed kinase regulation of membrane-trafficking pathways may occur at multiple stages of the endomembrane system (Pelkmans et al., 2005). In this study, we have investigated the kinase/kinases involved in modifying Sec4p to elucidate the cellular pathway(s) that control Sec4p phosphorylation.
Sec4p is encoded by an essential gene and is a critical mediator for the pathway that delivers budding yeast post-Golgi vesicles to the plasma membrane (Goud et al., 1988). This trafficking step is spatially and temporally regulated to the sites of active growth reflected in Sec4p localization at the tip of newly growing cells and at the neck region between dividing cells (Novick and Brennwald, 1993). The closest mammalian orthologues of Sec4p are Rab3A, Rab8, and Rab13 (Collins, 2005), and these proteins also regulate post-Golgi trafficking pathways. The consequences of Rab GTPase activation are transmitted to downstream effectors, proteins, or protein complexes that bind to the nucleotide-activated or GTP-bound conformation (Grosshans et al., 2006). Several effectors have been identified for Sec4p, including the octameric exocyst complex (Guo et al., 1999). The exocyst complex is also an effector for other Ras-related small GTPases and is known to be a central player that serves as an intersection point for multiple signal transduction pathways (Chang et al., 2004; Gromley et al., 2005; Chien et al., 2006; Blankenship et al., 2007; Goehring et al., 2007; He et al., 2007; Jiang et al., 2007; Nejsum and Nelson, 2007; Stuart et al., 2007; Bao et al., 2008). The results of our study suggest that Sec4p phosphorylation acts to control exocyst function during cytokinesis via the polo-kinase Cdc5p.
Results
Identification of phosphorylated Sec4p in vivo
To investigate and measure the phosphorylation status of Sec4p, antibodies were raised against modified Sec4p (Fig. 1). The results are shown for an antibody specifically raised to detect phosphorylation at the N terminus of Sec4p. Characterization of this custom antibody using chemically modified Sec4p peptides (residues 3–14 of Sec4p) reveals specific detection of serine 8 phosphorylation (pS8), and this detection is independent of the phosphorylation status of nearby serine residues (Fig. 1 A). Furthermore, affinity-tagged, purified Sec4p protein was found to produce a robust pS8 signal; however, this signal was lost upon serine 8 to alanine mutation, in additional to alkaline phosphatase treatment of the purified wild-type protein (Fig. 1 C). These data demonstrate a pool of phosphorylated Sec4p exists in vivo; however, the dynamics of this modification and sensitivity to signaling pathways remained unknown.
Detecting phosphorylation in the N terminus of the Rab GTPase Sec4p. (A) Schematic of the Rab GTPase Sec4p showing relative positions of the GTPase domain and phosphorylation sites. N- and C-terminal phosphorylation sites highlighted in red were targeted for antibody production. (B) Dot blot of phosphate-modified peptides (sequence shown and site of phosphate modifications highlighted in red) representing Sec4p3–14 probed with a custom antibody raised against phosphorylated Sec4p peptide sequences. (C) Western blot of S. cerevisiae–purified MBP-Sec4p (with and without alkaline phosphatase treatment) and MBP-Sec4pAla (S8, 10, 11, 201, and 204A) using pS8 antibody and pan-Sec4p antibody. Calf intestinal alkaline phosphatase (CIP) was incubated with purifed Sec4p for 30 min at 30°C to remove phosphorylation. MBP-Sec4p is expressed from an exogenous plasmid (pRS315 backbone) as the only cellular copy of Sec4p. (D) Regulatory control of Sec4p phosphorylation in response to nutrient depravation. Carbon and nitrogen starvation of wild-type cells. Cells were split and incubated at 25°C for 3 h in YPD, YP + 2% glycerol, YP + 2% sucrose, and SD-N medium before lysis. (E) Effects of TORC1 inhibition on Sec4p phosphorylation. Cells were split into media containing different concentrations of the TORC1 inhibitor rapamycin and incubated for 3 h at 25°C before lysis. WT, wild-type.
Detecting phosphorylation in the N terminus of the Rab GTPase Sec4p. (A) Schematic of the Rab GTPase Sec4p showing relative positions of the GTPase domain and phosphorylation sites. N- and C-terminal phosphorylation sites highlighted in red were targeted for antibody production. (B) Dot blot of phosphate-modified peptides (sequence shown and site of phosphate modifications highlighted in red) representing Sec4p3–14 probed with a custom antibody raised against phosphorylated Sec4p peptide sequences. (C) Western blot of S. cerevisiae–purified MBP-Sec4p (with and without alkaline phosphatase treatment) and MBP-Sec4pAla (S8, 10, 11, 201, and 204A) using pS8 antibody and pan-Sec4p antibody. Calf intestinal alkaline phosphatase (CIP) was incubated with purifed Sec4p for 30 min at 30°C to remove phosphorylation. MBP-Sec4p is expressed from an exogenous plasmid (pRS315 backbone) as the only cellular copy of Sec4p. (D) Regulatory control of Sec4p phosphorylation in response to nutrient depravation. Carbon and nitrogen starvation of wild-type cells. Cells were split and incubated at 25°C for 3 h in YPD, YP + 2% glycerol, YP + 2% sucrose, and SD-N medium before lysis. (E) Effects of TORC1 inhibition on Sec4p phosphorylation. Cells were split into media containing different concentrations of the TORC1 inhibitor rapamycin and incubated for 3 h at 25°C before lysis. WT, wild-type.
Sec4p phosphorylation is sensitive to nutrient availability
Considering that Sec4p is directly responsible for cellular growth by delivering membrane to the site of polarized exocytosis on the plasma membrane, we reasoned that phosphorylation may be sensitive to signaling pathways activated or inhibited under different nutrient availability conditions. To test this hypothesis, cells were grown under carbon- and nitrogen-starvation conditions for 3 h, and then the phosphorylation status of Sec4p was accessed via Western blot (Fig. 1 D). Phosphorylation was significantly reduced upon both starvation conditions. No significant change in phosphorylation was observed when sucrose was the only carbon source. Additionally, the nitrogen starvation conditions contained ample glucose, but still showed a significant decrease in Sec4p phosphorylation, suggesting that glucose repression was not responsible for the changes observed (Gancedo, 1998). Nutrient availability typically signals through the target of rapamycin (TOR) pathway, leading to the regulation cell growth, translation initiation, cell cycle progression from G1, and autophagy (Barbet et al., 1996; Cardenas et al., 1999; Kamada et al., 2000). Inhibition of TOR complex 1 (TORC1) using rapamycin shows a dose-dependent ablation of Sec4p phosphorylation (Fig. 1 E). Collectively, the status of Sec4p phosphorylation is altered in response to general nutrient availability and downstream of the TORC1 signaling pathway.
Sec4p phosphorylation accumulates on membrane-bound Sec4p and interrupts the interaction with effector protein Sec15p
Sec4p phosphorylation has previously been shown to negatively regulate Sec4p function (Heger et al., 2011). Sec4p mutants that replace phosphorylated serine residues with aspartic acid residues to mimic the constitutively phosphorylated state of the protein (phosphomimetic mutants) failed to functionally replace the wild-type protein, whereas the complementary serine-to-alanine mutations (Sec4pAla) were able to functionally replace wild-type Sec4p. However, the molecular mechanism by which phosphorylation negatively affects function is not understood. Using the pS8 antibody, we began to investigate how phosphorylation affects function. First, we investigated whether the nucleotide-bound state of the protein affected phosphorylation. Previous work had shown that nucleotide hydrolysis and exchange rates of phosphomimetic Sec4p were indistinguishable from wild-type protein in the presence of the GTPase-activating protein and guanine-nucleotide exchange factor, respectively (Heger et al., 2011). However, the effect of the nucleotide-bound state of Sec4p on phosphorylation was not investigated. Using GTP-hydrolysis (Q79L) and GDP exchange–deficient (S29V) (Rinaldi et al., 2015) mutants of Sec4p as the sole copy, probing Western blots with the Sec4p phospho-specific antibody revealed no significant change in Sec4p phosphorylation levels (Fig. 2 A). However, membrane-fractionation experiments to separate the soluble and membrane-bound Sec4p protein pools indicated that phosphorylation was enriched on the membrane-bound fraction of Sec4p (Fig. 2 B). Furthermore, using a GFP-tagged, temperature-sensitive phosphomimetic mutant of Sec4p previously characterized (Heger et al., 2011), we found the Sec4p mutant accumulated on vesicles in the cytosol at the restrictive (but not permissive) temperature (Fig. 2 C).
Sec4p phosphorylation accumulates on membrane-bound Sec4p and interrupts the interaction with effector protein Sec15p. (A) Phosphorylation status of GTP hydrolysis–deficient (Q79L) and GDP exchange–deficient (S29V) Sec4p mutants. (B) Membrane fractionation of MBP-Sec4p and MBP-Sec4pAla as the only copy of Sec4p and subsequent Western blot. 100,000 g centrifugation was used to separate the cytosolic (Cyto.) and membrane (Mem.) fractions. (C) Localization of GFP-Sec4p and temperature-sensitive phosphomimetic GFP-Sec4pDASD DIND (S8, 11, 201, and 204D) at permissive (25°C) and restrictive (37°C) temperatures. Cells were split, incubated at designated temperature for 2 h, and then fixed in 4% PFA before microscopy. (D) Pulldown of Sec15p-MBP that was coexpressed with GFP-Sec4p, GFP-Sec4pAla, or GFP-Sec4pAsp and subsequent Western blot analysis using GFP antibody to determine Sec15p interaction with Sec4p. WT, wild type.
Sec4p phosphorylation accumulates on membrane-bound Sec4p and interrupts the interaction with effector protein Sec15p. (A) Phosphorylation status of GTP hydrolysis–deficient (Q79L) and GDP exchange–deficient (S29V) Sec4p mutants. (B) Membrane fractionation of MBP-Sec4p and MBP-Sec4pAla as the only copy of Sec4p and subsequent Western blot. 100,000 g centrifugation was used to separate the cytosolic (Cyto.) and membrane (Mem.) fractions. (C) Localization of GFP-Sec4p and temperature-sensitive phosphomimetic GFP-Sec4pDASD DIND (S8, 11, 201, and 204D) at permissive (25°C) and restrictive (37°C) temperatures. Cells were split, incubated at designated temperature for 2 h, and then fixed in 4% PFA before microscopy. (D) Pulldown of Sec15p-MBP that was coexpressed with GFP-Sec4p, GFP-Sec4pAla, or GFP-Sec4pAsp and subsequent Western blot analysis using GFP antibody to determine Sec15p interaction with Sec4p. WT, wild type.
Because it appeared that Sec4p phosphorylation was negatively affecting the function of Sec4p on membranes, but before vesicle fusion, we decided to investigate the effector-binding capacity of the phosphomimetic protein. Yeast two-hybrid analysis had previously implicated interactions with the exocyst component and Sec4p effector protein Sec15p were disrupted by Sec4p phosphorylation (Heger et al., 2011). To investigate this suggestion biochemically, affinity-tagged Sec15p was affinity purified from cells ectopically expressing GFP-tagged Sec4pWT, Sec4pAla, and phosphomimetic Sec4p (Fig. 2 D). The results of this experiment show phosphomimetic Sec4p, but not Sec4pAla, has a diminished affinity for Sec15p. Failure of Sec4p to effectively engage with Sec15p is known to result in severe membrane-trafficking defects leading to cell death and could explain why the phosphomimetic Sec4p is a nonfunctional replacement of Sec4p.
Kinase overexpression screen to identify Sec4p kinases
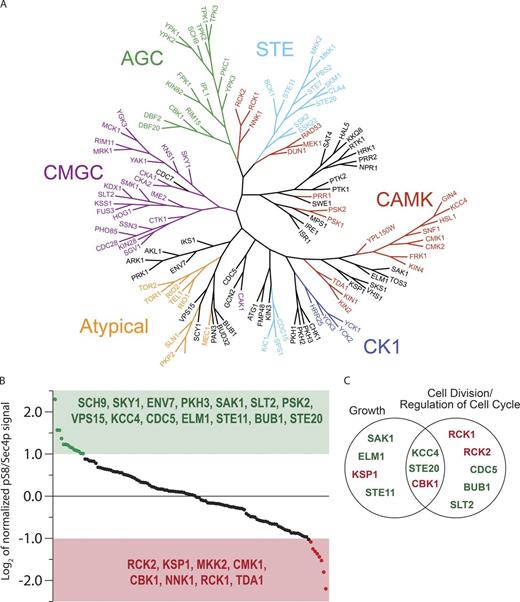
Our data indicate that effector binding, which is already known to be under the control of the nucleotide-bound state of Sec4p, can also be controlled by the phosphorylation status of Sec4p. This suggests that additional cellular pathways may impinge upon membrane trafficking through Sec4p effector protein recruitment, independent of its nucleotide-bound state. Indeed, the nutrient-starvation phenotype and sensitivity to rapamycin suggests a pathway downstream of TORC1, of which there are many, could be involved. To elucidate the particular cellular pathways/processes responsible for controlling Sec4p phosphorylation, we sought to identify the kinases directly responsible for modifying Sec4p. To accomplish this, a kinase overexpression library was constructed and a screen performed to measure the phosphorylation status of Sec4p in each overexpression strain. Because of the redundancy of many S. cerevisiae kinases, an overexpression screen was favored as opposed to a deletion screen in which paralogous kinases have the potential to moderate the effects of individual kinase deletions. Additionally, all essential kinases can be included in an overexpression screen, which is not the case for a deletion library. 127 kinases and predicted kinases were cloned onto multicopy plasmids with their endogenous promoters for the overexpression library (Fig. 3 A). For each kinase overexpression strain, the levels of Sec4p pS8 and total Sec4p protein were measured in duplicate via Western blot and normalized to a vector-only control (Fig. 3 B). Additionally, the data from the screen were processed to compare an increase in pS8 levels to a vector-only control in case kinase overexpression leads to aberration in Sec4p expression without disrupting the pool of phosphorylated Sec4p (Fig. S1). Several kinases were found to significantly increase Sec4p phosphorylation, making them prime candidates for bona fide Sec4p kinases (a significant increase was defined as a doubling of Sec4p phosphorylation per total Sec4p protein, relative to a vector-only control). Gene ontology of these kinases together with kinases that significantly lowered Sec4p phosphorylation was conducted, and cellular processes like growth and mitotic cell cycle were enriched (Fig. 3 C), consistent with previous observations of the impact of nutrient availability on Sec4p phosphorylation status.
Kinase overexpression screen to identify entities responsible for Sec4p phosphorylation. (A) Tree view of all 127 kinases/predicted kinases used in this study. (B) Summary of kinase overexpression screen with each point representing results from a single kinase assay. Kinases are ordered from most positive increase to most negative decrease in Sec4p phosphorylation relative to the total Sec4p signal (Table S1). Significance was assessed as a deviation from control of twofold (log2 = 1). (C) Gene ontology (SGD Gene Ontology Term Finder) of kinases that significantly affect Sec4p phosphorylation.
Kinase overexpression screen to identify entities responsible for Sec4p phosphorylation. (A) Tree view of all 127 kinases/predicted kinases used in this study. (B) Summary of kinase overexpression screen with each point representing results from a single kinase assay. Kinases are ordered from most positive increase to most negative decrease in Sec4p phosphorylation relative to the total Sec4p signal (Table S1). Significance was assessed as a deviation from control of twofold (log2 = 1). (C) Gene ontology (SGD Gene Ontology Term Finder) of kinases that significantly affect Sec4p phosphorylation.
Identification of the polo-like kinase Cdc5p
To better understand the relationship between kinase overexpression and Sec4p phosphorylation, we sought to reduce the activity of each candidate kinase and look for a subsequent reduction of Sec4p phosphorylation. Of the subsets of kinases for which overexpression significantly increased Sec4p phosphorylation, all but one were nonessential. Using the Resgen haploid deletion collection, pS8 levels were measured for each kinase deletion strain. Surprisingly, most kinase deletions did not significantly reduce Sec4p phosphorylation, with the exception of Vps15p and, to a lesser extent, Sch9p, but neither kinase deletion strain completely abolished Sec4p phosphorylation (Fig. 4 A). Vps15p is a vacuolar membrane–associated kinase for which deletion results in membrane-trafficking defects (Herman et al., 1991). General membrane-trafficking defects have been shown to attenuate pS8 levels in a similar fashion (Fig. S2); thus, we believe this to be a secondary effect. Additionally, Sec4p is not known to be associated with vacuolar traffic under conditions of normal cell growth. Sch9Δ only causes a minor reduction in phosphorylation; thus, we also believe this to be a secondary affect, although overexpression does increase Sec4p phosphorylation. Sch9p is a direct substrate of TORC1, and TORC1 activity was shown to be required for Sec4p phosphorylation, thus potentially explaining the overexpression phenotype (Urban et al., 2007).
Cdc5p is responsible for Sec4p phosphorylation. (A) Sec4p phosphorylation status in cells deleted for nonessential kinases identified in overexpression screen. 10 OD units of cells were assayed for Sec4p phosphorylation with significance as described for Fig. 3. (B, top) Sec4p phosphorylation status in wild-type (WT) and cdc5ts cell at the permissive (25°C) and restrictive (37°C) temperatures. Cells were split and grown in YPD for 2 h at the specified temperature before lysis and Western blot analysis. (B, bottom) Quantification of Sec4p phosphorylation relative to total Sec4p protein in cdc5ts cells at both permissive and restrictive temperature. The histogram shows the mean from three independent replicates quantified from Western blots and error bars show the SD. (C, left) Western blot of cells overexpressing Cdc5p under the GAL4 promoter with galactose (Gal) as the sole carbon source compared with vector-only control. (C, right) Bar graph showing the mean pS8 values from three independent replicates of both WT and Cdc5p overexpression quantified from Western blots, and error bars show the SD. (D, left) Disruption of hypothetical polo-box binding motif on Sec4p (S10,11A) and subsequent Western blot analysis to measure changes in Sec4p phosphorylation. (D, right) Quantification of three separate experiments measuring phosphorylation status relative to total Sec4p protein as in B and C. (E) In vitro kinase assay with purified Cdc5p and peptides representing Sec4p3-14 as substrates. Samples were incubated at 30°C for 1 h and then analyzed via dot blot of products probed with Sec4p pS8 antibody.
Cdc5p is responsible for Sec4p phosphorylation. (A) Sec4p phosphorylation status in cells deleted for nonessential kinases identified in overexpression screen. 10 OD units of cells were assayed for Sec4p phosphorylation with significance as described for Fig. 3. (B, top) Sec4p phosphorylation status in wild-type (WT) and cdc5ts cell at the permissive (25°C) and restrictive (37°C) temperatures. Cells were split and grown in YPD for 2 h at the specified temperature before lysis and Western blot analysis. (B, bottom) Quantification of Sec4p phosphorylation relative to total Sec4p protein in cdc5ts cells at both permissive and restrictive temperature. The histogram shows the mean from three independent replicates quantified from Western blots and error bars show the SD. (C, left) Western blot of cells overexpressing Cdc5p under the GAL4 promoter with galactose (Gal) as the sole carbon source compared with vector-only control. (C, right) Bar graph showing the mean pS8 values from three independent replicates of both WT and Cdc5p overexpression quantified from Western blots, and error bars show the SD. (D, left) Disruption of hypothetical polo-box binding motif on Sec4p (S10,11A) and subsequent Western blot analysis to measure changes in Sec4p phosphorylation. (D, right) Quantification of three separate experiments measuring phosphorylation status relative to total Sec4p protein as in B and C. (E) In vitro kinase assay with purified Cdc5p and peptides representing Sec4p3-14 as substrates. Samples were incubated at 30°C for 1 h and then analyzed via dot blot of products probed with Sec4p pS8 antibody.
The only remaining kinase that significantly increased Sec4p phosphorylation upon overexpression was the essential polo-like kinase Cdc5p. Cdc5p, as the ninth-ranked kinase in the overexpression screen, was not an obvious candidate. We reasoned that as Cdc5p is normally present at very low levels as a result of being targeted for degradation in G1 and that Cdc5p overexpression is toxic to cells, the results from lysates of cells overexpressing Cdc5p on episomal plasmids may not reflect the potential of Cdc5p to phosphorylate Sec4p. To address this deficiency, we used a regulatable promoter to selectively induce Cdc5p overexpression and found that the increase in Sec4p phosphorylation was significantly increased relative to episomal plasmid expression (Fig. 4 C).
Cdc5p is responsible for regulating a variety of cellular processes including, but not limited to, progression of cytokinesis via recruitment of Rho1, timely mitotic exit, nuclear shape during mitosis, and Cdc14 release during anaphase (Yoshida et al., 2006; Botchkarev et al., 2014; Walters et al., 2014). Cdc5p was also identified in the landmark cell division cycle screen; thus, a temperature-sensitive allele is available for further study (Hartwell et al., 1973). Using the cdc5ts allele, we sought to determine if Cdc5p activity was required for Sec4p phosphorylation. Indeed, at the restrictive temperature, Sec4p phosphorylation was almost completely ablated in the cdc5ts strain (Fig. 4 B). Additionally, there does appear to be a slight decrease in Sec4p pS8 levels even at the permissive temperature, consistent with a partially impaired Cdc5p protein.
The activity of Cdc5p is regulated by its spatial and temporal subcellular localization throughout cell cycle progression (Cheng et al., 1998; Park et al., 2004). Cdc5p protein levels are controlled in an antigen-presenting cell–dependent way, peaking in S/G2 and being actively degraded in G1 (Visintin et al., 2008). Furthermore, Cdc5p is targeted to its substrates via a polo-box domain (PBD) through recognition of a core S–pS/pT–P/X phosphopeptide motif (Song et al., 2000; Elia et al., 2003). Examination of the mass spectrometry–confirmed phosphorylation sites on Sec4p reveals a hypothetical core PBD recognition site on Sec4p amino acids 10–12 (Fig. 1 A). Disruption of this hypothetical PBD recognition motif via alanine mutagenesis (S10,11A) shows a dramatic decrease in Sec4p phosphorylation (Fig. 4 D). Additionally, based on the characterization of the pS8 antibody, we are confident that mutagenesis of S10 and S11 does affect substrate recognition.
To show that Cdc5p can directly modify Sec4p, Cdc5p was purified from S. cerevisiae and used for in vitro kinase assays with Sec4p3–14 peptides as substrates. Peptides were analyzed for modifications via a dot blot and subsequent probing with the pS8 antibody. Cdc5p was able to directly modify Sec4p peptide substrates (Fig. 4 E). The addition of a phosphate on S11 to mimic the hypothetical polo-box binding phosphopeptide did not affect the final total of phosphorylated Sec4p as compared with unmodified Sec4p peptide; however, the rates of phosphorylation were not addressed in this study.
Sec4p phosphorylation is a dynamic cell cycle–sensitive modification
To determine the physiological relevance of phosphorylation of Sec4p by Cdc5p, we examined the spatial and temporal localization of Cdc5p and Sec4p. Both Cdc5p and Sec4p change subcellular localization throughout the cell cycle, with Sec4p localizing to the bud tip, the periphery of the growing bud, and the bud neck for cytokinesis and Cdc5p localizing to the nucleus, spindle-pole bodies, and site of cytokinesis (Song et al., 2000). Using a GFP-tagged Sec4p and a 3x-mCherry–tagged Cdc5p, we examined the localization of both proteins at various stages of the cell cycle (Fig. 5 A). Early budded cells in G1 show Sec4p at the tip of the growing bud, whereas Cdc5p is either not visible or appears to be in the vacuole, consistent with Cdc5p being actively degraded. In S/G2, Cdc5p becomes localized to the nucleus or spindle pole bodies, whereas Sec4p is more distributed throughout the daughter cell for isotropic growth. During late M-phase or cytokinesis, we find that Sec4p and Cdc5p colocalize at the bud neck. Not only does this show that Cdc5p is in a position to phosphorylate Sec4p in vivo, but it also suggests that Sec4p is phosphorylated at a specific time point in the mitotic cell cycle.
Sec4p phosphorylation peaks during colocalization of Sec4p and Cdc5p during cytokinesis. (A) Microscopy of GFP-Sec4p and Cdc5p-3xmCherry at various stages of cell cycle based on bud size and Sec4p localization. (B, top) Nocodazole synchrony and release of GFP-Sec4p–expressing cells, in which Sec4p localization was monitored and categorized. (bottom) Nocodazole synchrony and release for wild-type cells, in which cells were pelleted and frozen in liquid nitrogen for each time point. (C) α-Factor synchrony and release of bar1Δ cells. At each indicated time point, cells were pelleted and frozen in liquid nitrogen for Western blot. (top) Analysis of Sec4p localization imaged by immunofluorescence at the indicated time points after α-factor release. (bottom) Cells were taken from the same culture and time points as for Western blot analysis, but fixed in 2% PFA for each time point.
Sec4p phosphorylation peaks during colocalization of Sec4p and Cdc5p during cytokinesis. (A) Microscopy of GFP-Sec4p and Cdc5p-3xmCherry at various stages of cell cycle based on bud size and Sec4p localization. (B, top) Nocodazole synchrony and release of GFP-Sec4p–expressing cells, in which Sec4p localization was monitored and categorized. (bottom) Nocodazole synchrony and release for wild-type cells, in which cells were pelleted and frozen in liquid nitrogen for each time point. (C) α-Factor synchrony and release of bar1Δ cells. At each indicated time point, cells were pelleted and frozen in liquid nitrogen for Western blot. (top) Analysis of Sec4p localization imaged by immunofluorescence at the indicated time points after α-factor release. (bottom) Cells were taken from the same culture and time points as for Western blot analysis, but fixed in 2% PFA for each time point.
To determine if Sec4p phosphorylation is a cell cycle–dependent modification, we performed cell synchrony and release experiments via two independent methods: the mating pheromone α-factor to halt cells in G1 and the microtubule-depolymerizing agent nocodazole to halt cells in metaphase. In both cases, Sec4p phosphorylation was found to be a highly dynamic process and only showing pS8 phosphorylation for ∼30 min per each cell cycle (Fig. 5, B and C). The localization of Sec4p during synchrony and release experiments was monitored by GFP-tagged Sec4p (nocodazole release) and immunofluorescence (α-factor release), and, in both cases, the rise and fall of bud neck localization of Sec4p corresponds with pS8 phosphorylation (Fig. 5, B and C). These data show that phosphorylation of Sec4p occurs in a cell cycle–dependent manner and specifically when Sec4p is at the bud neck, where it colocalizes with Cdc5p. Collectively, Cdc5p phosphorylates Sec4p in a cell cycle–dependent manner at the bud neck of late-budded cells during cytokinesis.
Sec4p phosphorylation is sensitive to cytokinetic defects and impacts cell size
Our data indicated Sec4p phosphorylation is a cell cycle modification that is spatially and temporally restricted to cytokinesis. To further investigate the physiological relevance of Sec4p phosphorylation in relation to cytokinesis, we examined the impact of cytokinetic defects on Sec4p phosphorylation. We hypothesized that Sec4p phosphorylation may provide a mechanism to link plasma membrane trafficking at the site of cell division with other events of cytokinesis. To induce cytokinetic defects, we used myo1Δ and hof1Δ cell lines, which are distinct mutants shown to be defective in cell separation, but still viable (Watts et al., 1987; Lippincott and Li, 1998; Korinek et al., 2000). Hof1p acts just before and during cytokinesis in regulation of the actin cytoskeleton and formins (Oh et al., 2013). Myo1p is a type II myosin heavy chain, that acts downstream of Hof1p and localizes to the actomyosin ring for cell separation after cytokinesis is initiated. hof1Δ cells also have enlarged cell size and defects in apical growth (Graziano et al., 2014). Interestingly, Hof1p is also a reported substrate for Cdc5p (Meitinger et al., 2011). We found that in hof1Δ cells, but not myo1Δ cells, there is a very large increase in Sec4p phosphorylation (Fig. 6, A and B). Furthermore, Sec4p localization in a hof1Δ cell line is in agreement with Graziano et al. (2014), and bud neck localization can be seen in groups of cells that have not completed budding (Fig. 6 C). This observation suggests that the increased Sec4p phosphorylation of hof1Δ cells reflects the fact that the hof1Δ cells spend a longer time in cytokinesis just before cell separation than do wild-type cells. Because phosphorylation was increased in the hof1Δ background, we next investigated the impact of removing Sec4p phosphorylation. For this experiment, we created a Sec4p allele in which each reported position of phosphorylation has been mutated to alanine (Hornbeck et al., 2015). This allele contains Sec4p with positions S2, 8, 10, 11, 201, 204, and T6 all converted to alanine residues; we refer to this mutant as Sec4pA7. When Sec4pA7 was expressed as the sole copy of Sec4p in hof1Δ, we saw that cells were significantly smaller, thus partially rescuing the hof1Δ phenotype although the cytokinetic defects remain (Fig. 6 D). This significant reversal of phenotype could be explained if the lack of phosphorylated Sec4p allows hof1Δ cells to bypass the period of waiting to physically separate, which is the major defect of these cells. The significant reversal of phenotype in the hof1Δ cells prompted us to further examine the impact of a nonphosphorylatable version of Sec4p in wild-type cells. If Sec4p phosphorylation is used to link growth with cytokinesis, then a cell unable to make use of this mechanism would be expected to have an associated cell size phenotype due to reduced coordination of plasma membrane growth with cell cycle division. We examined this in a wild-type genetic background, expressing Sec4pA7 as the sole copy, and observed that cells were reproducibly smaller (Fig. 6 E). These results suggest that Sec4p phosphorylation at the neck of dividing cells alters the timing of the membrane-trafficking events responsible for plasma membrane closure during cytokinesis. Phosphorylation blocks exocytosis in this region, and so wild-type cells grow slightly larger before cell division. In cells in which Sec4p cannot be phosphorylated, there is no such block at the neck, and cytokinesis progresses more rapidly, resulting in a slightly smaller cell size phenotype.
Sec4p phosphorylation is up-regulated upon cytokinetic stress and is required for proper cell size maintenance. (A) Sec4p phosphorylation status in myo1Δ strain compared with wild type (WT). Total Sec4p and Pgk1p were also probed for loading controls. Quantification of three independent measures is presented in a bar graph. (B) Sec4p phosphorylation status in hof1Δ strain compared with WT. Total Sec4p and Pgk1p were also probed for loading controls. Quantification of three independent measures is presented in a bar graph. (C) Microscopy of GFP-Sec4p in a hof1Δ background. (D) DIC microscopy of hof1Δ cells expressing wild-type Sec4p and Sec4p7A. (E) Quantification of cell size. Cell size was measured using ImageJ (National Institutes of Health) to take the area of the mother cell exclusively, as the size of the bud is more indicative of position within the cell cycle as opposed to overall cell size. Error bars are SD, and the significance was calculated using a two-tailed Student’s t test. (**) The confidence level was >99%. Measurements from 150 cells were made for each strain.
Sec4p phosphorylation is up-regulated upon cytokinetic stress and is required for proper cell size maintenance. (A) Sec4p phosphorylation status in myo1Δ strain compared with wild type (WT). Total Sec4p and Pgk1p were also probed for loading controls. Quantification of three independent measures is presented in a bar graph. (B) Sec4p phosphorylation status in hof1Δ strain compared with WT. Total Sec4p and Pgk1p were also probed for loading controls. Quantification of three independent measures is presented in a bar graph. (C) Microscopy of GFP-Sec4p in a hof1Δ background. (D) DIC microscopy of hof1Δ cells expressing wild-type Sec4p and Sec4p7A. (E) Quantification of cell size. Cell size was measured using ImageJ (National Institutes of Health) to take the area of the mother cell exclusively, as the size of the bud is more indicative of position within the cell cycle as opposed to overall cell size. Error bars are SD, and the significance was calculated using a two-tailed Student’s t test. (**) The confidence level was >99%. Measurements from 150 cells were made for each strain.
Discussion
Sec4p phosphorylation had previously been characterized to negatively regulate function; however, the signaling pathways involved, the molecular mechanism by which function is disrupted, the dynamics of these modifications, and the kinases involved were unclear (Heger et al., 2011). In this study, we demonstrate that Sec4p phosphorylation is a dynamic modification and observed the phosphorylation status changes according to nutrient availability. Nutrient starvation elicits numerous cellular responses, including halting cells in G1 (Barbet et al., 1996; Rohde et al., 2001), and further investigation revealed that Sec4p phosphorylation is a cell cycle–dependent modification that peaks during late M phase, specifically cytokinesis, and is removed during G1, suggesting that our data showing lack of phosphorylation during nutrient starvation were primarily reflective of cell cycle arrest. A previous genetic study examining interactions between cdc mutants and genes involved in polarized cell traffic had suggested a connection between pathways involved in cell cycle and polarity establishment (Finger and Novick, 2000), and in this study, we show that a genomic kinase screen for Sec4p phosphorylation reveals an enrichment for mitotic cell cycle kinases, further supporting the idea that changes in phosphorylation controlled by cell cycle progression are influential for membrane traffic.
The molecular mechanism by which phosphorylation negatively affects function appears to be via a disruption of the interaction between Sec4p and the exocyst component Sec15p. Indeed, phosphorylation is enriched on the membrane-bound pool of Sec4p relative to the cytosolic fraction and is in a physiologically relevant location to modulate the interaction between Sec4p and the exocyst complex. Temperature-sensitive phosphomimetic Sec4p is associated on the surface of accumulated vesicles, which is a phenotype similar to that observed in Sec15p temperature-sensitive mutants (Salminen and Novick, 1989). Furthermore, biochemical analysis of a full phosphomimetic shows reduced affinity for Sec15p relative to the wild-type and the complimentary alanine mutant. It is important to note that detection of phosphorylated Sec4p in this study was measured using pS8 as a marker; however, there are a total of seven phosphorylation sites that have been identified (Sadowski et al., 2013). It is possible that additional intermediate species of phosphorylated Sec4p exist and have distinct or overlapping functions. However, in this study, we were unable to distinguish potential differentially phosphorylated isoforms. The precise structure by which Sec4p associates with Sec15p is not currently known; however, it is known to include the regions on Sec4p that are modified by phosphorylation (Guo et al., 1999), and the structure of the human orthologue Rab3A with its effector Rabphilin includes the analogous N-terminal region that extends from the core GTPase domain (Stroupe and Brunger, 2000).
We also identified the polo-like kinase Cdc5p as a potential Sec4p kinase from a kinase overexpression screen and further demonstrated that Cdc5p activity is responsible for Sec4p phosphorylation, and Cdc5p is capable of directly modifying Sec4p in vitro. Cdc5 temperature-sensitive alleles show synthetic negative interaction with sec4-8 and sec15-1 (Fig. S4). Additionally, the spatial and temporal colocalization of Cdc5p and Sec4p corresponds to the rise of Sec4p phosphorylation during cytokinesis, and loss of phosphorylation correlates with the degradation of Cdc5p and Sec4p localization changes from the site of cytokinesis to the newly forming bud tip.
The pathway required to dephosphorylate Sec4p remains to be definitively identified. Previous work suggests that PP2A in complex with Cdc55p is a Sec4p phosphatase (Heger et al., 2011), and cdc55Δ cells have elongated buds as a result of excessive polarized exocytosis (Healy et al., 1991). We found that cdc55Δ cells do not have significant changes in Sec4p phosphorylation levels (Fig. S3). However, this result is difficult to interpret, as cdc55Δ cells show decreased Sec4p bud neck localization that could offset any expected rise in Sec4p phosphorylation caused by a lack of Cdc55p. Multiple phosphatases are known to regulate mitotic exit; diversity in this pathway and coordination with other cellular processes may also influence cell size and shape in other systems (Cundell et al., 2013).
Several interesting possibilities suggest themselves to explain the physiological role of Sec4p phosphorylation at the neck during cytokinesis. In animal cells, there is a specific requirement for the exocyst complex during cytokinesis (Neto et al., 2013), and this complex also participates in membrane delivery at the site of division in S. cerevisiae (Novick et al., 2006; Heider and Munson, 2012). Because the exocyst provides a link between cell-surface delivery and division, it represents an ideal nexus for the integration of different physiological signals (Lipschutz et al., 2000; Wiederkehr et al., 2003; Bodemann et al., 2011), some of which are already known to be subject to phosphorylation control (Macé et al., 2005). We show that cell cycle–dependent phosphorylation of Sec4p reduces the affinity for the exocyst component Sec15p and thus provides an intervention point for the integration of post-Golgi vesicle traffic and membrane tethering with cytokinesis (Fig. 7). Additionally, Cdc42p, which (like Sec4p) preferentially interacts with the exocyst complex in its GTP-bound state (Wu et al., 2010), is inhibited in a Cdc5p-dependent mechanism during mitotic exit (Atkins et al., 2013). Cdc42p interacts with exocyst subunit Exo70p when Cdc42p is directly associated with the plasma membrane. The Cdc5p-dependent inhibition of Cdc42p has also been shown to be important for proper cytokinesis and would affect the ability of the exocyst to tether vesicles to the plasma membrane.
Cartoon model for Sec4p phosphorylation by Cdc5p during cytokinesis. As the cell prepares for cytokinesis at the end of mitosis, Cdc5p kinase phosphorylates Sec4p on secretory vesicles at the site of future cell division. This phosphorylation event will block interaction with Sec15p, the downstream effector, and halt the addition of new plasma membrane at this location. Plasma membrane traffic is not halted at other locations so the cells can continue to enlarge during this period. As cytokinesis progresses, Sec4p dephosphorylation will facilitate exocyst function and allow the cells to form the new plasma membrane at the site of cytokinesis.
Cartoon model for Sec4p phosphorylation by Cdc5p during cytokinesis. As the cell prepares for cytokinesis at the end of mitosis, Cdc5p kinase phosphorylates Sec4p on secretory vesicles at the site of future cell division. This phosphorylation event will block interaction with Sec15p, the downstream effector, and halt the addition of new plasma membrane at this location. Plasma membrane traffic is not halted at other locations so the cells can continue to enlarge during this period. As cytokinesis progresses, Sec4p dephosphorylation will facilitate exocyst function and allow the cells to form the new plasma membrane at the site of cytokinesis.
General disruptions to membrane trafficking, particularly endocytosis, have been noted to occur during mitosis (Warren et al., 1984; Fielding et al., 2012). Phosphorylation of the exocyst component Exo84 by Cdc28p has been shown to cause dissociation of the exocyst complex (Luo et al., 2013). Exo84 phosphorylation occurs as early as metaphase, whereas Sec4p phosphorylation was found to occur after release from metaphase, suggesting that Exo84 phosphorylation to inhibit cell growth happens slightly before Sec4p phosphorylation. Furthermore, Cdc28p, which phosphorylates Exo84p, also phosphorylates Cdc5p, leading to its activation, and may link Sec4p and Exo84p phosphorylation (Simpson-Lavy and Brandeis, 2011).
Rab phosphorylation in general may provide a convenient mechanism to coordinate membrane trafficking with cell cycle changes. Indeed, Rab1 and Rab4 have previously been shown to be preferentially phosphorylated during mitosis (Bailly et al., 1991). Further work to characterize the phosphorylation status of other Rab proteins is needed to address the true functional significance of Rab phosphorylation during mitosis and to demonstrate such modifications represent a regulatory mechanism by which cell cycle signaling pathways can impinge upon critical membrane-trafficking pathways that remodel both organelle shape and distribution inside of cells and the surface that regulates the shape and size that cells present to the exterior.
Materials and methods
Yeast strains, media, and reagents
The S. cerevisiae strains and plasmids used in this study are listed in Tables 1and2, respectively, and were created using standard manipulations. For Western blot analysis, gels were transferred to polyvinylidene difluoride Immobilon membrane (EMD Millipore) before probing with antibodies including anti-GFP antibody (ab6556; Abcam), pS8 antibody (21st Century Biochemicals), and anti-Sec4p (this study). Blots were subsequently incubated with the appropriate secondary antibody coupled to alkaline phosphatase, imaged using CDP-Star chemiluminescence reagent (PerkinElmer), and recorded with an LAS3000 (Fujifilm).
Strain list
| Strain . | Genotype . | Source . |
|---|---|---|
| NY605 | MATa ura3-52 leu2-3,112 | Novick laboratorya |
| RCY1507 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [YCP50 SEC4] | Calero et al., 2003 |
| CUY318 | MATa ade1 ade2 lys2 tyr1 gal1 his7 cdc5ts | Huffaker laboratoryb |
| RCY3343 | MATa ura3-52 leu2-3,112 his3Δ0 | Collins laboratoryc |
| RCY3350 | MATa ura3Δ0 leu2-3,112 cdc55ΔKANR | Heger et al., 2011 |
| RCY5066 | MATa ura3-52 leu2-3,112 his3Δ0 BAR1ΔHIS3 | This study |
| RCY5009 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 MBP-SEC4] | This study |
| RCY5010 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 MBP-SEC4 S8,10,11,201,204A] | This study |
| RCY5085A | MATa ura3-52 leu2-3,112 his3Δ0 sch9ΔHIS3 | This study |
| RCY5090 | MATα his3Δ0 leu2Δ0 ura3Δ0 env7ΔKANR | Resgend |
| RCY5091 | MATα his3Δ0 leu2Δ0 ura3Δ0 sky1ΔKANR | Resgen |
| RCY5092 | MATα his3Δ0 leu2Δ0 ura3Δ0 pkh3ΔKANR | Resgen |
| RCY5081 | MATα his3Δ0 leu2Δ0 ura3Δ0 sak1ΔKANR | Resgen |
| RCY5093 | MATα his3Δ0 leu2Δ0 ura3Δ0 slt2ΔKANR | Resgen |
| RCY5094 | MATα his3Δ0 leu2Δ0 ura3Δ0 psk2ΔKANR | Resgen |
| RCY5095 | MATα his3Δ0 leu2Δ0 ura3Δ0 ste11ΔKANR | Resgen |
| RCY5096 | MATα his3Δ0 leu2Δ0 ura3Δ0 vps15ΔKANR | Resgen |
| RCY4607 | MATα his3Δ0 leu2Δ0 ura3Δ0 kcc4KANR | Resgen |
| RCY5097 | MATα his3Δ0 leu2Δ0 ura3Δ0 bub1ΔKANR | Resgen |
| RCY5130 | MATa ura3-52 leu2-3,112 his3Δ200 hof1ΔKANR SEC4ΔHIS3 [YCP50 SEC4] | This study |
| RCY5126 | MATa ura3-52 ade2-101 his3Δ200 myo1ΔLEU2 SEC4ΔHIS3 [YCP50 SEC4] | This study |
| RCY5120 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 SEC4 S2, 8, 10, 11, 201, 204A; T6A] | This study |
| Strain . | Genotype . | Source . |
|---|---|---|
| NY605 | MATa ura3-52 leu2-3,112 | Novick laboratorya |
| RCY1507 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [YCP50 SEC4] | Calero et al., 2003 |
| CUY318 | MATa ade1 ade2 lys2 tyr1 gal1 his7 cdc5ts | Huffaker laboratoryb |
| RCY3343 | MATa ura3-52 leu2-3,112 his3Δ0 | Collins laboratoryc |
| RCY3350 | MATa ura3Δ0 leu2-3,112 cdc55ΔKANR | Heger et al., 2011 |
| RCY5066 | MATa ura3-52 leu2-3,112 his3Δ0 BAR1ΔHIS3 | This study |
| RCY5009 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 MBP-SEC4] | This study |
| RCY5010 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 MBP-SEC4 S8,10,11,201,204A] | This study |
| RCY5085A | MATa ura3-52 leu2-3,112 his3Δ0 sch9ΔHIS3 | This study |
| RCY5090 | MATα his3Δ0 leu2Δ0 ura3Δ0 env7ΔKANR | Resgend |
| RCY5091 | MATα his3Δ0 leu2Δ0 ura3Δ0 sky1ΔKANR | Resgen |
| RCY5092 | MATα his3Δ0 leu2Δ0 ura3Δ0 pkh3ΔKANR | Resgen |
| RCY5081 | MATα his3Δ0 leu2Δ0 ura3Δ0 sak1ΔKANR | Resgen |
| RCY5093 | MATα his3Δ0 leu2Δ0 ura3Δ0 slt2ΔKANR | Resgen |
| RCY5094 | MATα his3Δ0 leu2Δ0 ura3Δ0 psk2ΔKANR | Resgen |
| RCY5095 | MATα his3Δ0 leu2Δ0 ura3Δ0 ste11ΔKANR | Resgen |
| RCY5096 | MATα his3Δ0 leu2Δ0 ura3Δ0 vps15ΔKANR | Resgen |
| RCY4607 | MATα his3Δ0 leu2Δ0 ura3Δ0 kcc4KANR | Resgen |
| RCY5097 | MATα his3Δ0 leu2Δ0 ura3Δ0 bub1ΔKANR | Resgen |
| RCY5130 | MATa ura3-52 leu2-3,112 his3Δ200 hof1ΔKANR SEC4ΔHIS3 [YCP50 SEC4] | This study |
| RCY5126 | MATa ura3-52 ade2-101 his3Δ200 myo1ΔLEU2 SEC4ΔHIS3 [YCP50 SEC4] | This study |
| RCY5120 | MATα ura3-52 leu2-3,112 his3Δ200 SEC4ΔHIS3 [pRS315 SEC4 S2, 8, 10, 11, 201, 204A; T6A] | This study |
Novick Laboratory, University of California, San Diego, San Diego, CA.
Huffaker Laboratory, Cornell University, Ithaca, NY.
Collins Laboratory, Cornell University, Ithaca, NY.
Resgen haploid delete collection: http://www-sequence.stanford.edu/group/yeast_deletion_project/.
Antibody testing
For characterization of pS8 antibody, 100 µg of synthetic peptides was loaded onto a nitrocellulose membrane using a dot blot apparatus (catalog no. 170–6545; Bio-Dot; Bio-Rad Laboratories). The membrane was probed with the pS8 antibody at a 1:1,000 dilution and a 1:2,500 goat anti–rabbit Ig HRP secondary before ECL detection and imaging.
Nutrient starvation and rapamycin treatments
For nutrient-starvation assays, one culture of cells (RCY3651) was grown overnight, split into four different media conditions with a starting OD600 of 0.3, then allowed to grow for 3 h at 25°C. Yeast extract peptone (YP) medium with 2% glucose, 2% sucrose, or 2% glycerol was used to compare different carbon sources. SD-N (0.17% yeast nitrogen base without amino acids and 2% glucose) was used to observe the effects of nitrogen starvation. Rapamycin was obtained from EMD Millipore (553210).
Protein purification
The protocol for Cdc5p purification was adapted from previous published studies (St-Pierre et al., 2009). In brief, 6X-His-SUMO–tagged Cdc5p under a GAL1/10 promoter (pRC5463A) was expressed in 2 liters of yeast culture, pelleted at 3,000 g for 10 min, resuspended in 30 mM KPO4, 0.5 M NaCl, 10 mM Tris, pH 8, 5% glycerol, 1 mM PMSF, and 5 mM β-glycerol phosphate, and lysed using glass beads. Cdc5p was purified using PrepEASE agarose-conjugated Ni resin (Affymetrix) and eluting with 250 mM imidazole. MBP-Sec4p (both wild-type and Ala mutants) for pS8 antibody characterization was purified from 50 ml RCY5009 and RCY5010. Cells were harvested by glass-bead lysis into 10 mM Tris, pH 7.5, 10 mM NaN3, 1 mM PMSF, 1 mM benzamidine, 0.2 mM DTT, and 1 mM MgCl2. MBP-Sec4p was purified using amylose resin (E8021L; New England Biolabs, Inc.) and eluted into 10 mM maltose, 100 mM NaCl, 50 mM Tris, pH 9.5, 1 mM MgCl2, and 0.2 mM DTT. Phosphatase treatment was performed by adding 20 units of Calf Intestinal Alkaline Phosphatase (M0290S; New England Biolabs, Inc.) and incubating samples for 1 h at 37°C.
Membrane fractionation
MBP-Sec4p wild-type and alanine mutants were grown to mid-log phase OD, and 15 OD units of cells were lysed using glass beads into 10 mM Tris, pH 7.5, 10 mM NaN3, 200 mM sorbitol, 1 mM PMSF, 1 mM benzamidine, 0.2 mM DTT, 5 mM β-glycerol phosphate, 50 mM NaF, and 2 mM EDTA. Samples were clarified for 10 min at 4°C and 10,000 g. The supernatant was then centrifuged for 1 h at 100,000 g to separate the insoluble membrane fraction from the cytosolic fraction. The supernatant was removed, and the membrane was resuspended in the original lysis buffer before preparation for Western blot.
Fluorescence microscopy
An Eclipse E600 microscope (Nikon) was used for fluorescence and differential interference contrast (DIC) microscopy equipped with 1× optivar (0.08 µm/pixel), 60× Oil objective (1.4 numerical aperture), and 10×/25 eye piece (CFI UW; Nikon), and imaged with a Clara CCD camera (DR-328G-C01-SIL; Andor Technology). A C-FL GFP HC HISN zero shift filter set and C-FL Texas Red HC HISN zero shift filter set were used for GFP and mCherry image acquisition, respectively. Images were taken as a series of z-stacks in 0.6-µm increments over 8 µm using NIS-Elements Advanced Research imaging software. Images were deconvolved using AutoQuant X software. For heat-shock experiments and cell-synchrony and release experiments, cells were fixed in 4% PFA for 30 min before washes and resuspension in PBS for imaging. Cdc5p and Sec4p colocalization experiments were performed on live cells.
Kinase overexpression library preparation and screen
127 yeast kinases were individually cloned into a pRS426 multi-copy 2µ plasmid using homologous recombination. The endogenous promoter and terminator were included for all kinases (except KNS1 and PBS2). A full list of library kinases can be found in Table S1. Plasmids were transformed into the yeast strain NY605, and then single colonies were picked and grown up in SCD-Ura growth medium overnight (Table 2). Samples were harvested in mid-log phase, and 10 OD units of cells were spun down, washed in TAZ buffer, 10 mM Tris, pH 7.5, and 10 mM NaN3, and then resuspended in 10 mM Tris, pH 7.5, 10 mM NaN3, 200 mM sorbitol, 1 mM PMSF, 1 mM benzamidine, 0.2 mM DTT, 5 mM β-glycerol phosphate, 50 mM NaF, and 2 mM EDTA for glass-bead lysis.
Plasmid list
| Plasmid identification no. . | Description . | Source . |
|---|---|---|
| pRC5316 | MBP-Sec4p/pRS315 | This study |
| pRC5313D | MBP-Sec4p S8,10,11,201,204A/pRS315 | This study |
| pRC1820 | Sec4p/pRS315 | Collins laboratory |
| pRC3423 | Sec4p Q79L/pRS315 | Heger et al., 2011 |
| pRC5475 | Sec4p S29V/pRS315 | This study |
| pRC5464A | Sec4p S10,11A/pRS315 | This study |
| pRC5463A | ppSUMO Cdc5p/pRS426 | This study |
| pRC651 | GFP-Sec4p/pRS315 | Collins laboratory |
| pRC3000 | GFP-Sec4p S8,11,201,204D/pRS315 | Heger et al., 2011 |
| pRC5473 | GFP-Sec4p S8,10,11,201,204D/pRS315 | This study |
| pRC5472 | GFP-Sec4p S8,10,11,201,204A/pRS315 | This study |
| pRC5474 | Sec15p-MBP/pRS426 | This study |
| pRC5460 | Cdc5p-3xmCherry/pRS316 | This study |
| pRC5468 | SEC4 S2,8,10,11,201,204A T6A/pRS315 | This study |
| Plasmid identification no. . | Description . | Source . |
|---|---|---|
| pRC5316 | MBP-Sec4p/pRS315 | This study |
| pRC5313D | MBP-Sec4p S8,10,11,201,204A/pRS315 | This study |
| pRC1820 | Sec4p/pRS315 | Collins laboratory |
| pRC3423 | Sec4p Q79L/pRS315 | Heger et al., 2011 |
| pRC5475 | Sec4p S29V/pRS315 | This study |
| pRC5464A | Sec4p S10,11A/pRS315 | This study |
| pRC5463A | ppSUMO Cdc5p/pRS426 | This study |
| pRC651 | GFP-Sec4p/pRS315 | Collins laboratory |
| pRC3000 | GFP-Sec4p S8,11,201,204D/pRS315 | Heger et al., 2011 |
| pRC5473 | GFP-Sec4p S8,10,11,201,204D/pRS315 | This study |
| pRC5472 | GFP-Sec4p S8,10,11,201,204A/pRS315 | This study |
| pRC5474 | Sec15p-MBP/pRS426 | This study |
| pRC5460 | Cdc5p-3xmCherry/pRS316 | This study |
| pRC5468 | SEC4 S2,8,10,11,201,204A T6A/pRS315 | This study |
Pulldown assay
Sec15-MBP and GFP-Sec4p wild-type and alleles (all Sec4p mutants expressed ectopically) were expressed in RCY3651 cells and grown in yeast peptone dextrose (YPD) to mid-log phase. 200 ml of culture was harvested, lysed with glass beads in 10 mM Tris, pH 7.5, 10 mM NaN3, 1 mM PMSF, 1 mM benzamidine, 0.2 mM DTT, and 1 mM MgCl2, 5 mM β-glycerol phosphate, and 50 mM NaF lysis buffer. Triton X-100 was added to a final concentration of 1% to solubilize membranes. Cell lysate was clarified at 2,000 g for 30 min, and then Sec15-MBP was purified using amylose resin (E8021L; New England Biolabs, Inc.) and eluted directly from resin with SDS-PAGE sample buffer containing DTT. GFP-Sec4p was probed for via Western blot with anti-GFP antibody (Ab6556; Abcam).
In vitro kinase assay
For each reaction, samples were incubated in 15 mM KPO4, 5 mM Tris, pH 8, 250 mM NaCl, 2.5% glycerol, 10 mM MgCl2, and 200 µM ATP. 2 µg of substrate peptides and 100 µl Cdc5p purification prep were added, and reactions were brought to a final volume of 200 µl. Reactions were started with the addition of Cdc5p and incubated for 1 h at 30°C. Aliquots were then assayed on nitrocellulose using Dot-Blot apparatus (catalog no. 170–6545; Bio-Dot; Bio-Rad Laboratories) and pS8 antibody analysis.
Cell cycle synchrony and release
To achieve cell synchrony, cells were treated with either 10 µg/ml nocodazole (Cayman Chemical) or 1 µg/ml α-factor (4003514; Bachem) and grown at 30°C for 2 h. Cells were imaged with DIC microscopy and checked to ensure >95% synchrony (large-budded cells or shmoo formation for nocodazole and α-factor, respectively). Cells were washed and resuspended in prewarmed 30°C YPD to initiate the release. Cells were grown in a 30°C shaker, and 3 OD600 units of cells were taken every 10 min, pelleted, washed in 10 mM Tris, pH 7.5, and 10 mM NaN3, pelleted once again, and snap frozen in liquid nitrogen (for each sample, it took ∼3 min to take cells from incubator until the pellet was snap frozen in liquid nitrogen). For immunofluorescence preparation, aliquots were taken every 10 min and mixed with 2× fixative (8% PFA). α-Factor synchrony experiments were performed in bar1Δ cells (RCY5066) for efficient synchrony and release. Endogenous Sec4p was monitored for phosphorylation via Western blot in both synchrony experiments, but nocodazole release localization experiments used GFP-Sec4p as the sole copy of Sec4p. α-Factor release experiments monitored endogenous Sec4p localization via immunofluorescence. For immunofluorescence experiments, cells were spheroblasted in 100 mM KPi, pH 7.5, 1.2 M sorbitol, zymolyase, and 0.2% 2-mercaptoethanol for 30 min, permeabilized with 0.1% SDS, and blocked in 10% goat serum PBS (30 min). Sec4p was detected using 1:500 mouse anti-Sec4p (generated in collaboration with Whittaker laboratory) and a goat anti–mouse Alexa Fluor 488 secondary.
Online supplemental material
Fig. S1 shows a summary of the kinase overexpression screen with each point representing the mean relative deviation of two independent measurements of pS8 levels in a specific kinase overexpression background from a vector-only control. Fig. S2 shows a Western blot analysis of pS8 and Sec4p levels in the membrane-trafficking temperature-sensitive strains after 1-h shift to 37°C. Fig. S3 shows a quantitative Western blot analysis of pS8 and Sec4p levels in wild-type and cdc55Δ cells from three independent samples, with error bars showing SD. Fig. S4 shows tetrad dissection and genetic analysis of haploid spores from a cdc5ts strain crossed with sec4-8 or sec15-1. Table S1 provides an analysis of the pS8/Sec4p ratio for kinase overexpression screen. Each number represents the relative deviation of the ratio pS8/Sec4p levels from a vector-only control as measured by quantification of Western blot data.
Acknowledgments
We thank the Whittaker laboratory for help in designing and isolating pan Sec4p antibodies, as well as the Huffaker laboratory for providing yeast strains.
This work was supported by National Institutes of Health Predoctoral Training in Cellular and Molecular Biology (5T32GM007273) and by a grant from the National Institutes of Health (5R01GM069596) to R.N. Collins.
The authors declare no competing financial interests.