Septin complexes display remarkable plasticity in subunit composition, yet how a new subunit assembled into higher-order structures confers different functions is not fully understood. Here, this question is addressed in budding yeast, where during meiosis Spr3 and Spr28 replace the mitotic septin subunits Cdc12 and Cdc11 (and Shs1), respectively. In vitro, the sole stable complex that contains both meiosis-specific septins is a linear Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 hetero-octamer. Only coexpressed Spr3 and Spr28 colocalize with Cdc3 and Cdc10 in mitotic cells, indicating that incorporation requires a Spr28-Spr3 protomer. Unlike their mitotic counterparts, Spr28-Spr3–capped rods are unable to form higher-order structures in solution but assemble to form long paired filaments on lipid monolayers containing phosphatidylinositol-4,5-bisphosphate, mimicking presence of this phosphoinositide in the prospore membrane. Spr28 and Spr3 fail to rescue the lethality of a cdc11Δ cdc12Δ mutant, and Cdc11 and Cdc12 fail to restore sporulation proficiency to spr3Δ/spr3Δ spr28Δ/spr28Δ diploids. Thus, specific meiotic and mitotic subunits endow septin complexes with functionally distinct properties.
Introduction
Septins are a family of GTP-binding proteins conserved in all eukaryotes (except higher plants; Pan et al., 2007; Nishihama et al., 2011). In organisms as evolutionarily distant as yeast and humans, septins assemble into a linear hetero-octameric complex composed of four different monomers arranged with twofold rotational symmetry (Bertin et al., 2008; Kim et al., 2011; Sellin et al., 2012). The resulting apolar rods can self-associate into long filaments and other, more complex higher-order structures. However, the genomes of yeast and humans encode, respectively, seven and thirteen different septins, raising important questions about the number of allowed combinatorial arrangements of these monomers and their respective physiological functions. Moreover, how are certain combinations favored over others when potentially redundant subunits are coexpressed? As we document here, the assembly properties and roles of a development-specific septin complex in yeast now provide important new insights that address these questions. This unique complex is formed during yeast meiosis and sporulation, a process closely akin to mammalian gametogenesis.
On a poor carbon source and limited nitrogen supply, a diploid (MATa/MATα) Saccharomyces cerevisiae cell undergoes meiosis within its own cytoplasm. The resulting four haploid nuclei are encased into spores, surrounded by the old cell wall (ascus; Fowell, 1969; Neiman, 2011). In this process, the nuclear envelope is remodeled, forming four lobes. Each lobe directs assembly of a closely allied membrane (the prospore membrane [PSM]) that becomes the spore plasma membrane, on which are deposited the spore wall and other protective coatings (Maier et al., 2007; Morishita and Engebrecht, 2008). The PSM assembles de novo from vesicles that dock and fuse, initially forming a cup-like cap above each nuclear lobe that expands and engulfs each incipient haploid nucleus (Moens, 1971; Riedel et al., 2005; Nakanishi et al., 2006). A septin-based structure is tightly associated with the developing PSM (De Virgilio et al., 1996; Fares et al., 1996; Pablo-Hernando et al., 2008).
In mitotic cells, five septins are expressed and assemble into two complexes differing only in the terminal subunit: Cdc11–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Cdc11 and Shs1–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Shs1. Cdc11-capped rods polymerize end-on-end into straight paired filaments when the salt concentration <150 mM (Bertin et al., 2008; Booth et al., 2015), whereas, under the same conditions, Shs1-capped rods associate laterally, not end to end (Booth et al., 2015), to form spirals and rings (Garcia et al., 2011). In meiotic cells, two new septins, Spr3 (Ozsarac et al., 1995; Fares et al., 1996) and Spr28 (De Virgilio et al., 1996), are produced (Brar et al., 2012). At the transcriptional level, SPR3, SPR28, CDC3, and CDC10 are induced during meiosis, whereas CDC11 and CDC12 are not (Kaback and Feldberg, 1985; Chu et al., 1998), and SHS1 is repressed (Friedlander et al., 2006). These findings are consistent with a model (McMurray and Thorner, 2008) in which, during meiosis, Cdc11 (and Shs1) and Cdc12 are replaced by Spr28 and Spr3, a pair of potentially interacting subunits, thereby generating a novel hetero-octameric complex unique to sporulating cells.
During sporulation, Spr3, Spr28, Cdc3, and Cdc10 are prominently localized to the PSM, and Cdc11 is detectable (Fares et al., 1996; Pablo-Hernando et al., 2008), whereas the bulk of Cdc12 and Shs1 are excluded from septin structures at the PSM (Douglas et al., 2005; McMurray and Thorner, 2008, 2009). Septins appear first on the nuclear-proximal side of the initial PSM. As the PSM cup expands, a U-shaped septin structure (“horseshoe”) forms, whose arms elongate as the PSM closes. After its closure, septins are distributed more evenly on the cytoplasmic face of the spore plasma membrane (Fares et al., 1996; Neiman, 2011).
In an spr28Δ/spr28Δ diploid, the horseshoe does not form and the other septins are dispersed over the PSM surface (Pablo-Hernando et al., 2008). In an spr3Δ/spr3Δ diploid, the horseshoe is also eliminated and association of other septins with the PSM is greatly reduced (Fares et al., 1996; Pablo-Hernando et al., 2008). Despite these drastic perturbations of normal meiotic septin organization, loss of Spr3 (Kao et al., 1989; Fares et al., 1996) or Spr28 (De Virgilio et al., 1996), or both (Fares et al., 1996; A. Neiman, personal communication), reportedly caused little, if any, reduction in spore formation.
In this study, we sought to determine how Spr3 and Spr28 contribute to overall septin architecture at the ultrastructural level and to examine both in vitro and in vivo the biochemical and biophysical properties of the complexes that contain them, especially their interaction with membranes. We also reinvestigated the phenotype of cells lacking Spr3 and Spr28 and found, contrary to prior studies, that absence of either of these septins markedly reduces sporulation proficiency, as documented here, as well as compromises the structural integrity of the spores that do manage to form, as described in detail elsewhere (Heasley and McMurray, 2016). Our findings provide novel insights about how alternative subunits endow septin complexes with unique properties.
Results
Expression and purification of recombinant septin complexes containing Spr3 and Spr28
In meiotic cells, Spr3 might replace Cdc12 and Spr28 might replace Cdc11 and Shs1, the subunits they most resemble (Fig. S1 and Table S1), thereby generating a unique septin complex with properties specific for execution of sporulation (Fig. 1 A). Analogously, as documented before (Garcia et al., 2011), in mitotic cells, Shs1 competes with and can substitute for Cdc11, its closest paralogue (Fig. S1 and Table S1), yet it accords very different properties on the resulting hetero-octamers.
The sporulation-specific septin complex is a hetero-octameric rod. (A) Model for organization of the sporulation-specific septin complex and its relationship to mitotic septin complexes. Adjacent monomers in linear septin complexes interact by two alternating contact modes, a G interface (involving residues in and around the GTP-binding pockets), and an NC interface (involving residues in and around the N- and C-terminal segments of the GTP-binding domains). Short side, G interface; long side, NC interface; wavy line, CTE; squiggle, coiled coil. (B) Septins were coexpressed in E. coli, the resulting (His)6Spr3-containing complexes were purified as described in Materials and methods, and proteins in the final purified fraction were resolved by SDS-PAGE and visualized by staining with Coomassie blue dye. (left) MM, molecular mass standards; lane 1, Cdc3 and Cdc10 coexpressed with (His)6Spr3; lane 2, Cdc3, Cdc10, and Spr28 coexpressed with (His)6Spr3. (right) Lane 1, Cdc3, Cdc10, and Cdc11 coexpressed with (His)6Cdc12 (mitotic septin complexes); lane 2, Cdc3, Cdc10, and Cdc11 coexpressed with (His)6Spr3; lane 3, Cdc3, Cdc10, Cdc11, and Spr28 coexpressed with (His)6Spr3. (C) Analytical scale size-exclusion chromatography of septin complexes. Cyan, Cdc11(Δα0)-(His)6Cdc12–Cdc3–Cdc10 hetero-octamers; the Δα0 mutation does not prevent rod assembly but blocks its end-to-end polymerization in solution (Bertin et al., 2008); gray, Shs1(Δα0)-(His)6Cdc12–Cdc3–Cdc10 complexes, which represent a mixture of octamers, heptamers, and hexamers (Garcia et al., 2011); green and dashed, Spr28– (His)6Spr3–Cdc3–Cdc10 complex; purple, (His)6Spr3-Cdc3-Cdc10 complex; and black, (His)6Cdc12–Cdc3–Cdc10 complexes, which are stable hetero-hexamers (Bertin et al., 2008). Slightly earlier elution of the (His)6Spr3-Cdc3-Cdc10 complex versus the (His)6Cdc12–Cdc3–Cdc10 complex is attributable to the higher molecular mass of Spr3 (59.8 kD) compared with Cdc12 (46.7 kD). EM images of the complexes from B, left, lane 1 (D) and lane 2 (E) in high-salt buffer, stained with uranyl formate. Globular particles are a contaminant, endogenous E. coli ArnA (formerly PmrI), a 70 kD Ni2+-binding polypeptide that copurifies with septin complexes to a variable extent from preparation to preparation because it forms two stacked trimers whose molecular mass is close to that of septin complexes (which, because of their rod shape, elute at a larger apparent size and are not well resolved from the ArnA homohexamer). Bar, 100 nm. (F) Four representative class averages for the (His)6Spr3–Cdc3–Cdc10 complex in high salt. Red arrows show presence of partially unfolded or highly mobile Spr3 indicated by the extra density at the ends of the Cdc3–Cdc10–Cdc10–Cdc3 hetero-tetrameric rods. Bar, 10 nm. (G) Four representative class averages for the Spr28– (His)6Spr3–Cdc3–Cdc10 complex in high salt. Yellow arrows, extra lateral densities representing either a coiled-coil interaction between the CTEs of Spr3 and Cdc3 or the N-terminal domain of Spr3. Bar, 10 nm.
The sporulation-specific septin complex is a hetero-octameric rod. (A) Model for organization of the sporulation-specific septin complex and its relationship to mitotic septin complexes. Adjacent monomers in linear septin complexes interact by two alternating contact modes, a G interface (involving residues in and around the GTP-binding pockets), and an NC interface (involving residues in and around the N- and C-terminal segments of the GTP-binding domains). Short side, G interface; long side, NC interface; wavy line, CTE; squiggle, coiled coil. (B) Septins were coexpressed in E. coli, the resulting (His)6Spr3-containing complexes were purified as described in Materials and methods, and proteins in the final purified fraction were resolved by SDS-PAGE and visualized by staining with Coomassie blue dye. (left) MM, molecular mass standards; lane 1, Cdc3 and Cdc10 coexpressed with (His)6Spr3; lane 2, Cdc3, Cdc10, and Spr28 coexpressed with (His)6Spr3. (right) Lane 1, Cdc3, Cdc10, and Cdc11 coexpressed with (His)6Cdc12 (mitotic septin complexes); lane 2, Cdc3, Cdc10, and Cdc11 coexpressed with (His)6Spr3; lane 3, Cdc3, Cdc10, Cdc11, and Spr28 coexpressed with (His)6Spr3. (C) Analytical scale size-exclusion chromatography of septin complexes. Cyan, Cdc11(Δα0)-(His)6Cdc12–Cdc3–Cdc10 hetero-octamers; the Δα0 mutation does not prevent rod assembly but blocks its end-to-end polymerization in solution (Bertin et al., 2008); gray, Shs1(Δα0)-(His)6Cdc12–Cdc3–Cdc10 complexes, which represent a mixture of octamers, heptamers, and hexamers (Garcia et al., 2011); green and dashed, Spr28– (His)6Spr3–Cdc3–Cdc10 complex; purple, (His)6Spr3-Cdc3-Cdc10 complex; and black, (His)6Cdc12–Cdc3–Cdc10 complexes, which are stable hetero-hexamers (Bertin et al., 2008). Slightly earlier elution of the (His)6Spr3-Cdc3-Cdc10 complex versus the (His)6Cdc12–Cdc3–Cdc10 complex is attributable to the higher molecular mass of Spr3 (59.8 kD) compared with Cdc12 (46.7 kD). EM images of the complexes from B, left, lane 1 (D) and lane 2 (E) in high-salt buffer, stained with uranyl formate. Globular particles are a contaminant, endogenous E. coli ArnA (formerly PmrI), a 70 kD Ni2+-binding polypeptide that copurifies with septin complexes to a variable extent from preparation to preparation because it forms two stacked trimers whose molecular mass is close to that of septin complexes (which, because of their rod shape, elute at a larger apparent size and are not well resolved from the ArnA homohexamer). Bar, 100 nm. (F) Four representative class averages for the (His)6Spr3–Cdc3–Cdc10 complex in high salt. Red arrows show presence of partially unfolded or highly mobile Spr3 indicated by the extra density at the ends of the Cdc3–Cdc10–Cdc10–Cdc3 hetero-tetrameric rods. Bar, 10 nm. (G) Four representative class averages for the Spr28– (His)6Spr3–Cdc3–Cdc10 complex in high salt. Yellow arrows, extra lateral densities representing either a coiled-coil interaction between the CTEs of Spr3 and Cdc3 or the N-terminal domain of Spr3. Bar, 10 nm.
To ascertain whether Spr3 and Spr28 possess an intrinsic ability to replace their mitotic counterparts, we expressed the sporulation-specific septins as recombinant proteins in bacterial cells alone, together, and with various combinations of the other five septin subunits. Expression of (His)6Cdc12 and untagged versions of Cdc3, Cdc10, and Cdc11 (or Shs1) reproducibly yields stoichiometric complexes of the mitotic septins because Cdc12 is the limiting subunit. Hence, we used (His)6Spr3 in the same way because of its resemblance to Cdc12 and the existing evidence that Spr3 may displace Cdc12 in meiotic cells (McMurray and Thorner, 2008). When (His)6Spr3 was coexpressed with Cdc3 and Cdc10, the three proteins consistently copurified in a stoichiometric complex even in high salt (Fig. 1 B, left, lane 1). When examined by size-exclusion chromatography, the particles in such preparations eluted very similarly to a known septin hetero-hexamer (Fig. 1 C). However, when diluted, dispersed on carbon-coated grids, and viewed under EM (Fig. 1 D), only a minority of the observed rods were hetero-hexamers (mainly pentamers and tetramers were found), indicating that Spr3 had dissociated from one or both ends, suggesting that the Spr3–Cdc3 junction is not very stable.
Next, we coexpressed Spr28 with (His)6Spr3, Cdc3 and Cdc10 and found that these four proteins consistently copurified in a stoichiometric complex even in high salt (Fig. 1 B, left, lane 2). When viewed using EM (Fig. 1 E), or when examined by size-exclusion chromatography against appropriate standards (Fig. 1 C), rods of appropriate length to be hetero-octamers were observed (as well as some of heptamer and hexamer length). By this criterion, presence of Spr28 stabilized the Spr3–Cdc3 interaction. Thus, in vitro, Spr3 and Spr28 together had the capacity to associate with the ends of Cdc3–Cdc10–Cdc10–Cdc3 hetero-tetramers to form Spr28–(His)6Spr3–Cdc3–Cdc10–Cdc10–Cdc3–(His)6Spr3–Spr28 hetero-octamers.
A potential explanation for the observed localization of some Cdc11 on the PSM might be that Spr3 bears sufficient resemblance to Cdc12 that Cdc11–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Cdc11 hetero-octamers are able to form. However, when Cdc11 was coexpressed with (His)6Spr3, Cdc3 and Cdc10, no detectable Cdc11 was incorporated into the resulting complexes, which contained only (His)6Spr3, Cdc3 and Cdc10 (Fig. 1 B, right, lane 2). Another possibility is that Spr28 retains enough similarity to Cdc11 to form a Cdc11–Spr28 junction, in analogy to the Cdc11–Cdc11 interface responsible for the end-to-end polymerization of mitotic Cdc11–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Cdc11 rods (Bertin et al., 2008). However, when Cdc11 was coexpressed with Spr28, (His)6Spr3, Cdc3 and Cdc10, no detectable Cdc11 was incorporated into the complexes, which contained only Spr28, (His)6Spr3, Cdc3, and Cdc10 (Fig. 1 B, right, lane 3). Thus, Cdc11 may be present at the PSM as a monomer (or, perhaps, in residual intact mitotic septin complexes that manage to survive during sporulation).
Subunit architecture in sporulation-specific septin complexes
As an independent means to determine the subunit arrangement in the complexes containing Spr3 and Spr28, we performed single-particle analysis. Large numbers of individual particles on EM grids were sorted into groups (classes) on the basis of shared distinctive features. The particles in each class were computationally aligned and averaged to produce a representative image (class average). The class averages of the complexes composed of (His)6Spr3, Cdc3 and Cdc10 were rod-shaped and had no additional density along the sides of the rod (Fig. 1 F). Given the stoichiometric complexes isolated (Fig. 1 B, left, lane 1) and their hydrodynamic behavior (Fig. 1 C), it was unexpected that a majority were tetrameric (Fig. 1 F, right-most panel). There were, however, classes that appeared pentameric (Fig. 1 F, middle two panels) and hexameric (Fig. 1 F, left-most panel) because they contained additional, albeit weak, density at one or both ends the rod. This behavior indicates that association of Spr3 with Cdc3 is relatively weak, that the terminal Spr3 molecules unfold relatively easily, or that Spr3 is attached via a linkage that allows for significant flexibility. In analogy to the Cdc12–Cdc3 interaction, Spr3 likely associates with Cdc3 via both its globular domain and via formation of a coiled coil between its C-terminal extension (CTE) and that of Cdc3 (Fig. 1 C). If the latter interaction is stronger and more mobile, it might explain the relatively weak end densities observed.
Class averages of complexes composed of Spr28, (His)6Spr3, Cdc3 and Cdc10 were also rod-shaped (Fig. 1 G), and the majority were clearly octameric (Fig. 1 G, left-most two panels); out of 1,990 total rods counted, 972 (49%) were octamers, 335 (17%) heptamers, and 683 (34%) hexamers. In the octamers, the penultimate protomer displayed a density as prominent as any other subunit, consistent with stabilization of Spr3 via its association with Spr28. The octamers contained a conspicuous density located between the second and third subunits (Fig. 1 G, left two panels) and situated on the same side of the rod. The CTEs at each NC interface in mammalian septin rods form coiled coils (de Almeida Marques et al., 2012) and, in their native state, must extend from the same side of the rod (Sirajuddin et al., 2007). Thus, the observed densities could represent a coiled coil between the CTEs of Spr3 and Cdc3 (Fig. 1 C). Alternately, this extra density could represent a stable fold adopted by the unique N-terminal domain in Spr3, which is 75 residues longer than that in Cdc12 (Fig. 1 A). Heptameric (Fig. 1 G, second panel from the right) and hexameric (Fig. 1 G, right-most panel) classes presumably arose from dissociation of Spr28 from one or both ends. Thus, Spr28 occupies the terminal position and Spr3 occupies the penultimate position in the hetero-octamers.
Higher-order assembly of sporulation-specific septin complexes
When diluted from high to low salt and deposited from solution onto EM grids, mitotic Cdc11–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Cdc11 hetero-octamers have polymerized into long paired filaments (Bertin et al., 2008; Fig. 2 A, left). When treated in the same manner, neither Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 hetero-octamers (Fig. 2 B, middle) nor Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3 hetero-hexamers (Fig. 2 A, right) formed any higher-order structure. The lack of observable self-assembly could indicate that sporulation-specific complexes are incapable of self-association or, if a higher-order structure forms, it is very fragile. Alternatively, the solution conditions chosen may not be suitable for interaction, or a molecular partner present in meiotic cells, but absent in our purified preparations, is necessary to promote higher-order assembly.
PtdIns4,5P2 promotes assembly of sporulation-specific septin complexes. (A) The indicated septin complexes were diluted from high-salt buffer into low-salt buffer, incubated for 1 h, deposited on grids, stained with uranyl formate, and viewed by EM. Left, Cdc11–(His)6Cdc12–Cdc3–Cdc10–Cdc10–Cdc3-(His)6Cdc12–Cdc11 (mitotic) hetero-octamers; middle, Spr28– (His)6Spr3–Cdc3–Cdc10–Cdc10–Cdc3–(His)6Spr3–Spr28 hetero-octamers; right, (His)6Spr3–Cdc3–Cdc10–Cdc10–Cdc3–(His)6Spr3 hetero-hexamers. (B) The sporulation-specific septin complex (top) and a control mitotic septin complex (bottom) whose polymerization into filaments is promoted on the surface of a PtdIn4,5P2-containing lipid monolayer (Bertin et al., 2010) were diluted into low-salt buffer in a droplet, whose meniscus was overlaid with a solution of a monolayer-forming lipid containing DOPC alone, DOPC containing PtdIns4P, or DOPC containing PtdIns4,5P2, as indicated. Any proteins associated with the headgroups of the lipid monolayer were visualized by capturing its hydrophobic side by binding to a holey carbon-coated grid, staining with uranyl formate, and inspecting the regions of the monolayer that span holes in the grid by EM. Right-most panels, magnified views of the indicated insets. Bar, 100 nm.
PtdIns4,5P2 promotes assembly of sporulation-specific septin complexes. (A) The indicated septin complexes were diluted from high-salt buffer into low-salt buffer, incubated for 1 h, deposited on grids, stained with uranyl formate, and viewed by EM. Left, Cdc11–(His)6Cdc12–Cdc3–Cdc10–Cdc10–Cdc3-(His)6Cdc12–Cdc11 (mitotic) hetero-octamers; middle, Spr28– (His)6Spr3–Cdc3–Cdc10–Cdc10–Cdc3–(His)6Spr3–Spr28 hetero-octamers; right, (His)6Spr3–Cdc3–Cdc10–Cdc10–Cdc3–(His)6Spr3 hetero-hexamers. (B) The sporulation-specific septin complex (top) and a control mitotic septin complex (bottom) whose polymerization into filaments is promoted on the surface of a PtdIn4,5P2-containing lipid monolayer (Bertin et al., 2010) were diluted into low-salt buffer in a droplet, whose meniscus was overlaid with a solution of a monolayer-forming lipid containing DOPC alone, DOPC containing PtdIns4P, or DOPC containing PtdIns4,5P2, as indicated. Any proteins associated with the headgroups of the lipid monolayer were visualized by capturing its hydrophobic side by binding to a holey carbon-coated grid, staining with uranyl formate, and inspecting the regions of the monolayer that span holes in the grid by EM. Right-most panels, magnified views of the indicated insets. Bar, 100 nm.
In the latter regard, the PSM in S. cerevisiae is highly enriched in PtdIns4,5P2, and production of this phosphoinositide is essential for sporulation (Rudge et al., 2004; Park and Neiman, 2012). Moreover, presence of PtdIns4,5P2 (and no other phosphoinositide) promotes polymerization of mitotic septin complexes on a lipid monolayer under high-salt conditions that do not permit filament formation in solution (Bertin et al., 2010). Similarly, mitotic septin complexes capped with Cdc11(Δα0), a mutation that weakens the Cdc11–Cdc11 interaction, do not form filaments in low-salt solution (Bertin et al., 2008) but readily form filaments on the surface of the PtdIns4,5P2-containing monolayer (Bertin et al., 2010).
For these reasons, we tested whether sporulation-specific septin complexes would display higher-order assembly when confronted with a PtdIns4,5P2-containing monolayer. Control lipid monolayers composed of 1,2-dioleoyl-sn-phosphatidylcholine (DOPC) alone, as well as DOPC doped with 15 mol% PtdIns4P, were unable to recruit either the Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 complex (Fig. 2 B, top) or the Cdc11(Δα0)–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Cdc11(Δα0) complex (Fig. 2 B, bottom) to their surface, even from low salt buffer. In marked contrast, DOPC doped with 15 mol% PtdIns4,5P2 robustly recruited both the sporulation-specific septin complex (Fig. 2 B, top) and the mutant mitotic septin complex (Fig. 2 B, bottom) to the surface and promoted formation of prominent and well-ordered filaments. Enlargement revealed that the filaments generated by the sporulation-specific septin complex are paired and laterally connected by an obvious “rungs-on-a-ladder” cross-bracing (Fig. 2 B, top), whereas many of the filaments generated by the mutant mitotic septin complex are in tight pairs (Fig. 2 B, bottom), as seen before (Bertin et al., 2010). Thus, PtdIns4,5P2 profoundly affected the assembly state of Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 hetero-octamers.
Sporulation-specific septins cannot support mitotic growth
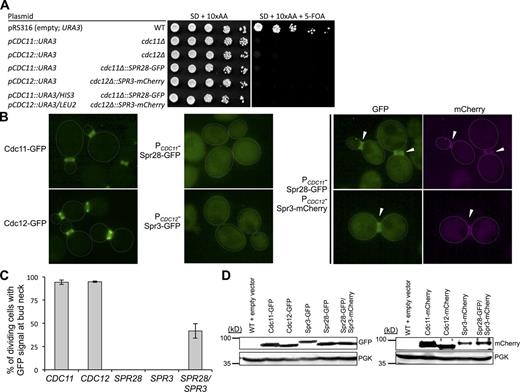
To test whether Spr28 and Spr3 can functionally substitute for Cdc11 and Cdc12, respectively, the sporulation-specific septins (marked with fluorescent tags) were integrated into the genome under control of the CDC11 and CDC12 promoters at their endogenous loci. The strains also carried URA3-marked plasmids that expressed a wild-type copy of the mitotic septin gene that was replaced with its sporulation-specific counterpart. Just like a cdc11Δ mutant, the cells harboring the integrated CDC11prom-SPR28-GFP construct were unable to propagate when the URA3-marked CDC11-expressing plasmid was selected against on medium containing 5-fluoro-orotic acid (5-FOA; Fig. 3 A, second and fourth lanes). Likewise, just like a cdc12Δ mutant, the cells harboring the integrated CDC12prom-SPR3-mCherry construct were unable to grow on 5-FOA medium (Fig. 3 A, third and fifth lanes). This latter finding agrees with the results of Fares et al. (1996) who showed that ectopic expression of Spr3 was unable to rescue growth at the nonpermissive temperature of strains carrying a cdc3ts, cdc10ts, cdc11ts, or cdc12ts allele. In our hands, even presence of both CDC11prom-SPR28-GFP and CDC12prom-SPR3-mCherry was unable to rescue the inviability of a cdc11Δ cdc12Δ double mutant (Fig. 3 A, line 6). Lack of complementation was not caused by lack of expression, as both sporulation-specific septins were stably produced in these cells at levels similar to tagged versions of Cdc11 and Cdc12, as confirmed by visualization of the cells (Fig. 3 B) and by immunoblotting (Fig. 3 D). Thus, the sporulation-specific subunits cannot functionally replace the corresponding mitotic septins.
Spr3 and Spr28 do not functionally substitute for Cdc12 and Cdc11 in vegetative cells and are competent to associate with mitotic septins only when coexpressed. (A) Haploid cells of the indicated genotype (middle column) and containing a URA3-marked plasmid (left column) expressing wild-type CDC11 or wild-type CDC12, or both (to ensure viability) were grown in selective (−Ura) medium and then serially diluted onto control medium (synthetic complete; left) or the same medium containing 5-FOA to select against the URA3-marked covering plasmid (right). Where indicated, sporulation-specific septins SPR28-GFP and SPR3-mCherry were expressed from the CDC11 and CDC12 promoters, respectively, at the corresponding chromosomal loci. (B) Otherwise wild-type cells expressing the indicated fluorescently-tagged septins were grown to mid-exponential phase in YPD and visualized by fluorescence microscopy. Faint dotted white lines, yeast cells periphery; arrowheads, fluorescent signal at the bud neck. Bar, 2 µm. (C) Quantification of B. Hundreds of cells in independent cultures expressing tagged Spr28 alone (n = 3), tagged Spr3 alone (n = 3), or both (n = 5) were examined by fluorescence microscopy. Bars, mean percentage of the budded cells that displayed a visible fluorescent signal at the bud neck; error bar, standard deviation of the mean. (D) To analyze expression of the indicated fluorescently tagged septins in the cells from B (left) and Fig. S1 (right), equivalent numbers of cells were lysed, resolved by SDS-PAGE, transferred to a nitrocellulose filter, and probed with appropriate antibodies (anti-GFP, anti-DsRed, or anti-Pgk1). Pgk1, control for equivalent protein loading.
Spr3 and Spr28 do not functionally substitute for Cdc12 and Cdc11 in vegetative cells and are competent to associate with mitotic septins only when coexpressed. (A) Haploid cells of the indicated genotype (middle column) and containing a URA3-marked plasmid (left column) expressing wild-type CDC11 or wild-type CDC12, or both (to ensure viability) were grown in selective (−Ura) medium and then serially diluted onto control medium (synthetic complete; left) or the same medium containing 5-FOA to select against the URA3-marked covering plasmid (right). Where indicated, sporulation-specific septins SPR28-GFP and SPR3-mCherry were expressed from the CDC11 and CDC12 promoters, respectively, at the corresponding chromosomal loci. (B) Otherwise wild-type cells expressing the indicated fluorescently-tagged septins were grown to mid-exponential phase in YPD and visualized by fluorescence microscopy. Faint dotted white lines, yeast cells periphery; arrowheads, fluorescent signal at the bud neck. Bar, 2 µm. (C) Quantification of B. Hundreds of cells in independent cultures expressing tagged Spr28 alone (n = 3), tagged Spr3 alone (n = 3), or both (n = 5) were examined by fluorescence microscopy. Bars, mean percentage of the budded cells that displayed a visible fluorescent signal at the bud neck; error bar, standard deviation of the mean. (D) To analyze expression of the indicated fluorescently tagged septins in the cells from B (left) and Fig. S1 (right), equivalent numbers of cells were lysed, resolved by SDS-PAGE, transferred to a nitrocellulose filter, and probed with appropriate antibodies (anti-GFP, anti-DsRed, or anti-Pgk1). Pgk1, control for equivalent protein loading.
In vegetative cells, either Cdc11-GFP (Fig. 3 B, left, top) or Cdc12-GFP (Fig. 3 B, left, bottom) were quantitatively incorporated into the filamentous collar at the bud neck, whereas Spr28-GFP (Fig. 3 B, middle, top) and Spr3-GFP (Fig. 3 B, middle, bottom) each displayed only cytosolic fluorescence in all cells examined. Revealingly, however, in cells coexpressing Spr28-GFP and Spr3-mCherry (Fig. 3 B, right), a significant proportion of the dividing cells (40–45%; Fig. 3 C) exhibited a clearly detectable fluorescent signal for both proteins at the bud neck, in addition to the diffuse cytosolic fluorescence (Fig. 3 B, right), even though the cells also expressed all five mitotic septins. Similar results were obtained when the fluorescent tags were swapped (Spr28-mCherry and Spr3-GFP; Fig. S2). Thus, in the cytoplasm of a mitotic cell, the sporulation-specific septins are competent for association with other septin subunits. Hence, the inability of Spr28 and Spr3 to complement cdc11Δ and cdc12Δ mutations, respectively, cannot be attributed to misfolding of these proteins. Second, incorporation of Spr28 and Spr3 at the bud neck in vivo required their simultaneous presence. Thus, the two proteins function as a unit and mutually promote their assembly into hetero-octamers, consistent with the conclusions of our in vitro biochemical and EM findings.
As shown here, Spr3 shares with Cdc12 the capacity to interact with Cdc3, and sporulation-specific hetero-octamers share with mitotic septin hetero-octamers the capacity to bind PtdIns4,5P2. Hence, expression of Spr3 and Spr28 in mitotic cells could interfere with normal growth. When Spr28-GFP was produced from the CDC11 promoter on a CEN vector, or Spr3-mCherry was made from the CDC12 promoter on another CEN vector (or both), there was no obvious effect on the growth rate of otherwise wild-type cells (Fig. S3). However, when we used any of three different means to ectopically over-produce the sporulation-specific septins in vegetative cells that also carried sensitizing mutations (alleles that compromise the function of particular septin subunits), mitotic growth rate was markedly reduced (Fig. S4) and cell morphology was altered in an increased proportion of the cell population (Fig. S5).
Mitotic septins alone cannot support sporulation
In the BY4743 genetic background (Brachmann et al., 1998), the homozygous spr3Δ/spr3Δ and spr28Δ/spr28Δ MATa/MATα diploids we constructed exhibited a marked reduction in overall sporulation proficiency, especially in production of four-spored asci, compared with the otherwise isogenic parental diploid (Fig. 4 A, left). Similarly, in a high-throughput screen (Enyenihi and Saunders, 2003), severe sporulation defects were seen in the absence of CDC10 in BY4743 cells. Moreover, diploid cells lacking both SPR3 and SPR28 failed to generate any detectable spores (Fig. 4 A, left). Importantly, in diploids in which the mitotic septins Cdc11 and Cdc12 were expressed from the native SPR28 and SPR3 promoters in place of the corresponding sporulation-specific septins, sporulation proficiency was scarcely increased (Fig. 4 A, left). Similarly, in the background of a hybrid between BY4743 and the SK1 strain typically used for synchronous and highly efficient meiosis (Börner and Cha, 2015) that was constructed and characterized (Heasley and McMurray, 2016), sporulation efficiency is very sensitive to the dose of Spr3 and Spr28 present and not fully restored by meiosis-specific expression of either Cdc12 and Cdc11 or Cdc12 and Shs1 (Fig. 4 A, right). Thus, just as Spr3 and Spr28 cannot functionally replace Cdc12 and Cdc11 to support mitosis, these mitotic septin subunits cannot functionally replace their meiosis-specific counterparts in the sporulation process.
Spr3- and Spr28-containing complexes are required for septin function and localization during sporulation. (A) Overall sporulation efficiencies and spore number per ascus were determined for diploids of the indicated genotypes. Left side, BY4743; MMY0221; MMY0154; MMY0231; GCFY7. The left-most three columns recapitulate data in Heasley and McMurray (2016) are included as a basis for comparison to the effect on sporulation proficiency of loss of both SPR3 and SPR28 or of replacing them with CDC12 and CDC11, respectively. Right side, MMY0228, MMY0184 x MMY0155; MMY0230; MMY0226. Asterisk, no asci detectable; error bars, standard error of the proportion. (B) FRAP analysis. Top micrograph, representative Cdc10-GFP-expressing cell; bottom micrograph, representative GFP-2X(PHOsh2)-expressing cell. Dashed circles, sites of signal bleaching and/or detection; filled white arrow, bleached site; outlined dark arrow, unbleached (control) site. Bar, 2 µm. (bottom) Cdc10-GFP-marked septin structures (black lines) or GFP-2X(PHOsh2)–marked portions of the PSM (green lines) were photobleached and recovery was visualized by time-course imaging, quantified, averaged over all trials, and plotted as the mean ± SEM. Top lines, unbleached (control) area; bottom lines, bleached area. Mean percent recovery: Cdc10-GFP, 9.4 ± 2.9% (n = 15); GFP-2X(PHOsh2), 28.4 ± 2.9% (n = 11). (C) Representative images of YFP fluorescence in sporulating cells expressing the indicated BiFC pairs. Bar, 5 µm.
Spr3- and Spr28-containing complexes are required for septin function and localization during sporulation. (A) Overall sporulation efficiencies and spore number per ascus were determined for diploids of the indicated genotypes. Left side, BY4743; MMY0221; MMY0154; MMY0231; GCFY7. The left-most three columns recapitulate data in Heasley and McMurray (2016) are included as a basis for comparison to the effect on sporulation proficiency of loss of both SPR3 and SPR28 or of replacing them with CDC12 and CDC11, respectively. Right side, MMY0228, MMY0184 x MMY0155; MMY0230; MMY0226. Asterisk, no asci detectable; error bars, standard error of the proportion. (B) FRAP analysis. Top micrograph, representative Cdc10-GFP-expressing cell; bottom micrograph, representative GFP-2X(PHOsh2)-expressing cell. Dashed circles, sites of signal bleaching and/or detection; filled white arrow, bleached site; outlined dark arrow, unbleached (control) site. Bar, 2 µm. (bottom) Cdc10-GFP-marked septin structures (black lines) or GFP-2X(PHOsh2)–marked portions of the PSM (green lines) were photobleached and recovery was visualized by time-course imaging, quantified, averaged over all trials, and plotted as the mean ± SEM. Top lines, unbleached (control) area; bottom lines, bleached area. Mean percent recovery: Cdc10-GFP, 9.4 ± 2.9% (n = 15); GFP-2X(PHOsh2), 28.4 ± 2.9% (n = 11). (C) Representative images of YFP fluorescence in sporulating cells expressing the indicated BiFC pairs. Bar, 5 µm.
Behavior of sporulation-specific septin complexes in vivo
One characteristic of septins stably assembled into filaments and higher-order structures in mitotic cells is a markedly diminished mobility, as judged by a lack of FRAP in cells expressing fluorescently-tagged septins (Caviston et al., 2003; Dobbelaere et al., 2003). Given that PtdIns4,5P2 is highly enriched in the PSM, and our observation that this lipid promotes polymerization of the Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 complex into filaments in vitro (Fig. 2 B), we used FRAP to determine whether the septin-containing “horseshoes” observed by light microscopy in sporulating cells display the FRAP behavior expected for a highly organized array of filaments. We found that the structures containing Cdc10-GFP never exhibited significant recovery of their fluorescent signal (<10%; n = 15) after photobleaching (Fig. 4 B, top), whereas areas bleached of GFP-2X(PHOsh2), a probe that binds to PtIns4P (a hydrolysis product of PtdIns4,5P2) on membranes (Roy and Levine, 2004), displayed substantially greater recovery of the fluorescent signal (22–55%, depending on the experiment; n = 11; Fig. 4 B, bottom). Thus, as assessed by FRAP, the septin-containing structures on the PSM display in vivo the hallmark of highly organized structures, consistent with their PtdIns4,5P2-promoted assembly into well ordered filaments in vitro.
We demonstrated before that the premade Cdc12 and Shs1 persist during sporulation, but are localized mainly in the ascal cytoplasm and not associated with the other septins (McMurray and Thorner, 2008), suggesting that their replacement by Spr3 and Spr28 may involve an active process for their eviction. We reasoned that if we could overcome this eviction mechanism by forcing stable interaction between Cdc12 and Cdc3, we could assess the consequences of maintaining some mitotic-like septin complexes in sporulating cells. To achieve this end, we used bimolecular fluorescence complementation (BiFC) because, once two associating proteins bring the two halves of the fluorescent reporter protein together, they are irreversibly “locked” together (Kerppola, 2008). As a control, we first coexpressed Cdc3-VC with Cdc10-VN, which we anticipated should fluorescently mark the septin horseshoes associated with the developing PSMs, as we indeed observed (Fig. 4 C, top). Likewise, we expected that when Cdc3-VC was coexpressed with Spr3-VN, only septin structures associated with developing PSMs would yield a prominent signal, as we also observed (Fig. 4 C, middle). In contrast, in sporulating cells containing Cdc3-VC and Cdc12-VN, most of the fluorescence was located in small puncta dispersed around the periphery of the ascus and not associated with PSMs at all (Fig. 4 C, bottom). Thus, presence of Cdc12 in septin complexes is not compatible with their ability to form the PSM-associated septin structures required for sporulation. These findings explain why, during meiosis, replacement of Cdc12 by Spr3 is critical for the formation the septin architecture necessary for proper execution of this developmental process.
Discussion
In phylogenetic comparisons of evolutionary relationships among septins (Pan et al., 2007; Momany et al., 2008; Nishihama et al., 2011), S. cerevisiae Spr28 is most closely related to Cdc11-like subunits and S. cerevisiae Spr3 is most closely related to Cdc12-like subunits. Such trees also indicate that Spr28 diverged before the split that separates the Cdc11-like group from its closest relative, the Shs1-like group. Moreover, Spr-like homologues, even in yeasts closely related to S. cerevisiae, are quite divergent (i.e., there is substantial sequence variation among Spr family members but strong conservation of Cdc11 and Cdc12 identity among the same species), suggesting that Spr subunit structure is not highly constrained. Nonetheless, we found that S. cerevisiae Spr3 and Spr28 each replace their closest mitotic septin relatives, thereby forming a linear sporulation-specific Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 complex with unique assembly and membrane-localizing properties. Thus, our findings reveal how displacement of resident septins by alternative subunits can confer distinctive supramolecular organization and function on septin complexes.
During sporulation in fission yeast (Schizosaccharomyces pombe), three new septins (Spn5, Spn6, and Spn7) assemble with Spn2 (Longtine et al., 1996) and supplant the vegetatively expressed septins (Spn1, Spn3, and Spn4) that associate with Spn2 (An et al., 2004). The three sporulation-specific subunits form a complex with Spn2 in vitro (although their order/organization has not been determined) and colocalize interdependently in vivo to the forespore membrane (equivalent to the PSM in S. cerevisiae), and loss of a sporulation-specific subunit results in less organized forespore membrane growth and decreases the number of viable spores formed (Onishi et al., 2010). Spn7 is most similar to Spr28 and Spn6 is most closely related to Spr3. Absence of Spn7 prevents incorporation of Spn6 into complexes with the other two septins (Onishi et al., 2010), similar to our observation that presence of both Spr28 and Spr3 promotes their mutual incorporation into hetero-octamers. The ability of coexpressed Spr28 and Spr3 to contribute to formation of hetero-octamers that are stable to EM processing in vitro and to incorporate into the bud neck in mitotic cells (when neither alone is competent to do so) suggests that they form a heterodimer in which Spr28 confers on Spr3 sufficient stability and affinity for Cdc3 to successfully compete with Cdc11-Cdc12. Perhaps all septin pairs, like Spr28-Spr3, first interact via their G interfaces forming a heterodimer before they assemble via their NC interfaces into hetero-octamers (Sirajuddin et al., 2007; Bertin et al., 2008; Weirich et al., 2008), akin to conclusions about the formation of mammalian septin complexes reached by Kim et al. (2012).
A striking feature of the sporulation-specific hetero-octamers was lack of self-association in low-salt solution. However, on lipid monolayers containing PtdIns4,5P2 as a plasma membrane mimic, sporulation-specific hetero-octamers were able to polymerize end-on-end into long paired filaments with a pronounced “railroad track” appearance and aligned extensively in rather well-organized parallel arrays. A basic motif in the so-called α0 helix just upstream of the conserved P-loop of the GTP-binding domain has been implicated in the ability of a septin subunit to interact with PtdIns4,5P2 (Zhang et al., 1999). In this regard, Spr3 is slightly more basic in this region (RELLNAKN) than Cdc12 (RYKIVNEE; Fig. S1 A), and Spr28 has just as many basic residues (six) in this region as does Cdc11 or Shs1 (Fig. S1 B). The corresponding basic patches in Cdc10, Cdc11 and Shs1 are necessary for membrane recruitment and function of these proteins (Finnigan et al., 2015).
In S. pombe, the forespore membrane is enriched in PtdIns4P, and two sporulation-specific septin subunits (Spn2 and Spn7) bind this phosphoinositide (Onishi et al., 2010). Cells expressing a Spn2 mutant unable to bind PtdIns4P still form septin complexes, but they fail to associate with the forespore membrane, which becomes disoriented, suggesting that septin binding to the forespore membrane helps guide its oriented growth (Onishi et al., 2010). Similar to what we show here for S. cerevisiae sporulation-specific septin complexes, recombinant S. pombe sporulation-specific septin complexes do not form filaments in solution (M. Onishi, personal communication). However, for S. cerevisiae sporulation-specific septin complexes, it is PtdIns4,5P2, not PtdIns4, that promotes formation of ordered filament ensembles. Our results further highlight the importance of phosphoinositides in regulating septin assembly and organization.
Although others reported that diploids lacking Spr3 (Fares et al., 1996) or Spr28 (De Virgilio et al., 1996) display only a mild, if any, decrease in the efficiency of sporulation compared with corresponding control cells, we found an obvious and severe reduction in sporulation proficiency in the absence of either of the sporulation-specific septins in BY4743 diploids. We also observed the same in a BY-SK1 hybrid strain (Heasley and McMurray, 2016) that displays an overall higher sporulation efficiency. Aside from the fact that we took great care to always use a uniform sporulation protocol, we have no obvious explanation for why, in our hands, diploids lacking Spr3 and/or Spr28 have a more profound phenotypic effect on sporulation than was previously described by others.
It is clear from our genetic complementation tests that Spr3 and Spr28 cannot substitute for the essential functions of Cdc12 and Cdc11 in mitotic cells; conversely, Cdc12 and Cdc11 cannot perform the function(s) executed by Spr3 and Spr28 in meiotic cells. In the latter regard, when Spr3 or Spr28 are absent, there is at least a 10-fold reduction in the frequency of sporulation. Also, the fitness of the few spores that are produced is drastically compromised with respect to the quality of their maturation and integrity, as documented elsewhere (Heasley and McMurray, 2016). Thus, Spr28–Spr3–Cdc3–Cdc10–Cdc10–Cdc3–Spr3–Spr28 complexes have significantly different properties and functions from either Cdc11–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Cdc11 or Shs1–Cdc12–Cdc3–Cdc10–Cdc10–Cdc3–Cdc12–Shs1 complexes. Moreover, our FRAP and BiFC analysis indicates that, in vivo, the PSM-associated horseshoes containing the sporulation-specific septins exclude the displaced mitotic septins and are highly organized structures, in agreement with the filaments assembled in vitro on PtdIns4,5P2-containing monolayers. Thus, sporulation-specific septin complexes, but not mitotic septin complexes, are capable of forming the proper higher-order structures and occupying the correct location to direct efficient spore morphogenesis.
In septin complex formation, guanine nucleotide binding has important roles both in intersubunit contacts at their G interface and for inducing assembly-promoting conformational changes (Sirajuddin et al., 2007, 2009). Moreover, compelling genetic evidence indicates a critical role for GTP binding in yeast septin subunit folding and hetero-octamer assembly in mitotic cells (Weems et al., 2014; Johnson et al., 2015). Crystal structures show that an Asp located in the G-2 motif of the GTP-binding domain in septins and other related small G proteins is important for GTP binding (Wittinghofer and Vetter, 2011). It seems that Spr28 lacks the corresponding Asp residue (Fig. S1 B). In this regard, it is of potential interest that a decrease in intracellular guanine nucleotide production promotes sporulation (Varma et al., 1985). Thus, it is tempting to speculate that, if Spr28 has evolved to no longer require GTP for its folding and assembly, this property and the drop in guanine nucleotide that occurs during meiosis may explain, in part, how formation of sporulation-specific septin complexes outcompetes assembly of residual mitotic septins into complexes.
Yeast sporulation is a form of gametogenesis. As in sporulating yeast, during mammalian spermiogenesis, gamete-specific subunits are incorporated into septin complexes and form higher-order structures distinct from those in mitotically-dividing cells (Lin et al., 2011). SEPT4 is expressed mainly in male germ cells (and postmitotic neural cells) and occupies the same central position in mammalian septin hetero-octamers as Cdc10 does in yeast hetero-octamers (Sandrock et al., 2011). In spermatozoa, SEPT4 is located in the annulus, a cortical ring that separates the middle and principal pieces of a mature sperm from its tail (Kwitny et al., 2010). Nullizygous sept4−/− male mice lack a normal annulus and are sterile because of defective sperm morphology and loss of motility of the flagellum (Ihara et al., 2005). SEPT12 is a testis-specific septin that occupies the same terminal position in human septin hetero-octamers as Spr28 does in the sporulation-specific yeast hetero-octamer. Mutations in SEPT12 cause infertility in men (Kuo et al., 2012), demonstrating the functional importance of gametogenesis-specific septins. However, nothing is yet known about the mechanisms involved in “remodeling” the septin repertoire during this (or any other) human cellular differentiation process.
Thus, the molecular organization and properties of the yeast sporulation-specific septin complexes, and the nature of their meiosis-specific interaction partners, will continue to be important models for understanding the unique roles of differentiation-specific septins. Hence, further study of the contributions of individual septin subunits and their development-specific posttranslation modifications in meiosis and other developmental processes in S. cerevisiae (e.g., pheromone response and filamentous growth) may shed additional light on general mechanisms that regulate the composition and function of septin complexes in diverse cell types, especially in organisms that, like humans, possess a large number of septin subunits.
Materials and methods
Expression and purification of septin complexes
In general, both the strategy for inducible heterologous expression in Escherichia coli and the purification procedure (metal ion affinity, size-exclusion chromatography, and anion exchange] to prepare yeast septin complexes, are described in detail elsewhere (Versele et al., 2004; Bertin et al., 2008; Garcia et al., 2011; Booth et al., 2015). Ligation-independent cloning (Aslanidis and de Jong, 1990) was used to incorporate CDC3, CDC10, SPR3, and/or SPR28 into bicistronic DUET (Invitrogen) vectors with compatible replication origins, and the resulting plasmids were introduced by DNA-mediated transformation into E. coli strain BL1(DE3). The desired transformants were selected on agar plates of Luria–Bertani broth; Bertani, 1951; Luria and Burrous, 1957; Miller, 1972) containing appropriate antibiotics for marker selection (40 µg/ml ampicillin, 34 µg/ml chloramphenicol, and 40 µg/ml kanamycin). Liquid cultures of Luria–Bertani broth containing antibiotics (20 µg/ml ampicillin, 17 µg/ml chloramphenicol, and 20 µg/ml kanamycin) to maintain selection were typically seeded with ∼10 colonies picked directly from the plates and grown overnight at 37°C. We have found that inoculation with multiple colonies yields greater reproducibility in final protein yield and quality from preparation to preparation. Samples (5 ml) of such overnight cultures were, in turn, used to inoculate larger (1 liter) cultures of Tartof–Hobbs medium (“Terrific broth”; Tartof and Hobbs, 1987) with the same antibiotics to maintain selection, which were grown at 37°C to a density of A595 nm = 0.7, whereupon expression was induced by addition of isopropyl-β-d-thiogalactoside (IPTG; 0.1 mM final concentration) and the culture shifted to 16°C. After 16 h, the cells were harvested by centrifugation at 3,000 g and the resulting pellet was resuspended in 10 ml of lysis buffer (40 µM GDP, 12% glycerol, 0.5% Tween-20, 300 mM KCl, 2 mM MgCl2, 20 mM imidazole, and 50 mM Tris-HCl, pH 8.0), flash-frozen by drop-wise addition of the cell resuspension in liquid N2, and stored at −80°C before use.
Frozen cell pellets were thawed in an ice-water bath, resuspended in lysis buffer-PLUS (5 ml; lysis buffer-PLUS was prepared just before use by adding 10 µl 1-thioglycerol, 40 µl Hercules endonuclease [Genscript], 800 µg lysozyme, and four Halt EDTA-free protease inhibitor tablets [Pierce/Thermo Scientific] to 25 ml of lysis buffer), incubated at 4°C with gentle agitation for 30 min, and then ruptured by four 30-s bursts of sonic irradiation (separated by 2-min periods on ice between each pulse) at a power output of 6 W. The crude lysate was clarified by centrifugation at 25,000 g, and the resulting crude extract was applied using a peristaltic pump to a bed (5 ml) of prepacked Ni2+-charged affinity resin (HisTrap HP; GE Healthcare) at a flow rate of 2 ml/min. After washing with 75 ml wash buffer (25 mM imidazole, 0.1% 1-thioglycerol, 300 mM KCl, and 50 mM Tris-HCL, pH 8.0) at a flow rate of 4 ml/min, bound protein was eluted with 30 ml elution buffer (500 mM imidazole, 0.1% 1-thioglycerol, 300 mM KCl, and 50 mM Tris-HCl, pH 8.0) at a flow rate of 1 ml/min and collected as 1-ml fractions. Protein content of the resulting fractions was assessed using the dye-binding method of Bradford (1976). Fractions with the highest content of septin protein were pooled (6 ml total) and passed through a PVDF membrane (0.2 µm) to remove any particulate material, and the resulting filtrate (5 ml) was loaded onto the top of a bed (in a 120-ml column) of prep-grade Hi-load 16/60 Superdex 200 (GE Healthcare) and eluted with 300 mM KCl, 0.1% 1-thioglycerol, and 50 mM Tris-HCl, pH 8.0, at a flow rate of 0.6 ml/min. In most cases, these two steps were sufficient to yield a purity of ≥90%. If not, the pooled peak fractions from size-exclusion chromatography (determined by A280 nm) were applied to a 1-ml Resource Q column (GE Healthcare) and eluted with a linear salt gradient from 10 mM KCl, 0.1% 1-thioglycerol, 50 mM Tris-HCl, pH 8.0, to 1 M KCl, 0.1% 1-thioglycerol, and 50 mM Tris-HCl, pH 8.0. Aliquots of the peak fractions (as determined by A280 nm) were flash-frozen in liquid N2 and stored at −80°C until used for experiments.
Analytical size-exclusion chromatography
A septin complex (150 pmol) of interest was loaded onto a Superose 6 PC column, eluted with buffer (75 mM KCl, 2 mM MgCl2, 0.1% 1-thioglycerol, and 50 mM Tris-HCl, pH 8.0) at a flow rate of 0.04 ml/min, and the resulting profile analyzed using an Ettan LC apparatus (GE Healthcare).
Electron microscopy and image processing
Purified septin complexes were diluted to 0.01 mg/ml in either high-salt buffer (300 mM NaCl, 2 mM MgCl2, and 50 mM Tris-HCl, pH 8.0) or low-salt buffer (10 mM NaCl, 2 mM MgCl2, and 50 mM Tris-HCl, pH 8.0) and applied to the surface of a carbon-coated copper EM grid prepared by glow-discharge using an Auto 306 Thermal Evaporator (Edwards). The grids were then washed with water and stained with 2% uranyl formate. Electron micrographs of the adsorbed protein were taken using a Tecnai T12 electron microscope (FEI) operated at 120 kV. Unless otherwise indicated, micrographs were taken at 30,000 magnification and at −1 µm defocus. Data were collected using Leginon (Potter et al., 1999) with a 4k × 4k complementary metal oxide semi-conductor (CMOS) camera (TVIPS TemCam F416). Images of individual complexes (particles) were windowed out of the images with a box size of 135 by 135 pixels using the Boxer program within the EMAN software package (Ludtke et al., 1999). Particles were then aligned and classified using SPIDER (Frank et al., 1996) within the Appion pipeline (Lander et al., 2009). The first round of alignment and classification was reference-free, and class averages representative of the full diversity in length and curvature of the particles in the sample were obtained. These class averages were used as references in subsequent iterations of alignment and classification. After each round, new references were chosen from the class averages produced, and Iterations of alignment and classification were continued until the class averages did not change from one round to the next. Typically, three iterations were performed.
Septin assembly on lipid monolayers
Association of purified recombinant septin complexes with lipid monolayers were performed by slight modifications of prior methods (Kubalek et al., 1991; Taylor et al., 2007; Bertin et al., 2010). In brief, protein samples (20 µl; 100 nM) in low-salt buffer (50 mM KCl and 20 mM Tris-HCl, pH 8.0) were dispensed into polytetrafluoroethylene (Teflon) wells (20 µl), yielding a convex meniscus at the surface. A stock solution (10 mg/ml) of DOPC in chloroform (Avanti Polar Lipids) was diluted to 0.25 mg/ml on the day of use. Stock solutions (1 mg/ml) of either PtdIns4,5P2 in 9:1::chloroform:methanol (vol/vol; Avanti Polar Lipids) or PtdIns4P in 9:1::chloroform:methanol (vol/vol; Avanti Polar Lipids) were mixed with the DOPC stock solution such that the final phosphoinositide concentration was 15.2 mol% and then diluted to 0.25 mg/ml on the day of use. The desired lipid solution (0.5 µl) was gently spread onto the surface of the protein-containing solution in the Teflon well, which caused a slight flattening of the meniscus. After incubation for 15 h at 4°C in a humidified chamber, the carbon-coated side of a C-flat grid (Proto-chips) with 1.2-µm holes was carefully placed on the surface of the well for 45 s, allowing the lipid monolayer (and any protein associated with its opposite face) to adsorb to the carbon surface. The grids then were lifted vertically from the wells and immediately stained with 2% uranyl formate, air-dried, and viewed by EM as described in the previous section. Micrographs were cropped for publication using ImageJ (National Institutes of Health).
Yeast strains and strain constructions
All yeast strains (Table 1) are derived from BY4741 or BY4742 (Brachmann et al., 1998) and were cultivated and manipulated using standard methods (Amberg et al., 2005). For some experiments, rich (“YP”) medium contained tryptone instead of peptone, which had no noticeable effect on growth. BY4742 sum1Δ0::kanMX was purchased from Thermo Fisher Scientific. Strain YMVB1 cdc12(T48N) (Versele and Thorner, 2004) was crossed with BY4742 sum1Δ0::kanMX and, after sporulation, tetrads were dissected to obtain cdc12(T48N) sum1Δ haploid. To create a cdc12-6 derivative of BY4741, the cdc12-6 allele was first introduced into a CDC12-GFP gene on a HIS3-marked CEN plasmid (pLP29; Lippincott and Li, 1998) by digestion with BfuAI and cotransformation of the cut plasmid into YMVB61 (cdc12Δ::kanMX [CDC12 URA3]; McMurray et al., 2011) along with cdc12-6 DNA PCR-amplified from strain DDY1462 (gift of D. Drubin, University of California, Berkeley, Berkeley, CA). Transformants were plated on 5-FOA medium (Boeke et al., 1987), and plasmids were recovered from those colonies that displayed temperature-sensitive growth by rescue in E. coli and sequenced to confirm the presence of the cdc12-6 allele (K391N L392stop). The CDC12 ORF (including an out-of-frame GFP coding sequence and downstream HIS3 marker) from one such plasmid (designated YCpH-Cdc12-6) was PCR-amplified and used to transform BY4741, creating the cdc12-6 mutant used here (MMY0000). JTY3993 (BY4742 CDC10-mCherry::kanMX; McMurray et al., 2011) was transformed with BamHI–cut pSC193 (Chu et al., 1998), integrating at the NDT80 locus a URA3-marked copy of NDT80-HA behind the GAL1/10 promoter, creating strain JTY5200. Derivatives of BY4741 carrying cdc10-1::kanMX allele (encoding a D182N mutant of Cdc10) and the cdc12-1::kanMX allele (encoding a G247E mutant of Cdc12) were obtained from a collection of temperature-sensitive mutants (Li et al., 2011), verified by sequencing the relevant septin gene, and mated with JTY5200 to obtain by subsequent sporulation and tetrad dissection the cdc10-1 PGAL-NDT80 strain (MMY0046) and the cdc12-1 PGAL-NDT80 strain (MMY0047). A PGAL-NDT80 derivative of BY4741 with untagged Cdc10 (MMY0048) was created using pSC193, in a similar manner.
Yeast strains used in this study
| Strain | Genotype | Reference |
|---|---|---|
| SF838-1Dα | MATα ura3-52 leu2-3,112 HIS4 ade6 pep4-3 gal2 | Rothman and Stevens, 1986 |
| DDY1462 | MATα ura3-52 cdc12-6 | Originally from P. Novick, University of California, San Diego, La Jolla, CA |
| YMVB1 | BY4741; cdc12(T48N)::URA3 | Versele and Thorner, 2004 |
| YMVB61 | BY4741; cdc12Δ::KanR + pMVB39 | McMurray et al., 2011 |
| BY4741 | MATa his3Δ leu2Δ ura3Δ met15Δ | Brachmann et al., 1998 |
| BY4742 | MATα his3Δ leu2Δ ura3Δ lys2Δ | Brachmann et al., 1998 |
| BY4743 | MATa/MATα his3Δ/his3Δ leu2Δ/leu2Δ ura3Δ/ura3Δ LYS2/lys2Δ MET15/met15Δ | Brachmann et al., 1998 |
| JTY5167 | BY4742 sum1Δ::KanR | Genome Deletion Collection |
| JTY3993 | BY4742 CDC10-mCherry::KanR | McMurray et al., 2011 |
| CBY06417a | BY4741 cdc10-1::KanR | Li et al., 2011 |
| CBY05110a | BY4741 cdc12-1::KanR | Li et al., 2011 |
| GCFY1 | BY4742 cdc11Δ::KanR + pSB1 | This study |
| GCFY2 | BY4742 cdc11Δ::SPR28-GFP-ADH1(t)-HygR [pJT1520] | This study |
| GCFY3 | BY4741 cdc12Δ::SPR3-mCherry-ADH1(t)-KanR [pJT1622] | This study |
| GCFY4 | BY4741 cdc12Δ::SPR3-GFP-ADH1(t)-HygR [pJT1622] | This study |
| GCFY5b | MATα cdc11Δ::SPR28-GFP-ADH1(t)-HygR cdc12Δ::SPR3-mCherry-ADH1(t)-KanR [pGCF1/pGCF2] | This study |
| GCFY6b | MATa cdc11Δ::SPR28-mCherry-ADH1(t)-KanR cdc12Δ::SPR3-GFP-ADH1(t)-HygR [pGCF1/pGCF2] | This study |
| JTY5168c | sum1Δ::KanR cdc12(T48N)::URA3 | This study |
| MMY0000d | BY4741 cdc12-6::HIS3 | This study |
| JTY5200e | JTY3993 prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0046f | cdc10-1::KanR prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0047g | cdc12-1::KanR prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0048h | BY4741 prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0219 | BY4741 spr3Δ::KanR | Genome Deletion Collection |
| MMY0220 | BY4742 spr3Δ::KanR | Genome Deletion Collection |
| MMY0221 | BY4743 spr3Δ::KanR/spr3Δ::KanR | This study |
| MMY0152 | BY4741 spr28Δ::KanR | Genome Deletion Collection |
| MMY0153 | BY4742 spr28Δ::KanR | Genome Deletion Collection |
| MMY0154 | BY4743 spr28Δ::KanR/spr28Δ::KanR | This study |
| MMY0231 | BY4743 spr3Δ::KanR/spr3Δ::KanR spr28Δ::KanR/spr28Δ::KanR | This study |
| GCFY7 | BY4743 spr28Δ::CDC11::eGFP::NatR / spr28Δ::CDC11::eGFP::HygR spr3Δ::CDC12::mCherry::KanR / spr3Δ::CDC12::mCherry::KanR | This study |
| FY2742 | MATα his3Δ1 leu2Δ0 lys2Δ0 ura3Δ0 MKT1(G30) RME1 TAO3(Q1493) | F. Winston, Harvard Medical School, Boston, MA |
| FY2839 | MATa his3Δ1 leu2Δ0 lys2Δ0 ura3Δ0 MKT1(G30) RME1 TAO3(Q1493) | F. Winston |
| MMY0185 | FY2839 spr3Δ::KanR | This study |
| MMY0222 | FY2742 spr3Δ::KanR | This study |
| MMY0186 | FY2839 spr28Δ::KanR | This study |
| MMY0155 | FY2742 spr28Δ::KanR | This study |
| MMY0149 | FY2742 CDC12-eCitrine::his5MX | This study |
| MMY0228 | rme1/RME1 mkt1/MKT1 tao3/TAO3 CDC12/CDC12-eCitrine::his5MX | This study |
| MMY0229 | FY2742 spr28Δ::KanR CDC12-eCitrine::his5MX | This study |
| MMY0230 | rme1/RME1 mkt1/MKT1 tao3/TAO3 spr3Δ::CDC12/SPR3+ spr28Δ::CDC11/SPR28+ CDC12/CDC12-eCitrine::his5MX | This study |
| MMY0226 | rme1/RME1 mkt1/MKT1 tao1/TAO1 SPR28/spr28Δ::SHS1::Vc::HIS3MX6 SPR3/spr3Δ::CDC12::mCherry::KanR | This study |
| YO685 | MATα his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC3-Vc::HIS3MX6 | Oh et al., 2013 |
| YEF5690 | MATa his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC10-Vn::kanMX6 | Oh et al., 2013 |
| YEF5692 | MATa his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC12-Vn::TRP1 [pRS316 CDC12] | Oh et al., 2013 |
| MMY0224 | BY4742 SPR3-Vn::URA3 | This study |
| Strain | Genotype | Reference |
|---|---|---|
| SF838-1Dα | MATα ura3-52 leu2-3,112 HIS4 ade6 pep4-3 gal2 | |
| DDY1462 | MATα ura3-52 cdc12-6 | Originally from P. Novick, University of California, San Diego, La Jolla, CA |
| YMVB1 | BY4741; cdc12(T48N)::URA3 | |
| YMVB61 | BY4741; cdc12Δ::KanR + pMVB39 | |
| BY4741 | MATa his3Δ leu2Δ ura3Δ met15Δ | |
| BY4742 | MATα his3Δ leu2Δ ura3Δ lys2Δ | |
| BY4743 | MATa/MATα his3Δ/his3Δ leu2Δ/leu2Δ ura3Δ/ura3Δ LYS2/lys2Δ MET15/met15Δ | |
| JTY5167 | BY4742 sum1Δ::KanR | Genome Deletion Collection |
| JTY3993 | BY4742 CDC10-mCherry::KanR | |
| CBY06417 | BY4741 cdc10-1::KanR | |
| CBY05110 | BY4741 cdc12-1::KanR | |
| GCFY1 | BY4742 cdc11Δ::KanR + pSB1 | This study |
| GCFY2 | BY4742 cdc11Δ::SPR28-GFP-ADH1(t)-HygR [pJT1520] | This study |
| GCFY3 | BY4741 cdc12Δ::SPR3-mCherry-ADH1(t)-KanR [pJT1622] | This study |
| GCFY4 | BY4741 cdc12Δ::SPR3-GFP-ADH1(t)-HygR [pJT1622] | This study |
| GCFY5 | MATα cdc11Δ::SPR28-GFP-ADH1(t)-HygR cdc12Δ::SPR3-mCherry-ADH1(t)-KanR [pGCF1/pGCF2] | This study |
| GCFY6 | MATa cdc11Δ::SPR28-mCherry-ADH1(t)-KanR cdc12Δ::SPR3-GFP-ADH1(t)-HygR [pGCF1/pGCF2] | This study |
| JTY5168 | sum1Δ::KanR cdc12(T48N)::URA3 | This study |
| MMY0000 | BY4741 cdc12-6::HIS3 | This study |
| JTY5200 | JTY3993 prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0046 | cdc10-1::KanR prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0047 | cdc12-1::KanR prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0048 | BY4741 prGAL1/10-NDT80-HA::URA3 | This study |
| MMY0219 | BY4741 spr3Δ::KanR | Genome Deletion Collection |
| MMY0220 | BY4742 spr3Δ::KanR | Genome Deletion Collection |
| MMY0221 | BY4743 spr3Δ::KanR/spr3Δ::KanR | This study |
| MMY0152 | BY4741 spr28Δ::KanR | Genome Deletion Collection |
| MMY0153 | BY4742 spr28Δ::KanR | Genome Deletion Collection |
| MMY0154 | BY4743 spr28Δ::KanR/spr28Δ::KanR | This study |
| MMY0231 | BY4743 spr3Δ::KanR/spr3Δ::KanR spr28Δ::KanR/spr28Δ::KanR | This study |
| GCFY7 | BY4743 spr28Δ::CDC11::eGFP::NatR / spr28Δ::CDC11::eGFP::HygR spr3Δ::CDC12::mCherry::KanR / spr3Δ::CDC12::mCherry::KanR | This study |
| FY2742 | MATα his3Δ1 leu2Δ0 lys2Δ0 ura3Δ0 MKT1(G30) RME1 TAO3(Q1493) | F. Winston, Harvard Medical School, Boston, MA |
| FY2839 | MATa his3Δ1 leu2Δ0 lys2Δ0 ura3Δ0 MKT1(G30) RME1 TAO3(Q1493) | F. Winston |
| MMY0185 | FY2839 spr3Δ::KanR | This study |
| MMY0222 | FY2742 spr3Δ::KanR | This study |
| MMY0186 | FY2839 spr28Δ::KanR | This study |
| MMY0155 | FY2742 spr28Δ::KanR | This study |
| MMY0149 | FY2742 CDC12-eCitrine::his5MX | This study |
| MMY0228 | rme1/RME1 mkt1/MKT1 tao3/TAO3 CDC12/CDC12-eCitrine::his5MX | This study |
| MMY0229 | FY2742 spr28Δ::KanR CDC12-eCitrine::his5MX | This study |
| MMY0230 | rme1/RME1 mkt1/MKT1 tao3/TAO3 spr3Δ::CDC12/SPR3+ spr28Δ::CDC11/SPR28+ CDC12/CDC12-eCitrine::his5MX | This study |
| MMY0226 | rme1/RME1 mkt1/MKT1 tao1/TAO1 SPR28/spr28Δ::SHS1::Vc::HIS3MX6 SPR3/spr3Δ::CDC12::mCherry::KanR | This study |
| YO685 | MATα his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC3-Vc::HIS3MX6 | |
| YEF5690 | MATa his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC10-Vn::kanMX6 | |
| YEF5692 | MATa his3-Δ200 leu2-Δ1 lys2-801 trp1-Δ63 ura3-52 CDC12-Vn::TRP1 [pRS316 CDC12] | |
| MMY0224 | BY4742 SPR3-Vn::URA3 | This study |
Sequence analysis of the relevant coding regions from these strains confirmed the mutant alleles to be cdc10(D182N; cdc10-1) and cdc12(G247E; cdc12-1).
In both GCFY5 and GCFY6, SPR28 is under control of the endogenous CDC11 promoter and SPR3 is under control of the endogenous CDC12 promoter, and both GCFY5 and GCFY6 may also harbor the original, URA3-marked covering plasmids pSB1/pJT1520 and/or pMVB39/pJT1622. Subsequent strain propagation was performed to select for the presence of either pGCF1 or pGCF2 (or both) for certain experiments.
Spore from cross of YMVB1 with JTY5167.
BY4741 was transformed with a PCR product amplified from YCpH-cdc12-6 that included the 3′ end of the cdc12-6 coding sequence, the out-of-frame GFP coding sequence, and downstream HIS3 marker.
JTY3993 was transformed with BamHI–cut pSC193.
Spore from cross of JTY5200 with CBY06417.
Spore from cross of JTY5200 with CBY05110.
BY4741 was transformed with BamHI-cut pSC193.
To construct yeast strains expressing fluorescently-tagged Spr3 or Spr28 (or both) during vegetative growth, a plasmid-derived DNA fragment encoding the sporulation-specific septin was integrated in place of the endogenous locus for a mitotic septin subunit (CDC12 or CDC11, respectively), as follows. A PCR fragment containing the CDC11prom-SPR28-GFP-ADH(t)-HygR (treated with DpnI restriction endonuclease to destroy any intact vector) was used to transform a cdc11Δ::KanR yeast strain (covered by pRS316-CDC11), where the cdc11Δ::KanR allele represents a full deletion of the CDC11 coding sequence and the drug-resistance cassette is in the same orientation as the ORF, leaving the promoter region and the MX4 cassette terminator, which is identical between the drug-resistance cassettes (Goldstein and McCusker, 1999), to mediate the homologous recombination to achieve integration at the CDC11 locus. A similar strategy was used to integrate CDC12prom-SPR3-GFP-ADH(t)-HygR. To create a strain coexpressing both SPR28 and SPR3, the individual strains (one MATa, the other MATα) were first transformed with differentially marked covering plasmids expressing the corresponding wild-type septin (CDC11::URA3-HIS3 and CDC12::URA3-LEU2, respectively), then mated together. After diploid selection and sporulation, tetrads were dissected and the desired haploid spores were identified by selection for the drug resistance markers diagnostic of the integrated alleles and the covering plasmids and then confirmed by growth phenotype, diagnostic PCR, and immunoblot analysis and fluorescence microscopy.
MMY0231 (MATa/MATα spr3Δ/spr3Δ spr28Δ/spr28Δ) was created by mating appropriate spore clones isolated from a cross of MMY0220 and MMY0152 (Table 1). To generate strain GCFY7, which lacks Spr28 and Spr3 and expresses in their place (and in a meiosis-specific manner) both Cdc11 and Cdc12, respectively, the SPR28 locus was deleted in BY4742, yielding GFY-853 (MATα spr28Δ::KanR), followed by deletion of the SPR3 locus producing GFY-885 (MATα spr28Δ::KanR spr3ΔHygR). Next, plasmids were constructed that express CDC11-GFP::NatR (pGF-IVL291) and CDC12-mCherry::KanR (pGF-IVL295) under control of the SPR28 and SPR3 promoters, respectively, using in vivo ligation and homologous recombination (Finnigan and Thorner, 2015). After PCR amplification with appropriate primers containing the necessary regions of homology and DpnI treatment, the CDC11-GFP-containing PCR product was integrated at the SPR28 locus (to generate strain GFY-920) and the CDC12-mCherry-containing PCR product was integrated at the SPR3 locus (creating strain GFY-983). Then, a URA3-based Gal-inducible HO-expressing plasmid (pJT2800) was introduced into GFY-983 to generate a derivative in which the mating type was switched from MATα to MATa (and the plasmid was removed by selection on medium containing 5-FOA), yielding GFY-1041. Lastly, the NatR marker at the SPR28 locus in GFY-983 was swapped to HygR, generating strain GFY-1030. Multiple diagnostic PCR reactions and nucleotide sequence analysis confirmed proper integration at each modified locus, as well as MAT identity. Finally, GCFY7 was created by (1) mating GFY-1030 with GFY-1041 on YPD medium for 24 h at 30°C and (2) two successive growth selections for clonal diploid isolates on rich medium containing both Nourseothricin (clonNat) and Hygromycin. BY4743 spr3Δ::kanMX/spr3Δ::kanMX and BY4743 spr28Δ::kanMX/spr28Δ::kanMX strains were constructed by first recreating the haploid deletion strains in BY4741 and BY4742, via transformations of those strains with PCR products that amplified the spr3Δ::kanMX or spr28Δ::kanMX cassette, including several hundred base pairs upstream and downstream of the former SPR gene ORF. These haploid mutants were then mated together to form the homozygous diploid strains. The same haploid transformation method was used to introduce the deletion alleles into strains carrying the SK1 alleles at TAO3, MKT1, and RME1. To make strain MMY0225, a PCR product made with a primer annealing to the 5′ region of the SHS1 ORF and including 40 nt upstream of the SPR28 ORF was used with an appropriate reverse primer including 40 nt downstream of the SPR28 stop codon to amplify SHS1-VC::HIS3MX6 from genomic DNA of Y0619, and this product was transformed into GFY-983. This strain was mated with FY2839 to create MMY0226. To integrate CDC12-eCitrine, PCR was used to amplify the CDC12-eCitrine::his5MX cassette from plasmid pML113, and the DpnI-digested product was transformed into the appropriate strain.
Plasmids and their construction
Plasmids (Table 2) expressing fluorescently tagged Spr28 and Spr3 under control of the endogenous CDC11 and CDC12 promoters were constructed by in vivo ligation and homologous recombination (Muhlrad et al., 1992; Finnigan and Thorner, 2015) in strain SF838-1Da (Rothman and Stevens, 1986), as follows. Plasmid pRS315 containing 500 bp of the CDC11 promoter was gapped and cotransformed into yeast with PCR-generated linear DNA fragments containing full-length SPR28 ORF and the GFP-ADH1(t)-HygR cassette (Goldstein and McCusker, 1999), each with corresponding 30-bp homologous tails. A similar procedure was used to create the SPR3-mCherry–expressing vector. To create vectors containing wild-type CDC11 or CDC12 that were differentially marked (in addition to containing URA3 for counterselection on 5-FOA), the same in vivo ligation approach was used to create pRS313-CDC11-ADH1(t)-CaURA3 and pRS315-CDC12-ADH1(t)-CaURA3. Constructs were recovered from yeast by rescue in E. coli and confirmed via diagnostic PCR and DNA sequencing.
Plasmids used in this study
| Plasmid | Description | Reference |
|---|---|---|
| pRS315 | CEN, LEU2 | Sikorski and Hieter, 1989 |
| pRS313 | CEN, HIS3 | Sikorski and Hieter, 1989 |
| pLP29 | pRS313; CDC12-GFP | Lippincott and Li, 1998 |
| pSB1 / pJT1520 | CEN, URA3, CDC11 | Versele et al., 2004 |
| pMVB39 / pJT1622 | CEN, URA3, CDC12 | Versele and Thorner, 2004 |
| pSC193 | URA3, prGAL1/10-NDT80-HA | Chu and Herskowitz, 1993 |
| YCpH-cdc12-6a | pLP29; cdc12-6 | This study |
| pGCF1 | pRS313; prCDC11-CDC11-ADH1(t)-CaURA3 | This study |
| pGCF2 | pRS315; prCDC12-CDC12-ADH1(t)-SpHIS5 | This study |
| pGCF3 | pRS315; prCDC11-SPR28-GFP-ADH1(t)-HygR | This study |
| pGCF4 | pRS313; prCDC12-SPR3-mCherry-ADH1(t)-KanR | This study |
| p3.8 | CDC3, CDC10, His6-SPR3 in E. coli | This study |
| p4.7 | CDC3, CDC10, His6-SPR3, SPR28 in E. coli | This study |
| p4.10 | CDC3, CDC10, His6-SPR3, CDC11 in E. coli | This study |
| pJT2485 | p406-GFP-2XPHOsh2 | Roy and Levine, 2004 |
| pML113 | CEN, LEU2, CDC12-eCitrine::his5MX | Nagaraj et al., 2008 |
| Plasmid | Description | Reference |
|---|---|---|
| pRS315 | CEN, LEU2 | |
| pRS313 | CEN, HIS3 | |
| pLP29 | pRS313; CDC12-GFP | |
| pSB1 / pJT1520 | CEN, URA3, CDC11 | |
| pMVB39 / pJT1622 | CEN, URA3, CDC12 | |
| pSC193 | URA3, prGAL1/10-NDT80-HA | Chu and Herskowitz, 1993 |
| YCpH-cdc12-6 | pLP29; cdc12-6 | This study |
| pGCF1 | pRS313; prCDC11-CDC11-ADH1(t)-CaURA3 | This study |
| pGCF2 | pRS315; prCDC12-CDC12-ADH1(t)-SpHIS5 | This study |
| pGCF3 | pRS315; prCDC11-SPR28-GFP-ADH1(t)-HygR | This study |
| pGCF4 | pRS313; prCDC12-SPR3-mCherry-ADH1(t)-KanR | This study |
| p3.8 | CDC3, CDC10, His6-SPR3 in E. coli | This study |
| p4.7 | CDC3, CDC10, His6-SPR3, SPR28 in E. coli | This study |
| p4.10 | CDC3, CDC10, His6-SPR3, CDC11 in E. coli | This study |
| pJT2485 | p406-GFP-2XPHOsh2 | |
| pML113 | CEN, LEU2, CDC12-eCitrine::his5MX |
The cdc12-6 allele was first introduced into a CDC12-GFP gene on pLP29 by digestion with BfuAI and co-transformation of the cut plasmid into YMVB61 along with the PCR-amplified cdc12-6 gene from DDY1462. Plasmids from transformants that displayed temperature sensitivity upon loss of the URA3-marked CDC12 plasmid were rescued to E. coli and sequenced to confirm the presence of the cdc12-6 allele (K391N L392stop).
Assessment of yeast growth rates and morphologies
In a 96-well plate, eight 100-µl cultures of each of six yeast strains were grown in YP containing 2% raffinose (“YPRaf”) or 2% raffinose plus 0.05% galactose (“YPRafGal”) at 25.4°C inside a BioTek Synergy HT plate reader, and A630 nm was measured every 10 min over 15 h (in between reads, the plate was agitated on the medium setting). Growth rates and associated standard errors were calculated using Prism 5.0d (GraphPad Software). Cells from these cultures, or from agar plates, were resuspended in H2O and examined at 22°C by transmitted light using an EVOSfl microscope (Advanced Microscopy Group) equipped with a monochrome charge-coupled device (ICX285AL; Sony), 2/3” 1,360 × 1,024, 1.4-Megapixel camera and an Olympus 60× PlanApo 1.42 NA objective and categorized. Images were captured using the software built into the microscope, and cropped and contrast-adjusted using Adobe Photoshop. In other experiments, yeast cultures were grown overnight in synthetic medium with 2% glucose with selection for covering plasmids; for cells coexpressing both Spr28 and Spr3, selection for the vectors containing the wild-type copies of CDC11 and CDC12 was used. In complementation tests, fivefold serial dilutions of the strains were spotted on solid synthetic-complete medium with 2% glucose lacking or containing 5-FOA, and scored after incubation at 30°C for 2–3 d.
Fluorescence microscopy and imaging
Yeast were grown overnight in synthetic medium selective for covering plasmid(s), diluted to an A600 nm of 0.25/ml in YPD medium, and grown for 4–4.5 h (until A600 nm was ∼1). The resulting cells were harvested, washed with water, and examined using a BH-2 epifluorescence microscope (Olympus) under a 100× objective. Images were obtained using a charge-coupled device camera (Olympus), Magnafire SP software (Optronics), and Adobe Photoshop. To quantify the fraction of cells with septin localization at the bud neck, images of cells (n = 100–250) for two technical replicates from at least three independent trials were scored (error represents the standard deviation of the mean of these measurements). Images of control cells (n = 50–100) expressing GFP fused to the C termini of Cdc11 or Cdc12 were also scored; similar images and quantification were obtained mCherry was used in place of GFP. Cells that displayed an extreme elongated morphology (caused by loss of the wild-type septin) were excluded from the analysis. For the strain expressing both Spr28-GFP and Spr3-mCherry, only cells displaying detectible GFP signal were scored.
Preparation of cell extracts and immunoblotting
Yeast strains were grown overnight in selective medium, diluted to an A600 nm = 0.25/ml in fresh medium and grown to A600 nm ∼1. Cells from samples (2 ml) of the cultures were harvested and frozen at −80°C. The frozen cells were resuspended and lysed in 1.85 M NaOH and 7.4% 2-mercaptoethanol (2-ME) and protein was precipitated using 50% trichloroacetic acid on ice for 10 min. After washing the resulting precipitates in acetone, protein was solubilized in 5% SDS and 0.1 M Tris, followed by addition of 0.2 volumes of a 5× stock of SDS gel sample buffer (10% SDS, 50% glycerol, 7.5 mM bromophenol blue, 0.715 M 2-ME, and 0.25 M Tris, pH 6.8). After boiling and clarification by brief centrifugation in a microfuge, samples (12 µl) were resolved by SDS-PAGE, transferred to nitrocellulose, probed with rabbit polyclonal anti-Pgk1 (Baum et al., 1978), mouse monoclonal anti-GFP (Roche Applied Sciences), or rabbit polyclonal anti-DsRed (Rockland) primary antibodies and appropriate infrared dye-conjugated secondary antibodies (Li-Cor) and visualized using an Odyssey infrared imaging device (Li-Cor).
Assessment of sporulation proficiency
The cells in triplicate samples (250 µl) of liquid cultures of the MATa/MATα strains to be tested, which had been grown to saturation in rich medium (YPGlc), were collected by centrifugation, washed twice with sterile H2O, and resuspended in 2 ml sporulation medium (1% potassium acetate, 0.05% glucose, 20 mg/ml leucine, and 40 mg/ml uracil). The resulting cell suspensions were incubated at room temperature (∼22°C) with gentle aeration on a rollerdrum and then examined after ∼5 d. A total of at least 300 individual cells were scored per sample; values obtained for any given genotype were averaged for at least three-to-six independent trials. Mature spores were identified as bright spheres of ≤5 µm diameter because completed spore walls refract the illuminating light when viewed by standard transmission microscopy. This method for scoring sporulation efficiency was validated by two independent means: (1) introducing into the strains tested a plasmid expressing a fluorescently tagged histone, Htb2-mCherry (Westfall et al., 2008), to accentuate the number of nuclear lobes present at meiosis II; and (2) introducing into the strains tested a plasmid expressing a fluorescently tagged exo–β-glucanase, Spr1-GFP (Suda et al., 2009), that specifically decorates the wall of mature spores (Heasley and McMurray, 2016). The differences between the sporulation efficiency (% asci) of the wild-type controls and the other strains examined were statistically significant to better than the 95% confidence level based on the standard one-tailed Fisher’s exact test.
FRAP
FRAP was performed as previously described (Maddox et al., 2000; Molk et al., 2004). In brief, cells expressing either CDC10-GFP or GFP-2X(PHOsh2) were imaged at 25°C using an inverted TiE microscope (Nikon) equipped with a Xyla CMOS camera (Andor) and a 100× PlanApo 1.4 NA objective. Photobleaching was performed using a Coherent 488 nm Sapphire 50-mW laser through an LU4A laser module (Nikon) controlled by Nikon Elements software. Three prebleach images (600-ms exposure) were acquired to establish a baseline control for the initial fluorescence. A single focused 50-ms laser pulse was used to photobleach a Cdc10-GFP–containing horseshoe in sporulating cells (or, for GFP-2X(PHOsh2), a well-decorated portion of the PSM). The photobleaching treatment eliminated 75–95% of the original fluorescence. Immediately thereafter, single plane images (600-ms exposures) were acquired every 10 s for ∼2 min to follow any recovery. Such photobleaching experiments were performed on 15 different cells. Using ImageJ analysis software (National Institutes of Health), the observed fluorescence intensity values were corrected for background and photobleaching during image acquisition and displayed as the mean relative value for the bleached and unbleached regions for all 15 cells.
Bimolecular fluorescence complementation
MATa/MATα diploids containing the proteins of interest fused to the two nonfluorescent halves of the Venus derivative (F46L F64L M153T V163A S175G; Rekas et al., 2002) of the YFP variant of GFP (Griesbeck et al., 2001) were made by mating the appropriate haploid strains, the majority of which were generously provided by E. Bi (University of Pennsylvania, Philadelphia, PA). The diploids were induced to sporulate as described in the Assessment of sporulation proficiency section and spotted in water directly from the sporulation cultures onto “pads” of 2% agarose, and any resulting yellow fluorescence arising from the reconstitution of the chromophore upon association of the interacting proteins (Kerppola, 2008) was imaged at 22°C using the EVOSfl microscope and 60× objective with a YFP filter cube (excitation, 500 nm; emission, 542 nm). Images were captured using the software built into the microscope and cropped and contrast-adjusted using Photoshop and/or ImageJ.
Online supplemental material
Table S1 catalogs the sequence relatedness among all seven S. cerevisiae septins. Fig. S1 depicts sequence features of the sporulation-specific septins. Fig. S2 shows that switching fluorescent tags on Spr3 and Spr28 does not alter their corecruitment to the bud neck in vegetative cells. Fig. S3 shows that ectopic expression of SPR3 and/or SPR28 does not impede the growth of wild-type mitotic cells. Fig. S4 shows that ectopic expression of sporulation-specific septins does exacerbate the growth defects of mitotic cells carrying mutations in mitosis-specific septins. Fig. S5 shows that expression of sporulation-specific septins driven by low-level ectopic expression of transcription factor Ndt80 in a cdc12-1 mutant causes an increased frequency of mitotic cells with an aberrant morphology.
Acknowledgments
We thank A. Neiman, M. Onishi, and J. Pringle for helpful discussions and the communication of unpublished results; A. Vershon for providing the NDT80 plasmid; E. Bi and Fred Winston for the generous gift of strains; and the QB3 MacroLab (University of California, Berkeley), established with and supported by funds provided by the W.M. Keck Foundation, for help preparing some of the constructs used for expression and purification of the sporulation-specific septins.
This work was supported by a National Science Foundation predoctoral fellowship (to G. Garcia III), a postdoctoral fellowship from the Adolph C. and Mary Sprague Miller Institute for Basic Research in Science, University of California Berkeley (to G.C. Finnigan), a National Institutes of Health Predoctoral Traineeship GM008730 (to L.R. Heasley), a National Institutes of Health R01 research grant GM099820 (to C.G. Pearson), a National Institutes of Health K99/R00 Pathway to Independence Award GM086603 (to M.A. McMurray), and a National Institutes of Health R01 research grant GM101314 (to E. Nogales and J. Thorner). E. Nogales is a Howard Hughes Medical Institute Investigator.
The authors declare no competing financial interests.
References
Author notes
G. Garcia III, G.C. Finnigan, and L.R. Heasley contributed equally to this paper.
G. Garcia III’s present address is Dept. of Biochemistry and Biophysics and Cardiovascular Research Institute, University of California, San Francisco, San Francisco, CA 94158.