In healthy hearts, myofilaments become more sensitive to Ca2+ as the myocardium is stretched. This effect is known as length-dependent activation and is an important cellular-level component of the Frank–Starling mechanism. Few studies have measured length-dependent activation in the myocardium from failing human hearts. We investigated whether ischemic and non-ischemic heart failure results in different length-dependent activation responses at physiological temperature (37°C). Myocardial strips from the left ventricular free wall were chemically permeabilized and Ca2+-activated at sarcomere lengths (SLs) of 1.9 and 2.3 µm. Data were acquired from 12 hearts that were explanted from patients receiving cardiac transplants; 6 had ischemic heart failure and 6 had non-ischemic heart failure. Another 6 hearts were obtained from organ donors. Maximal Ca2+-activated force increased at longer SL for all groups. Ca2+ sensitivity increased with SL in samples from donors (P < 0.001) and patients with ischemic heart failure (P = 0.003) but did not change with SL in samples from patients with non-ischemic heart failure. Compared with donors, troponin I phosphorylation decreased in ischemic samples and even more so in non-ischemic samples; cardiac myosin binding protein-C (cMyBP-C) phosphorylation also decreased with heart failure. These findings support the idea that troponin I and cMyBP-C phosphorylation promote length-dependent activation and show that length-dependent activation of contraction is blunted, yet extant, in the myocardium from patients with ischemic heart failure and further reduced in the myocardium from patients with non-ischemic heart failure. Patients who have a non-ischemic disease may exhibit a diminished contractile response to increased ventricular filling.
Introduction
The Frank–Starling relationship is critical for normal cardiac function because it allows ventricular output to increase in response to elevated filling pressures (Starling, 1918; Frank, 1959). The precise mechanisms underlying the Frank–Starling response remain unclear but are thought to include intracellular effects of the cardiomyocyte being stretched. (Allen and Kentish, 1985; Farman et al., 2010; Campbell, 2011; Sagawa et al., 1988; de Tombe et al., 2010). Length-dependent changes in calcium handling contribute to the response (Fabiato and Fabiato, 1975; Parmley and Chuck, 1973; Vahl et al., 1998; Allen and Kurihara, 1982; Kentish et al., 1986), but the primary mechanisms driving the Frank–Starling relationship probably stem from the myofilaments (i.e., the thick filaments of myosin and thin filaments of actin that are responsible for myocardial force production; Farman et al., 2010; Hibberd and Jewell, 1982; Smith et al., 2009; Cazorla et al., 2001; McDonald and Moss, 1995; Milani-Nejad et al., 2015; Hanft and McDonald, 2010). As chemically permeabilized cardiac cells are stretched, they become more sensitive to Ca2+ and develop more force. This contractile response is known as length-dependent activation and Ca2+ sensitization is thought to be the most important component of the Frank–Starling response (Campbell, 2011).
Given its physiological importance, it is surprising that length-dependent activation has only been studied a few times using human myocardium. Important questions about the effect on patients remain unanswered. In particular, it is unclear whether length-dependent increases in Ca2+ sensitivity are systematically modulated in human heart failure. Several publications have shown that length-dependent activation is reduced or eliminated in failing human tissue (Kitzman et al., 1991; van Dijk et al., 2012; Sequeira et al., 2013; Schwinger et al., 1994; McDonald et al., 2020), while others demonstrate that the length-dependence of Ca2+ sensitivity is maintained in diseased hearts (Milani-Nejad et al., 2015; Vahl et al., 1997; Holubarsch et al., 1996). We were intrigued by these reports and hypothesized that the variability of the responses might reflect differences in the clinical phenotype of the patients who donated samples.
Clinicians characterize heart failure in different ways, but the diagnoses documented in medical records are often simpler than scientists might expect. For example, the genotype of patients who receive cardiac transplants is often unknown. At the University of Kentucky, (a US academic medical center that performs 0.5–1% of all cardiac transplants worldwide each year) the most common distinction noted in the medical records is whether a patient developed heart failure subsequent to myocardial infarction (“ischemic heart failure”) or due to some other, often unknown, reason (“non-ischemic heart failure”). We reasoned that ischemic and non-ischemic diseases might produce distinct cellular-level phenotypes and developed an experimental design to test this hypothesis.
Our study compared Ca2+-activated force production in permeabilized myocardial strips isolated from the left ventricle of organ donors and patients who had ischemic or non-ischemic heart failure. Maximal force increased in samples from all hearts as the tissue was stretched from a sarcomere length of 1.9 to 2.3 μm. The myocardium from organ donors exhibited a robust length-dependent increase in Ca2+ sensitivity, indicating that the tissue was activated at a lower Ca2+ concentration when it was stretched. This length-dependent activation response was blunted—but still present—in preparations from patients with ischemic heart failure. The length-dependent increase in Ca2+ sensitivity was eliminated with non-ischemic heart failure. Comparable changes in cardiac troponin I (cTnI) phosphorylation accompanied these differences in Ca2+ sensitivity, in that cTnI phosphorylation was greatest among donors, decreased moderately with ischemic heart failure, and was lowest in samples from patients with non-ischemic heart failure. These findings suggest that patients who have non-ischemic heart failure could exhibit a smaller cellular-level contractile response to increased ventricular filling than patients who have an ischemic disease and that decreases in cTnI phosphorylation may compromise length-dependent activation with heart failure.
Materials and methods
Human tissue samples
Contractile mechanics and hydroxyproline assays used cardiac samples that were obtained from 12 patients who received heart transplants at the University of Kentucky and from 6 organ donors who did not have heart failure (Tables 1 and 2). Their mean age was 47 (range 10–61) for donors, 57 (range 49–71) for patients with ischemic heart failure, and 51 (range 20–65) for patients with non-ischemic heart failure. 66% of these donors and 50% of these patients with heart failure were female.
Cardiac sample characteristics for organ donors
| Sample ID . | Age (yr) . | Sex . | Race . | Diabetic . | Medications . | Cause of death . |
|---|---|---|---|---|---|---|
| FC3CBa,b | 10 | F | Whited | No | No data | Anaphylaxis, anoxia |
| 24713a | 47 | F | Whited | No | No data | Head trauma |
| B23E3a,b,c | 50 | M | Whited | No | No data | Stroke |
| BE497a,b | 56 | M | Black | No data | No data | Cardiac arrest |
| 31331a,b | 58 | F | Whited | No | No data | Anoxia |
| CF462a | 61 | F | Whited | Yes | No data | Stroke |
| 4B3FAb,c | 37 | F | White | No | β-blocker | Anoxia |
| 632FDb,c | 32 | M | White | No | Aspirin | Anoxia |
| 8CB30b,c | 61 | F | White | No | β-blocker, vasopressor | Head trauma |
| Sample ID . | Age (yr) . | Sex . | Race . | Diabetic . | Medications . | Cause of death . |
|---|---|---|---|---|---|---|
| FC3CBa,b | 10 | F | Whited | No | No data | Anaphylaxis, anoxia |
| 24713a | 47 | F | Whited | No | No data | Head trauma |
| B23E3a,b,c | 50 | M | Whited | No | No data | Stroke |
| BE497a,b | 56 | M | Black | No data | No data | Cardiac arrest |
| 31331a,b | 58 | F | Whited | No | No data | Anoxia |
| CF462a | 61 | F | Whited | Yes | No data | Stroke |
| 4B3FAb,c | 37 | F | White | No | β-blocker | Anoxia |
| 632FDb,c | 32 | M | White | No | Aspirin | Anoxia |
| 8CB30b,c | 61 | F | White | No | β-blocker, vasopressor | Head trauma |
Tissue samples were used for the permeabilized muscle mechanics experiments, and hydroxyproline assays.
Tissue samples were used for the Phos-tag assays.
Tissue samples were used for the cMyBP-C extraction for the Pro-Q diamond phospho-stain assays.
Not Hispanic or Latino.
Cardiac sample characteristics for patients with heart failure
| Sample ID . | Age (yr) . | Sex . | Race . | Heart failure . | Diabetic . | β-blockers . | ACE inhibitor or ARB . | Statin . | Aspirin . | Aldosterone antagonist . | Inotrope . |
|---|---|---|---|---|---|---|---|---|---|---|---|
| B8BE2a,b,c | 49 | M | Whited | Ischemic | No | Yes | Yes | Yes | No | Yes | No |
| 0894Ea,b | 50 | F | Whited | Ischemic | Yes | Yes | No | Yes | Yes | Yes | Yes |
| EF5CBa,b,c | 56 | M | Whited | Ischemic | No | Yes | No | Yes | No | Yes | Yes |
| 7CE52a,b | 57 | F | Whited | Ischemic | No | Yes | No | No | Yes | Yes | Yes |
| 4E8C1a,b,c | 60 | F | Whited | Ischemic | Yes | Yes | Yes | Yes | Yes | No | Yes |
| 246D8a,b,c | 71 | M | Whited | Ischemic | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 14C39b | 38 | M | Whited | Ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| 6634Fb | 60 | F | Black | Ischemic | Yes | Yes | Yes | No | Yes | Yes | Yes |
| 8E8D8b | 51 | M | White | Ischemic | No | Yes | No | Yes | Yes | No | Yes |
| B0644b | 49 | F | Whited | Ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| A3CD2a | 20 | M | Black | Non-ischemic | No | Yes | No | No | Yes | No | No |
| 70146a,b,c | 48 | M | Whited | Non-ischemic | No | No | Yes | No | No | Yes | Yes |
| 117DDa | 56 | F | Whited | Non-ischemic | No | No | Yes | No | No | No | Yes |
| 105A1a,b | 57 | M | Whited | Non-ischemic | No | Yes | Yes | No | Yes | Yes | Yes |
| 668C5a | 60 | F | Whited | Non-ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| 73CE5a | 65 | F | Whited | Non-ischemic | No | Yes | No | Yes | Yes | Yes | Yes |
| DC737b,c | 17 | F | Whited | Non-ischemic | No | Yes | Yes | No | No | No | No |
| C9BAEb,c | 23 | F | Whited | Non-ischemic | No | Yes | No | No | No | No | No |
| B25DFb,c | 54 | M | Whited | Non-ischemic | No | Yes | Yes | No | No | Yes | No |
| AFAA5b | 68 | M | Black | Non-ischemic | No | No | No | Yes | No | Yes | Yes |
| 61BACb | 51 | M | Black | Non-ischemic | Yes | Yes | No | Yes | No | Yes | No |
| 93EEDb | 61 | M | Black | Non-ischemic | No | Yes | No | Yes | Yes | Yes | Yes |
| 32B32b | 67 | M | Whited | Non-ischemic | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 406DEb | 47 | M | Black | Non-ischemic | Yes | No | No | No | No | Yes | Yes |
| Sample ID . | Age (yr) . | Sex . | Race . | Heart failure . | Diabetic . | β-blockers . | ACE inhibitor or ARB . | Statin . | Aspirin . | Aldosterone antagonist . | Inotrope . |
|---|---|---|---|---|---|---|---|---|---|---|---|
| B8BE2a,b,c | 49 | M | Whited | Ischemic | No | Yes | Yes | Yes | No | Yes | No |
| 0894Ea,b | 50 | F | Whited | Ischemic | Yes | Yes | No | Yes | Yes | Yes | Yes |
| EF5CBa,b,c | 56 | M | Whited | Ischemic | No | Yes | No | Yes | No | Yes | Yes |
| 7CE52a,b | 57 | F | Whited | Ischemic | No | Yes | No | No | Yes | Yes | Yes |
| 4E8C1a,b,c | 60 | F | Whited | Ischemic | Yes | Yes | Yes | Yes | Yes | No | Yes |
| 246D8a,b,c | 71 | M | Whited | Ischemic | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 14C39b | 38 | M | Whited | Ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| 6634Fb | 60 | F | Black | Ischemic | Yes | Yes | Yes | No | Yes | Yes | Yes |
| 8E8D8b | 51 | M | White | Ischemic | No | Yes | No | Yes | Yes | No | Yes |
| B0644b | 49 | F | Whited | Ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| A3CD2a | 20 | M | Black | Non-ischemic | No | Yes | No | No | Yes | No | No |
| 70146a,b,c | 48 | M | Whited | Non-ischemic | No | No | Yes | No | No | Yes | Yes |
| 117DDa | 56 | F | Whited | Non-ischemic | No | No | Yes | No | No | No | Yes |
| 105A1a,b | 57 | M | Whited | Non-ischemic | No | Yes | Yes | No | Yes | Yes | Yes |
| 668C5a | 60 | F | Whited | Non-ischemic | No | Yes | Yes | Yes | Yes | Yes | Yes |
| 73CE5a | 65 | F | Whited | Non-ischemic | No | Yes | No | Yes | Yes | Yes | Yes |
| DC737b,c | 17 | F | Whited | Non-ischemic | No | Yes | Yes | No | No | No | No |
| C9BAEb,c | 23 | F | Whited | Non-ischemic | No | Yes | No | No | No | No | No |
| B25DFb,c | 54 | M | Whited | Non-ischemic | No | Yes | Yes | No | No | Yes | No |
| AFAA5b | 68 | M | Black | Non-ischemic | No | No | No | Yes | No | Yes | Yes |
| 61BACb | 51 | M | Black | Non-ischemic | Yes | Yes | No | Yes | No | Yes | No |
| 93EEDb | 61 | M | Black | Non-ischemic | No | Yes | No | Yes | Yes | Yes | Yes |
| 32B32b | 67 | M | Whited | Non-ischemic | Yes | Yes | Yes | Yes | No | Yes | Yes |
| 406DEb | 47 | M | Black | Non-ischemic | Yes | No | No | No | No | Yes | Yes |
ACE, angiotensin converting enzyme; ARB; angiotensin receptor blocker.
Tissue samples were used for the permeabilized muscle mechanics experiments, and hydroxyproline assays.
Tissue samples were used for the Phos-tag assays.
Tissue samples were used for the cMyBP-C extraction for the Pro-Q diamond phospho-stain assays.
Not Hispanic or Latino.
Phos-tag gel electrophoresis assays used separate cardiac samples from 20 patients receiving heart transplants and from 7 organ donors who did not have heart failure (Tables 1 and 2). Their mean age was 43 (range 10–61) for donors, 54 (range 38–71) for patients with ischemic heart failure, and 49 (range 17–68) for patients with non-ischemic heart failure. 57% of these donors and 35% of these patients with heart failure were female.
As previously described (Blair et al., 2016), the hearts were passed to a researcher as soon as they were excised from the body, immediately placed in ice-cold saline slush, and transported back to the laboratory in an insulated box (a 3-min walk), where tissue samples (∼500 mg) were snap-frozen in liquid nitrogen and stored at −150°C within ∼20 min. Mid-myocardial sections of the left ventricular free wall were shipped overnight on dry ice to Washington State University and stored at −80°C for 1–3 wk, until they were dissected for mechanics and biochemical experiments. Phos-tag gels were performed at King’s College London using samples that were shipped to Britain using a dry shipper that maintained the samples in the vapor phase of liquid nitrogen. All procedures were approved by the University of Kentucky Institutional Review Board, and the subjects or their legally authorized representatives gave written informed consent (Protocol 46103).
Permeabilized myocardial strips
Muscle mechanics solution concentrations were formulated by solving equations describing ionic equilibria according to Godt and Lindley (1982), and all concentrations are listed in mM unless otherwise noted. Skinning solution: 50 BES, 30.83 K propionate, 10 Na azide, 20 EGTA, 6.29 MgCl2, 6.09 ATP, 1 DTT, 20 BDM, 50 µM Leupeptin, 275 µM Pefabloc, and 1 µM E-64, with 1% Triton-X100 (wt/vol), pH 7.0, and 50% glycerol (wt/vol). Storage solution: same as skinning solution without Triton-X100. Relaxing solution: pCa 8.0 (pCa = −log10[Ca2+]), 5 EGTA, 5 MgATP, 1 Mg2+, 0.3 Pi, 35 phosphocreatine, 300 U/ml creatine kinase, pH 7.0, and 200 ionic strength adjusted with Na methanesulfonate. Activating solution: same as relaxing solution, but with pCa 4.8.
Frozen tissue sections were thawed in ice-cold skinning solution and dissected into thin strips (∼180 μm in diameter and 700 μm long). Strips were skinned overnight at 4°C, transferred to the storage solution, and stored at −20°C for 3–5 d. On the day of the experiments, the strips were mounted between a motor (P841.40; Physik Instrumente) and a strain gauge (AE801; Kronex), lowered into a 30-μl droplet of relaxing solution maintained at physiological temperature (37°C), and stretched to a sarcomere length of 1.9 or 2.3 μm as measured by digital Fourier transform analysis (IonOptix Corp).
Strips were activated in solutions with pCa values ranging from 8.0 to 4.8 to measure the isometric force–pCa relationship. Force values were normalized to the cross-sectional area of each preparation and reported as stress values with units of kN m−2. Each strip was only measured at one sarcomere length. Preliminary attempts to measure back-to-back force–pCa curves were inconsistent, even under identical solution conditions at a single sarcomere length. A portion of this may follow from the experiments being performed at physiological temperature, wherein we saw a binary response regarding preparation stability throughout an experiment; preparations either produced a robust force–pCa response or they failed to activate well and did not maintain a force response consistent enough to fit to Eq. 1. A total of 158 strips were analyzed (three to five from each heart at each sarcomere length).
Biochemical analysis
Myocardial collagen content was assessed using colorimetric assays for hydroxyproline. 15 mg of frozen tissue from each heart sample was boiled in 200 µl of 6 M HCl at 110°C overnight. 5 µl aliquots of hydrolyzed tissue were added to 80 µl of 100% isopropanol, then mixed with 40 µl of reaction buffer 1, and allowed to react for 5 min at room temperature. Next, 0.5 ml of reaction buffer 2 was added to the mixture and incubated in a water bath at 55°C for 30 min. Vials were then placed on ice for 5 min and centrifuged at 5,000 × g for 1 min at 4°C. 200 µl aliquots were placed in a 96-well plate and the absorbance was measured at 558 nm. Reaction buffer 1 was composed of a 1:4 solution ratio between 7% chloramine-T solution (w/v, in water) and acetate citrate buffer (0.695 M sodium acetate, 0.174 M citric acid, 0.435 M NaOH, and 38.5% [v/v] isopropanol). Reaction buffer 2 was composed of a 3:13 solution ratio between Ehrlich’s solution (6 g of p-dimethylaminobenzaldehyde mixed with 20 ml of ethanol and 1.35 ml of sulfuric acid) and isopropanol. A standard curve of trans-4-hydroxy-L-proline (0–500 μM) was used to determine the hydroxyproline concentration for each sample.
The relative phosphorylation of cTnI was measured using Phos-tag gel electrophoresis and Western blot. Human ventricular tissue was pulverized in a liquid nitrogen–cooled mortar, resuspended in SDS-PAGE loading buffer containing 4 mol/liter urea, and the samples were denatured by incubation at 100°C for 3–4 min. Debris was removed by centrifugation at 15,000 × g for 5 min at 4°C and the clear supernatant was transferred to a fresh Eppendorf tube. Samples were run on Phos-tag–SDS-PAGE gels (containing 50 μmol/liter Phos-tag reagent and 100 μmol/liter MnCl2) for 2–3 h at 120 V. Phos-tag gels were washed first with transfer buffer (in mmol/liter: 25 Tris-HCl, pH 8.3, 192 glycine, 0.08% [v/v] SDS, and 15% [v/v] methanol) containing 10 mmol/liter EDTA for 10 min, followed by three washes in transfer buffer without EDTA. Proteins were blotted onto nitrocellulose (BIO-RAD) membranes for 1 h at 1 mA/cm2 in the transfer buffer using a Trans-Blot SD Semi-Dry Electrophoretic Transfer Cell (BIO-RAD). After blotting, membranes were blocked with Tris-buffered saline containing 0.05% (v/v) Tween-20 (TBS-T) and 5% (w/v) semi-skimmed milk powder for 1 h at room temperature. Membranes were incubated overnight in TBS-T containing 5% (w/v) semi-skimmed milk powder and rabbit monoclonal α-RLC antibody (EPR3741, 1:2,000 dilution; Abcam) or mouse anti-cTnI antibody (clone 84; 1:2,000 dilution; HyTest). Membranes were washed three times in TBS-T and incubated for 1 h at room temperature with secondary antibody (1:1,000 dilution, HRP-conjugated α rabbit; Abcam; and HRP-conjugated goat anti-mouse IgG; BIO-RAD) in TBS-T containing 1% (w/v) semi-skimmed milk powder. Blots were washed in TBS-T, immersed in ECL Plus reagent (BIO-RAD), and the bands were visualized on a BIO-RAD Gel-Doc Imager. Blots were quantified using ImageJ software.
Phosphorylation of regulatory light chain (RLC) in human donor samples was further analyzed using a S15 phospho-specific antibody for RLC (Affinity, #AF8618, 1:1,000 dilution in TBT-T containing 5% [w/v] BSA) and compared with total protein Western blots as described for the Phos-tag gels.
cMyBP-C was purified from human heart samples using the method published by Copeland et al. (2010) and run on 4–20% SDS-PAGE gradient gels. Gels were stained with Pro-Q Diamond phospho-protein stain and Coomassie total protein stain according to the manufacturer’s instructions (Life Technologies) and imaged on a BIO-RAD Gel-Doc Imager.
Statistical analysis
Parameters from the Hill fits to the individual stress–pCa curves were analyzed in SAS 9.1.3 (SAS Institute) using linear-mixed-effects models by incorporating two main effects (heart failure condition and sarcomere length) and their interaction. This approach links data from the same hearts to optimize statistical power, with each heart being treated as a random variable in the mixed-models analysis (Haynes et al., 2014). Compound symmetry was assumed for the covariance structure, and post-hoc analyses were performed using Tukey–Kramer corrections. Data quantifying hydroxyproline content and protein phosphorylation were analyzed using one-way ANOVA. Linear regression was used to illustrate the relationships between stress and hydroxyproline content (Fig. 4) and RLC phosphorylation of donor samples from two different methods (Fig. 6 C). P values <0.05 were considered significant.
Results
Length-dependent increase in Ca2+ sensitivity was eliminated with non-ischemic heart failure
Fig. 1 shows isometric stress measured at physiological temperature (37°C) plotted against activating [Ca2+], shown as pCa = −log10[Ca2+], for myocardial strips isolated from donor hearts (left panels), patients with ischemic heart failure (center panels), and patients with the non-ischemic disease (right panels). The top row of plots in Fig. 1 shows the absolute force values at short and long sarcomere lengths, with solid lines illustrating the best fit of Eq. 1 to each dataset. The bottom row of plots in Fig. 1 shows relative force values (Ca2+-activated force normalized to the maximum value within each experimental group). Parameter values from fits to the absolute force–pCa relationships for each myocardial strip are summarized in Fig. 2 (pCa50 values and Hill coefficients) and Fig. 3 (Fact and Fpas).
Force–pCa relationships at 1.9 and 2.3 µm sarcomere length for the three heart groups at physiological temperature (37°C) show that length-dependent activation of contraction was eliminated for non-ischemic heart failure. Top row: Absolute calcium-activated force values plotted against pCa, with force values normalized to the cross-sectional area for each myocardial strip. Bottom row: Relative force values for each strip were normalized to the average, maximal calcium-activated force within each experimental group. Lines represent fits to Eq. 1, with average pCa50 and nH values listed in lower panels for each experimental group. Summary data show mean ± SE from 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition).
Force–pCa relationships at 1.9 and 2.3 µm sarcomere length for the three heart groups at physiological temperature (37°C) show that length-dependent activation of contraction was eliminated for non-ischemic heart failure. Top row: Absolute calcium-activated force values plotted against pCa, with force values normalized to the cross-sectional area for each myocardial strip. Bottom row: Relative force values for each strip were normalized to the average, maximal calcium-activated force within each experimental group. Lines represent fits to Eq. 1, with average pCa50 and nH values listed in lower panels for each experimental group. Summary data show mean ± SE from 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition).
Calcium sensitivity of contraction increased as sarcomere length increased from 1.9 and 2.3 µm for organ donors and ischemic heart failure, but not for non-ischemic heart failure at physiological temperature (37°C). (A and B) pCa50 values (A) and nH values (B) from force–pCa fits to Eq. 1. Data are shown for 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition). Super plots show the distribution of data for each myocardial strip (smaller symbols), as well as the mean (larger symbols) for each donor or patient within an experimental condition for each group (Lord et al., 2020).
Calcium sensitivity of contraction increased as sarcomere length increased from 1.9 and 2.3 µm for organ donors and ischemic heart failure, but not for non-ischemic heart failure at physiological temperature (37°C). (A and B) pCa50 values (A) and nH values (B) from force–pCa fits to Eq. 1. Data are shown for 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition). Super plots show the distribution of data for each myocardial strip (smaller symbols), as well as the mean (larger symbols) for each donor or patient within an experimental condition for each group (Lord et al., 2020).
Maximal calcium-activated force and passive force increased as sarcomere length increased from 1.9 and 2.3 µm for all groups at physiological temperature (37°C). (A and B) Parameter values showing (A) maximal calcium-activated force and (B) relaxed, or passive, force from the force–pCa fits to Eq. 1. Data are shown for 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition). Super plots show the distribution of data for each myocardial strip (smaller symbols), as well as the mean (larger symbols) for each donor or patient within an experimental condition for each group (Lord et al., 2020).
Maximal calcium-activated force and passive force increased as sarcomere length increased from 1.9 and 2.3 µm for all groups at physiological temperature (37°C). (A and B) Parameter values showing (A) maximal calcium-activated force and (B) relaxed, or passive, force from the force–pCa fits to Eq. 1. Data are shown for 156 total strips from 18 total hearts (6 hearts from each group, with 3–5 technical replicates per heart at each condition). Super plots show the distribution of data for each myocardial strip (smaller symbols), as well as the mean (larger symbols) for each donor or patient within an experimental condition for each group (Lord et al., 2020).
pCa50 of the force–pCa relationship represents the Ca2+ concentration required to produce half-maximal force and can be used to quantify Ca2+ sensitivity of contraction in myocardial preparations. Ca2+ sensitivity increased with sarcomere length in myocardial strips from organ donors and patients with ischemic heart failure (i.e., pCa50 values increased; Fig. 2 A). Specifically, pCa50 increased by 0.07 pCa units at the longer sarcomere length for donors (P < 0.001) and for patients with ischemic heart failure (P = 0.003). In contrast, pCa50 did not change with sarcomere length in myocardial strips from patients with non-ischemic heart failure (ΔpCa50 = 0.02, P = 0.156). Hill coefficients exhibited similar group-level effects (Fig. 2 B), with nH decreasing in strips from organ donors and patients with ischemic heart failure as sarcomere length increased. The value of nH did not change with sarcomere length in strips from patients with non-ischemic disease. These data show that, in humans, length-dependent increases in the Ca2+-activated force response depend on the etiology of the heart failure.
Ca2+-activated force is reduced with heart failure
As sarcomere length increased from 1.9 to 2.3 µm, myocardial strips from all three heart conditions produced greater maximal force at the longer sarcomere length (P < 0.001 for the main effect of length; Fig. 3 A). Under relaxed conditions, passive force values increased with sarcomere length in all three groups as well (P < 0.001 for a main effect of length; Fig. 3 B). These increases in passive force likely stem from titin and collagen being stretched as sarcomere length increases (Granzier and Irving, 1995). Hydroxyproline content, a measure of myocardial collagen, was elevated in patients with non-ischemic heart failure (P = 0.001; Fig. 4 A). Passive force and hydroxyproline content were not correlated (Fig. 4 B), but maximal Ca2+-activated force decreased as hydroxyproline content increased (Fig. 4 C). This latter correlation may imply that greater collagen content reduces the density of contractile myocardium per tissue mass, which could underlie the subtle decrease in maximal force with ischemic heart failure and the ∼25–30% decrease in maximal force with non-ischemic heart failure, compared with donors (top row of Fig. 1).
Cardiac fibrosis increased with non-ischemic heart failure, which was inversely correlated with maximal calcium-activated force. (A) Hydroxyproline content, which is a relative metric of collagen content and cardiac fibrosis, is shown for each of the six hearts from donors and heart failure patients (three technical replicates were performed on a single sample from each heart). Black bars represent the mean for each heart sample group. (B and C) Average passive force values (B) and average Ca2+-activated force values (C) were plotted against hydroxyproline content for each heart. Force data in B and C represent the identical data shown in Fig. 3 for 2.3 µm sarcomere length (mean ± SE) for each heart, from the fits to Eq. 1. Correlations between the average force data and hydroxy proline content are shown by the solid lines within B and C, the dashed lines show 95% confidence interval of the regression, and the dotted line shows 95% confidence intervals of the data point.
Cardiac fibrosis increased with non-ischemic heart failure, which was inversely correlated with maximal calcium-activated force. (A) Hydroxyproline content, which is a relative metric of collagen content and cardiac fibrosis, is shown for each of the six hearts from donors and heart failure patients (three technical replicates were performed on a single sample from each heart). Black bars represent the mean for each heart sample group. (B and C) Average passive force values (B) and average Ca2+-activated force values (C) were plotted against hydroxyproline content for each heart. Force data in B and C represent the identical data shown in Fig. 3 for 2.3 µm sarcomere length (mean ± SE) for each heart, from the fits to Eq. 1. Correlations between the average force data and hydroxy proline content are shown by the solid lines within B and C, the dashed lines show 95% confidence interval of the regression, and the dotted line shows 95% confidence intervals of the data point.
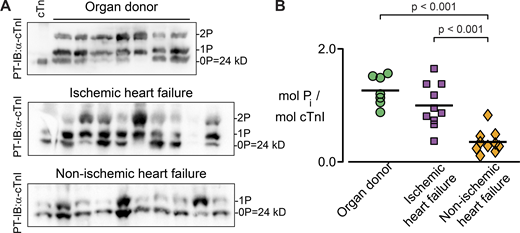
cTnI phosphorylation decreased with heart failure
Western blots of Phos-tag gels show cTnI phosphorylation levels for each group (Fig. 5 A). cTnI was phosphorylated to about 1.25 mol Pi/mol cTnI in the donor group, decreased to ∼1.0 mol Pi/mol cTnI for patients with ischemic heart failure, and to ∼0.3 mol Pi/mol cTnI for patients with non-ischemic heart failure (Fig. 5 B). cTnI phosphorylation levels were not different between donors and patients with ischemic heart failure, but cTnI phosphorylation was significantly reduced in patients with non-ischemic heart failure. The relative ratio of cTnI phosphorylation species (i.e., 0P, 1P, and 2P) was also different between heart groups, where most notably the non-ischemic samples showed no bisphosphorylated cTnI.
Heart failure type-specific decrease in cTnI phosphorylation. (A) Phosphorylation levels in left ventricular samples were determined by Phos-tag SDS-PAGE, followed by a Western blot against cTnI (top). Unphosphorylated cTnI (indicated by 0P, where human cTnI weighs 24,008 D) migrates faster than single (1P) or doubly (2P) phosphorylated cTnI. Unphosphorylated recombinant troponin (cTn, left lane) was used as a negative control, and each lane represents a single cardiac sample from a donor or patient. (B) Densitometry analysis for each lane in A was used to calculate the level of cTnI phosphorylation. A single measurement was made for each of the 7 donor hearts and 10 hearts from each heart failure group, indicated in Tables 1 and 2. Source data are available for this figure: SourceData F5.
Heart failure type-specific decrease in cTnI phosphorylation. (A) Phosphorylation levels in left ventricular samples were determined by Phos-tag SDS-PAGE, followed by a Western blot against cTnI (top). Unphosphorylated cTnI (indicated by 0P, where human cTnI weighs 24,008 D) migrates faster than single (1P) or doubly (2P) phosphorylated cTnI. Unphosphorylated recombinant troponin (cTn, left lane) was used as a negative control, and each lane represents a single cardiac sample from a donor or patient. (B) Densitometry analysis for each lane in A was used to calculate the level of cTnI phosphorylation. A single measurement was made for each of the 7 donor hearts and 10 hearts from each heart failure group, indicated in Tables 1 and 2. Source data are available for this figure: SourceData F5.
Myosin RLC phosphorylation was not different among the organ donors or patients with heart failure
Western blots of Phos-tag gels show that RLC phosphorylation levels were similar for each group (Fig. 6 A), with RLC phosphorylated to ∼0.2–0.25 mol Pi/mol RLC. This phosphorylation level is similar to the RLC phosphorylation level for organ donors and some patients with dilated cardiomyopathy in a recent study using cardiac tissue samples from Dr. Campbell’s laboratory (Vikhorev et al., 2022). RLC phosphorylation was also analyzed in donor samples using a phospho-specific antibody for Ser15 and an antibody for total RLC (Fig. 6 C), showing good agreement between RLC phosphorylation levels determined via Phos-tag blots.
Myosin RLC phosphorylation did not differ between the three groups. (A) Phosphorylation levels in left ventricular samples were determined by Phos-tag SDS-PAGE, followed by a Western blot against RLC. Unphosphorylated RLC (indicated by 0P, where human ventricular RLC weighs 18,789 D) migrates faster than phosphorylated RLC (1P). Unphosphorylated recombinant troponin (RLC [−], first lane for heart failure groups) was used as a negative control, and each lane represents a single cardiac sample from a donor or patient. (B) Densitometry analysis for each lane in A was used to calculate the level of RLC phosphorylation. (C) Relative RLC phosphorylation for each donor was assessed using phospho-specific antibody staining (top) versus total protein antibody staining (middle). Linear regression showed good agreement between densitometry calculations for RLC phosph-specific antibody staining versus Phos-tag analysis for donor samples. Molecular weights (kD) are listed to the right of the Ponceau stain (bottom). A single measurement was made for each of the 7 donor hearts and 10 hearts from each heart failure group, indicated in Tables 1 and 2. Source data are available for this figure: SourceData F6.
Myosin RLC phosphorylation did not differ between the three groups. (A) Phosphorylation levels in left ventricular samples were determined by Phos-tag SDS-PAGE, followed by a Western blot against RLC. Unphosphorylated RLC (indicated by 0P, where human ventricular RLC weighs 18,789 D) migrates faster than phosphorylated RLC (1P). Unphosphorylated recombinant troponin (RLC [−], first lane for heart failure groups) was used as a negative control, and each lane represents a single cardiac sample from a donor or patient. (B) Densitometry analysis for each lane in A was used to calculate the level of RLC phosphorylation. (C) Relative RLC phosphorylation for each donor was assessed using phospho-specific antibody staining (top) versus total protein antibody staining (middle). Linear regression showed good agreement between densitometry calculations for RLC phosph-specific antibody staining versus Phos-tag analysis for donor samples. Molecular weights (kD) are listed to the right of the Ponceau stain (bottom). A single measurement was made for each of the 7 donor hearts and 10 hearts from each heart failure group, indicated in Tables 1 and 2. Source data are available for this figure: SourceData F6.
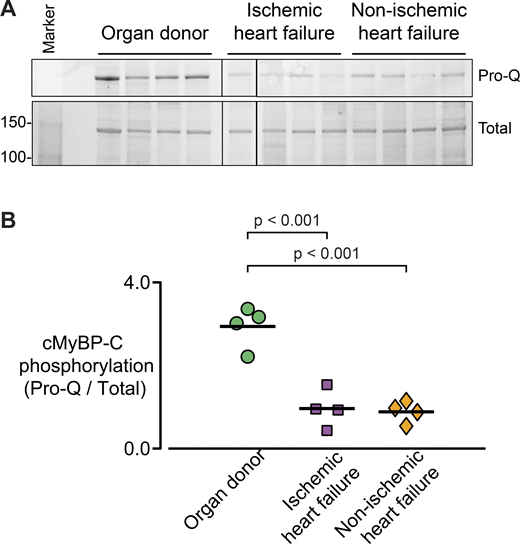
Cardiac myosin binding protein-C (cMyBP-C) phosphorylation decreased with heart failure
In a subset of samples (four for each heart group; Tables 1 and 2), cMyBP-C was extracted from samples and cMyBP-C phosphorylation was quantified using Pro-Q diamond phospho-stain (Fig. 7). cMyBP-C phosphorylation was approximately three times higher for organ donors than either of the heart failure groups.
Decreased cMyBP-C phosphorylation with heart failure. (A) cMyBP-C was extracted from four heart samples from each group (Tables 1 and 2) and phosphorylation levels were determined from Pro-Q diamond phospho-stain versus total protein stain. (B) Densitometry analysis for each lane in A was used to calculate the level of cMyBP-C phosphorylation. A single measurement was made for each of the four hearts from each group, indicated in Tables 1 and 2. Molecular weights (kD) are listed to the left of A. Source data are available for this figure: SourceData F7.
Decreased cMyBP-C phosphorylation with heart failure. (A) cMyBP-C was extracted from four heart samples from each group (Tables 1 and 2) and phosphorylation levels were determined from Pro-Q diamond phospho-stain versus total protein stain. (B) Densitometry analysis for each lane in A was used to calculate the level of cMyBP-C phosphorylation. A single measurement was made for each of the four hearts from each group, indicated in Tables 1 and 2. Molecular weights (kD) are listed to the left of A. Source data are available for this figure: SourceData F7.
Discussion
In this study we investigate length-dependent activation in permeabilized human myocardium at physiological temperature (37°C). There are two effects of sarcomere length on myocardial function at the cellular level: increased sensitivity to the activating Ca2+ concentration and greater maximal force production. Myocardial strips from organ donors showed a robust length-dependent increase in Ca2+ sensitivity (Figs. 1 and 2). This increase in Ca2+ sensitivity was largely preserved in the myocardium from patients with ischemic heart failure, but Ca2+ sensitivity did not differ with sarcomere length in samples from patients with non-ischemic heart failure. Maximal Ca2+-activated force increased with sarcomere length for all three heart groups, indicating that a portion of the mechanism(s) underlying length-dependent activation was preserved among all groups. We conclude that tissues from non-ischemic failing hearts produce a smaller cellular-level Frank–Starling response than the myocardium from organ donors or patients with ischemic disease.
Ca2+ sensitivity of contraction
Subtle shifts in Ca2+ sensitivity can have a major impact on in vivo function because the intracellular Ca2+ concentration does not saturate during a heartbeat; for example, consider the length-dependent ΔpCa50 of 0.07 shown for the tissue from organ donors (Fig. 1 D). This small pCa50 shift may seem unimportant, yet the force–pCa curves are so steep that the 0.07 increase in pCa50 can double the contractile force. pCa50 increased with sarcomere length in the myocardium from organ donors and patients with ischemic heart disease but did not change with length in samples from patients with non-ischemic heart failure. pCa50 was not systematically affected by heart failure status (P = 0.472 for heart failure status; Fig. 2 A), though there was a significant main effect of sarcomere length (P < 0.001). Comparing the range of pCa50 values among all conditions, we found three interesting results: (1) average pCa50 occurred near 5.39 for the donor and ischemic groups at 1.9 µm sarcomere length, (2) average pCa50 was greater at 1.9 µm sarcomere length in the non-ischemic group than the other two groups, and (3) pCa50 was ∼5.46–5.48 at 2.3 µm sarcomere length for all groups. This suggests an inability of the myocardium to properly relax or “turn off” at short sarcomere length for patients with non-ischemic heart failure, which may contribute to the elimination of length-dependent activation in this group. This could also impair relaxation function during diastole for these patients.
Several prior studies have shown that Ca2+ sensitivity of contraction increases in human heart failure without, in most cases, differentiating between ischemic and non-ischemic failure (van Dijk et al., 2012; Sequeira et al., 2013; Schwinger et al., 1994; Hamdani et al., 2010; Van der Velden et al., 2003a; Wolff et al., 1996). Increased Ca2+ sensitivity has been linked with lower cTnI phosphorylation levels in heart failure (van Dijk et al., 2012; Hamdani et al., 2010; Sequeira et al., 2013; Van der Velden et al., 2003b). This interpretation is supported by in vitro recombinant cTnI protein exchange experiments that emulate dephosphorylation (Ser23/24 substitutions for Ala) or phosphorylation (Ser23/24 substitutions to Asp; Wijnker et al., 2014b; Wijnker et al., 2014a; Hanft et al., 2013), and by studies with transgenic mice (Arteaga et al., 2000; Konhilas et al., 2003). The loss of, or the inability to, phosphorylate PKA target sites at Ser23/24 also reduced length-dependent increases in Ca2+ sensitivity, yet length-dependent increases in Ca2+ sensitivity were restored with PKA treatment or exchange of cTnI with phosphomimetic mutation of Ser23/24.
Phosphorylation of cMyBP-C has also been reported to preserve or amplify length-dependent increases in Ca2+ sensitivity of contraction and myocardial power output (Kumar et al., 2015; Hanft et al., 2020; Cazorla et al., 2006; Mamidi et al., 2016). cMyBP-C and cTnI are both PKA targets in the β-adrenergic signaling cascade that amplifies cardiac contractility, and Kumar et al. (2015) estimated that the PKA-mediated contribution to length-dependent activation was approximately twofold greater for cMyBP-C than cTnI (67 vs. 33%, respectively). A portion of this also appears to be a length-dependent response where cMyBP-C dephosphorylation augments pCa50 at shorter sarcomere lengths, and stretching the myocardium produces a nominal increase in Ca2+ sensitivity of contraction when cMyBP-C is dephosphorylated (Cazorla et al., 2000; Kumar et al., 2015; Mamidi et al., 2016). Therefore, the loss of cMyBP-C phosphorylation could diminish length-dependent activation with heart failure, similar to decreases in cMyBP-C phosphorylation in hypertrophic and dilated cardiomyopathies (Copeland et al., 2010; Vikhorev et al., 2022).
However, our cMyBP-C phosphorylation data show an inconsistency with these prior reports, where cMyBP-C phosphorylation is reduced in both heart failure groups (Fig. 7) even though length-dependent increases in Ca2+ sensitivity of contraction are only eliminated for the non-ischemic group. Due to technical limitations, cMyBP-C phosphorylation was analyzed in four cardiac tissue samples from each group. We recognize this is a limitation of our study. The ischemic samples we used for cMyBP-C biochemistry experiments came from the same hearts that were used for mechanics experiments, although both measurements only coincided for one heart in each of the other two groups (Tables 1 and 2). It is possible that this different sample population could explain the divergence we see between cMyBP-C phosphorylation and length-dependent activation between the two heart failure groups compared with prior studies. It is also possible that cMyBP-C phosphorylation has a smaller effect on length-dependent activation in the myocardial strips we used from the patients with ischemic versus non-ischemic heart failure.
Our data agree with prior length-dependent activation studies where dephosphorylation of either or both cTnI or cMyBP-C compromises length-dependent activation. Also, the largest effects of dephosphorylation appear to influence shorter sarcomere lengths from which stretching the myocardium shows a blunted length-dependent increase in pCa50. This phenomenon remains consistent with our mechanical and biochemical data from patients with non-ischemic heart failure, in that this population showed a higher pCa50 at 1.9 µm sarcomere length, while also showing the lowest cTnI and cMyBP-C phosphorylation levels. Together, this suggests that phosphorylation at cTnI Ser23/24 and cMyBP-C is important for maintaining the length-dependent activation in humans (Wijnker et al., 2014b; Wijnker et al., 2014a; Kumar et al., 2015; Cazorla et al., 2006; Mamidi et al., 2016). Throughout a heartbeat, sarcomere length typically starts at longer lengths and becomes shorter throughout systole, upon which the myocardium relaxes at the shortest sarcomere lengths. Our data would suggest that the myocardium from patients with non-ischemic heart failure may not be capable of properly relaxing at short sarcomere lengths (Fig. 2 A). It is also possible this inability to relax could prolong systole, both of which could underlie diastolic dysfunction with the non-ischemic disease. In turn, given the phospho-sensitive mechanism of the length-dependent activation described just above, the systolic function could also be compromised if the Frank–Starling response was diminished. A portion of the low cTnI or cMyBP-C phosphorylation levels could be exacerbated due to patients with heart failure being prescribed β-blockers (Table 2), thereby suppressing β-adrenergic signaling for PKA activation in the heart.
Maximal force production
All three heart groups demonstrated greater maximal Ca2+-activated force at longer sarcomere lengths (Fig. 3 A). While there were slight decreases (5–10%) in maximal force for the ischemic group, non-ischemic strips produced the lowest maximal force values among all groups—generating roughly 25–30% less force than the myocardial strips from donors (Fig. 1 and Fig. 3 B). These differences in maximal force production were not driven by any sarcomere-length-dependent differences in the passive force among the three heart groups (Fig. 3 B and Fig. 4 B). However, these decreases in maximal force were negatively correlated with cardiac fibrosis from each heart (Fig. 4), with greater fibrosis in the non-ischemic samples than in either the donor or ischemic samples. Thus, heart failure might influence the underlying mechanisms that facilitate increased Ca2+ sensitivity of contraction, but not the maximal force-generating capacity of the myocardium.
Maximal force production could be affected by the relative amount of contractile proteins per tissue volume, and increased fibrosis would compromise the density of contractile myocardium versus fibrotic infiltration and extracellular matrix. Although the maximal force values presented herein are lower than what we typically report using comparable permeabilized myocardial preparations from rodent hearts, we do not think our primary findings and data interpretations are changed due to tension values being <25 kN m−2. We recently tested the possible impact of freezing human tissue samples by comparing the mechanical characteristics of fresh versus frozen myocardial preparations (Milburn et al., 2022). In that study, we found no differences in force production or the Hill coefficient of force–pCa relationships, but there was a small, yet statistically significant increase in pCa50 for the frozen versus fresh samples. The range of maximal forces we measured (8–35 kN m−2) covers the same range of tension values from comparable studies using multicellular myocardial tissue preparations from human hearts (Haynes et al., 2014; Milburn et al., 2022; Awinda et al., 2020; Donaldson et al., 2012). These multicellular preparations typically show lower maximal force values than permeabilized mechanics measurements from human cardiomyocytes (30–45 kN m−2), though heart failure leads to lower force production by single cardiomyocytes (8–10 kN m−2; McDonald et al., 2020; Nijenkamp et al., 2020; Sequeira et al., 2013; Wolff et al., 1996). Together, our biochemical and mechanics data support the idea that maximal force would decrease as fibrosis increases with heart failure (Figs. 3 and 4).
We previously observed increased fibrosis and collagen content in samples from heart failure patients, although we had not differentiated between ischemic and non-ischemic groups (Haynes et al., 2014). This prior study did not find any increases in passive force, consistent with our current data (Fig. 3 B). However, Haynes et al. (2014) measured an increased ratio of the N2BA to N2B titin isoforms with heart failure compared with donors; the human myocardium typically expresses a ∼1:1 N2BA:N2B isoform ratio. Given that N2BA is longer than N2B, more N2BA could reduce the titin contribution to intracellular passive tension (or stiffness), which could counteract the greater contribution to passive force due to increased fibrosis with heart failure. Although we did not specifically test the N2BA:N2B isoform ratio in our current study, it is possible that a similar mechanism could underlie the equivalent passive force values for the failing and donor samples used herein.
Alternative regulatory pathways
The underlying mechanism(s) driving length-dependent activation has been difficult to define for decades. One of the prevailing theories had been the lattice-spacing hypothesis, where increases in sarcomere length reduced the distance between the thick and thin filaments, thereby increasing the probability of crossbridge binding (Fuchs and Martyn, 2005; Matsubara et al., 1984; Shiels and White, 2008; Yagi et al., 2004; Zhao and Kawai, 1993). Other studies have shown that proximity of the myosin heads to the thin filament (as indicated by the I1,1/I1,0 intensity ratio from small-angle x-ray diffraction studies) may be a better indicator of increased Ca2+ sensitivity of contraction than lattice spacing as the sarcomere length increases. Simultaneous x-ray diffraction and mechanics experiments also suggest that the orientation of myosin heads (as in well-ordered thick-filament head structure) perpendicular to the long axis of the thick filament is also well-correlated with length-dependent increases in maximal force (Farman et al., 2011; Ait-Mou et al., 2016). Increasing evidence suggests that mechanosensation along the thick filaments links the structural and mechanical aspects of contractile activation of both the thick- and thin-filament regulatory mechanisms (Reconditi et al., 2017; Caremani et al., 2021; Ma et al., 2021). Within this context, stretching a muscle imparts a mechanical signal along the thick filaments that transitions quiescent myosin heads from the OFF state (=super-relaxed state, or interacting heads motif) to the ON state (=disordered relaxed state), where they become available to bind actin and generate force (Zhang et al., 2017b; Brunello et al., 2020; Reconditi et al., 2017; Park-Holohan et al., 2021). There is ongoing research in this area to better understand the possibility of multiple structural versus biochemical states of the thick filament under relaxed and activated conditions. Altogether these studies imply that increases in sarcomere length could augment both passive and active tension values to amplify crossbridge recruitment from the OFF to ON state, which may contribute to length-dependent activation and the Frank–Starling mechanism.
We were unable to test the dual-filament mechanisms directly for the current study, as measuring the OFF–ON transition requires x-ray diffraction or fluorescent polarization techniques in concert with myocardial force measurements. Ongoing fluorescent polarization experiments in Dr. Kampourakis’ laboratory are focused on characterizing the OFF–ON equilibrium among these three heart groups. Findings from Zhang et al. (2017a) suggest that length-dependent increases in cardiac contractility involve complementary, yet distinct, thick-filament and thin-filament mechanisms. Increases in maximal Ca2+-activated force production may follow from changes in the thick-filament structure (i.e., mechanotransduction to further recruit crossbridges), while increases in calcium sensitivity of contraction at lower calcium levels may follow from changes in the thin-filament structure (i.e., Ca2+ activation of thin filaments to expose additional sites for myosin to bind). In combination, our mechanics, fibrosis, and cTnI phosphorylation data would support this idea. Therefore, thick-filament activation mechanisms may be preserved among all heart conditions that we tested, but cooperative activation of thin filaments may be compromised with ischemic heart failure and further eliminated with non-ischemic heart failure. Testing the impact of these dynamic regulatory mechanisms on human myocardial function is an exciting direction for future work.
Variation in human samples and study limitations
We studied hearts from patients and organ donors of different ages, sexes, and different medical histories (Tables 1 and 2). Our muscle mechanics measurements were acquired a couple of years before the biochemical measurements, which resulted in some of our tissue samples being unavailable for the biochemical experiments. Age and sex matching were balanced for the mechanics experiments, showing no significant differences between or within groups. Age and sex matching were less balanced for the biochemical studies, with a significant sex difference between groups (P = 0.02 from a one-way ANOVA) due to two females in the non-ischemic group being younger than the others. Thus, there could be some incongruities between the mechanics and the cTnI or cMyBP-C phosphorylation data, although we tried to measure a larger group of donors and patients for the biochemistry to describe each group more broadly. Within our ongoing studies, we are working to better match samples between multiple laboratories and use a greater number of donor or patient samples to maximize biological replicates rather than technical replicates within a heart. As a result, our data show more within-group variance among all mechanical and biochemical measurements from these human hearts than would be expected in well-controlled studies using animal models of human heart disease (Janssen et al., 2016; Libby, 2015). Although we observed some statistically significant effects, the genomic and phenotypic heterogeneity found in human samples is an inherent clinical variable that could limit statistical findings but, nonetheless, provide value when addressing precision health outcomes. We also recognize that myocardial samples from organ donors may not fully represent tissue from healthy human hearts. However, they are the best available option for this type of study and are considered the gold standard for most research analyzing human myocardial function.
Numerous studies using animal models of heart disease have shown that the relative phosphorylation of sarcomeric proteins can influence length-dependent activation, including cTnI, cMyBP-C, and RLC. A variety of changes in protein content and phosphorylation have been correlated with altered Ca2+ sensitivity of force production during human heart failure, being associated with both dilated and hypertrophic cardiomyopathies (van Dijk et al., 2012; Sequeira et al., 2013; Bodor et al., 1997; Zakhary et al., 1999; Van der Velden et al., 2003a; Bollen et al., 2017; Messer et al., 2007; Walker et al., 2011; Walker et al., 2013; Hamdani et al., 2010; Van Der Velden et al., 2003b; Copeland et al., 2010). Given that cTnI Ser23/24 phosphorylation and length-dependent activation may be tightly coupled (Wijnker et al., 2014b; Wijnker et al., 2014a; Hanft et al., 2013), our observation that cTnI phosphorylation decreased as length-dependent increases in pCa50 decreased represents one possible regulatory mechanism underlying the differences between non-ischemic heart failure and the other two heart groups (Figs. 1, 2, and 5). Characterizing additional regulatory pathways to better describe differentiating mechanisms between ischemic and non-ischemic myocardial function is an ongoing focus of our future work.
Most heart transplant patients demonstrate a dilated cardiomyopathy phenotype, and few patients undergo genetic testing to link their phenotype with any specific genetic mutation. This genotype-to-phenotype link is evolving with a growing number of research hospitals starting to expand genetic testing for patients with heart failure. Different tissue procurement strategies and timing between excising a sample until it is flash-frozen can also produce different measures of regulatory protein content and modulate phosphorylation levels, presumably due to endogenous phosphatase and kinase activities (Walker et al., 2011; Walker et al., 2013). Therefore, a range of structural and functional protein responses in human studies of heart failure present challenges that limit a definitive link between a single molecular cause of heart failure and patient phenotype. It is our hope that focused research efforts to overcome these challenges will ultimately benefit patient care and improve patient outcomes.
Conclusions
This study shows that the length-dependent increase in Ca2+ sensitivity is preserved in the myocardium from patients with ischemic heart failure but eliminated in the myocardium from patients with non-ischemic heart failure. Our data show that this contractile effect may reflect reduced bisphosphorylation of cTnI in the non-ischemic disease. A portion of the compromised length-dependent increases in Ca2+ sensitivity of contraction may also stem from reduced cMyBP-C phosphorylation in the heart failure groups compared with the organ donors. It may be important to adjust sarcomere-targeting therapies based on the etiology of heart failure.
Acknowledgments
Henk L. Granzier served as editor.
The authors appreciate helpful comments and advice from Brandon Biesiadecki, Kerry McDonald, and Ivanka Sevrieva.
This work was supported by grants from the American Heart Association (19TPA34860008 to B.C.W. Tanner and K.S. Campbell, and GRNT25460003 to K.S. Campbell), the National Science Foundation (1656450 to B.C.W. Tanner), and the National Institutes of Health (HL149164 to B.C.W. Tanner and K.S. Campbell, and TR001988 to K.S. Campbell).
The authors declare no competing financial interests.
Author contributions: B.C.W. Tanner and K.S. Campbell conceptualized the study. C.A. Blair, M.S. Thompson, and K.S. Campbell procured and distributed the tissue samples. B.C.W. Tanner, P.O. Awinda, K.B. Agonias, S. Attili, and T. Kampourakis performed the experiments. B.C.W. Tanner, P.O. Awinda, L.A. Walker, T. Kampourakis, and K.S. Campbell analyzed and curated the data. B.C.W. Tanner wrote the original draft of the manuscript. B.C.W. Tanner, P.O. Awinda, L.A. Walker, T. Kampourakis, and K.S. Campbell reviewed, edited, and revised additional versions of the manuscript, and all authors approved the final version submitted for review.
References
This work is part of a special issue on Myofilament Function 2022.






![Myosin RLC phosphorylation did not differ between the three groups. (A) Phosphorylation levels in left ventricular samples were determined by Phos-tag SDS-PAGE, followed by a Western blot against RLC. Unphosphorylated RLC (indicated by 0P, where human ventricular RLC weighs 18,789 D) migrates faster than phosphorylated RLC (1P). Unphosphorylated recombinant troponin (RLC [−], first lane for heart failure groups) was used as a negative control, and each lane represents a single cardiac sample from a donor or patient. (B) Densitometry analysis for each lane in A was used to calculate the level of RLC phosphorylation. (C) Relative RLC phosphorylation for each donor was assessed using phospho-specific antibody staining (top) versus total protein antibody staining (middle). Linear regression showed good agreement between densitometry calculations for RLC phosph-specific antibody staining versus Phos-tag analysis for donor samples. Molecular weights (kD) are listed to the right of the Ponceau stain (bottom). A single measurement was made for each of the 7 donor hearts and 10 hearts from each heart failure group, indicated in Tables 1 and 2. Source data are available for this figure: SourceData F6. Refer to the image caption for details.](https://cdn.rupress.org/rup/content_public/journal/jgp/155/3/10.1085_jgp.202213200/1/m_jgp_202213200_fig6.png?Expires=1779459036&Signature=T9pZAba95hM4TdbuTwZNCN8zhWhZB9pL7NmbteFF9MtbTlcwEqw3Ug~kTuTcp9mIubHrFP~dz5pe1ObGTebVSButxaxuBRGChe6AMW4b142Af4wNayZu4NChYCyBH1qCTTpPLo2L9hysHgAMAlxje~9HrKL3-W58wgFC7XBfGfyvZ6OPtuqSO6XIYql8z5pCpVz5x~Stb7ck5I5iVLKFJL1Nh~qdw3WMcDIdRwbg5wYhIdnjXduWC7IcaKG8thinsB1UV-t45GAud8zFanSf~IAuRA8X8ySXPw9-nKAN3PxLvmQUi18NO-PZyxzTikGtiD2sO7GJkQB7wdPBYIn6fQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)