CD4 tissue-resident memory T cells (TRM) are crucial adaptive immune components involved in preventing influenza A virus (IAV) infection. Despite their importance, their physiological role in the upper respiratory tract, the first site of contact with IAV, remains unclear. Here, we find that, after IAV infection, antigen-specific CD4 TRM persist in the nasal tissue (NT) compartment after infection and provide protection upon heterosubtypic challenge. Single-cell RNA-sequencing analysis reveals that NT CD4 TRM are heterogeneous and transcriptionally distinct as compared with their lung counterparts. Mechanistically, we demonstrate that the CXCR6–CXCL16 axis promotes CD4 TRM residency in the NT. Furthermore, we show that the NT of mice and humans contains a high frequency of Th17 CD4 TRM that aid in local viral clearance and in reducing tissue damage. Collectively, our results support a robust physiological role for NT CD4 TRM in local protection during heterosubtypic IAV infection.
Introduction
Influenza A virus (IAV) continues to pose a threat to society by causing yearly morbidity and deaths and providing a fertile ground for opportunistic bacterial infections (Paget et al., 2019). Seasonal vaccines elicit antibodies against the immunodominant surface glycoprotein hemagglutinin (HA), which is, however, prone to antigenic drift. Therefore, vaccines show limited effectiveness and are updated annually (Campbell et al., 2020). In addition, there is a hovering risk of pandemics caused by the recombination of animal and human viruses (The Lancet, 2024). Under such circumstances, antibody-mediated protection is usually inefficient. However, memory T cells recognize IAV internal proteins, which are relatively conserved across different strains (Vijayanand et al., 2020), thus mediating heterosubtypic responses.
Among the memory T cells, tissue-resident memory T cells (TRM) are gaining traction as a “ready to deploy” memory arm that is formed at the site of infection and hence acts as a barricade before the pathogens reach the circulation or lymphoid organs where effector memory or central memory T cells (TCM) can be activated (Marchesini Tovar et al., 2024). Indeed, during IAV infection, both CD4 and CD8 TRM in the lungs protect against heterologous viruses (Teijaro et al., 2011; Zheng et al., 2023). Compared to CD8 TRM, CD4 TRM are understudied, despite their multifunctionality. They can mediate viral clearance, act by direct cytotoxicity on virus-infected cells, guide the formation of IAV-specific lung CD8 TRM, and orchestrate local B cell responses (Borkner et al., 2021; Brown et al., 2012; Son et al., 2021; Swarnalekha et al., 2021; Teijaro et al., 2011). Specifically, lung CD4 TRM are diverse and exhibit different functions depending on their antigenic specificity. For example, two subsets of IAV nucleoprotein (NP)-specific lung CD4 TRM were recently identified: TRM1, which are Th1 like, and T follicular helper like cells, which are involved in promoting local antibody response and delivering help to local CD8 T cells (Son et al., 2021; Swarnalekha et al., 2021). On the other hand, M2e (ectodomain of virus matrix protein)–specific lung CD4 TRM were shown to be mostly of the Th17 subtype and protect from tissue injury during IAV infection (Omokanye et al., 2022). However, our knowledge about CD4 TRM in other tissues relevant for respiratory viral infection is still limited.
The upper respiratory tract (URT) is the port of entry for any inhaled pathogen and the first site of contact with the immune cells of the body. The immune system within the mouse URT is a complex network of draining cervical LNs (cLN), nasal-associated lymphoid tissues (NALT), and immune cells dispersed within the nasal chamber outside the NALT (referred to as nasal tissues; NT) (Pizzolla et al., 2017a; Randall, 2015). The NALT is the site for the recall expansion of memory T cells and priming of naïve T cells, whereas cLN act as a priming site (Liu et al., 2024; Pizzolla et al., 2017b). Increasing evidence suggest that protective CD4 TRM are generated during bacterial and SARS-CoV-2 infection in the URT (Borkner et al., 2021; Diallo et al., 2023; Kinnear et al., 2018; Lim et al., 2022; O’Hara et al., 2020; Wimmers et al., 2023; Xu et al., 2023; Yount et al., 2023). Nevertheless, the cues needed for URT CD4 TRM formation and tissue residency remain elusive. For IAV, some studies have demonstrated the protective role of URT CD8 TRM in reducing viral burden and transmission (Pizzolla et al., 2017a; Richard et al., 2020; Uddbäck et al., 2024). Surprisingly, despite the importance of CD4 TRM and the extensive spread of IAV, the role of IAV-specific CD4 TRM in the URT has not been explored.
Here, we investigate antigen-specific CD4 TRM within the URT of IAV-infected mice and healthy human volunteers. We demonstrate that CD4 TRM form in the NT of the URT and protect upon a heterologous IAV infection. Furthermore, we discover transcriptional differences between TRM populations in the NT versus lungs and define factors required for the formation of antigen-specific CD4 TRM in the NT. Our data provide a comprehensive characterization of previously unexplored, IAV-specific CD4 TRM that reside in the URT.
Results
IAV infection generates long-lived antigen specific CD4 TRM in the NT
To assess whether IAV-specific CD4 TRM arise in the URT, splenic CD4 T cells derived from OVA-specific OT-II TCR-transgenic mice that could be identified as CD45.1+ or tdTomato+ were transferred into congenic C57BL/6J (CD45.2+) mice. Subsequently, we infected the mice i.n. with a sublethal dose of mouse-adapted IAV strain, PR8 H1N1, that expresses the OVA323–339 peptide (PR8-OVA) and examined NALT and NT for OVA-specific CD4 TRM (OT-II CD4 TRM) on day 30 after infection. We used lungs as a positive control because IAV-specific CD4 TRM are well known to form there (Teijaro et al., 2011). Throughout all our experiments, we administered fluorochrome-conjugated CD4 antibody i.v. 5 min before sacrifice to distinguish CD4 T cells in the tissue from those in the vasculature (Fig. 1 A). CD4 memory T cells were identified as being CD3+, CD4+, CD44+, and CD62L−. We differentiated CD4 TRM from their circulating counterparts as CD3+CD4+CD44+CD62L− i.v.− CD69+ (Fig. S1 A). CD69 is a well-known tissue retention molecule that prevents egress of T cells from the tissue (Shiow et al., 2006; Szabo et al., 2019b). OT-II CD4 TRM were present in the NT at a similar frequency as in the lungs. However, we found less OT-II CD4 TRM in the NALT (Fig. 1, B and C).
To localize OT-II CD4 T cells anatomically within the URT, we isolated, fixed, and decalcified heads of PR8-OVA–infected mice on day 30 after infection. OT-II CD4 T cells were found throughout the nasal chamber, including the olfactory epithelium, respiratory epithelium lining the nasal septum (S), maxilloturbinates (MT), and NALT (Fig. 1 D and Fig. S1 B). Given the localization of OT-II CD4 T cells in proximity to the epithelium of the NT, we examined the expression of CD103, an integrin involved in the adhesion of T cells to E-cadherin on epithelial cells. In mice, CD103 is often associated with CD8 TRM (Beura et al., 2019; Szabo et al., 2019b; Zens et al., 2017), but its expression on CD4 TRM is poorly characterized. Still, single-cell RNA-sequencing (scRNA-seq) of lung CD4 TRM after Klebsiella pneumoniae vaccination showed low Itgae (the gene encoding for CD103) expression (Iwanaga et al., 2021). Indeed, CD103 was poorly expressed on lung CD4 TRM in comparison with NT CD4 TRM (5 versus 22%) (Fig. 1, E and F; and Fig. S1 C). Apart from CD103, we analyzed NT CD4 TRM for the expression of CD11a, another marker associated with TRM residency at late days after infection (Szabo et al., 2019b; Teijaro et al., 2011). Nearly all OT-II CD4 TRM in the NT and lungs expressed CD11a (Fig. 1, E and F; and Fig. S1 C).
Having demonstrated that NT CD4 TRM are established in a transfer model, we wanted to verify whether they would also form when precursor frequency is at physiological levels. Hence, we took advantage of MHC-II tetramers, presenting immunodominant peptides I-Ab HA91–107 and I-Ab NP306–322 (Besavilla et al., 2023; Miller et al., 2015). To follow the kinetics of antigen-specific CD4 TRM, we infected mice with PR8 and harvested the lungs and NT at different time points after infection (Fig. 1 G). We found that I-Ab HA91–107 tetramer+ and I-Ab NP306–322 tetramer+ CD4 TRM arose as early as 10 days post infection (dpi). The number of tetramer+ CD4 TRM declined from day 10 to day 30. Although there was a steady decrease in tetramer-specific CD4 TRM in the NT and lungs, we could still detect antigen-specific CD4 TRM in the NT on 120 dpi in most of the mice, suggesting this to be a relatively stable population (Fig. 1, H and I; and Fig. S1, D and F). We observed a more rapid decline of OT-II+ CD4 TRM in the OT-II transfer model (Fig. 1 I; and Fig. S1, D and E).
IAV-specific CD4 TRM in the lungs are self-sustaining without being replenished from the lymphoid reservoirs (Turner et al., 2014). To determine whether the same was true for NT CD4 TRM, we analyzed their expression of Ki-67, a marker for ongoing cell division (Miller et al., 2018). As expected, at the early stage after infection (10 dpi), ∼60% of the IAV-specific NT CD4 TRM expressed Ki-67 (Fig. 1 J and Fig. S1 G), suggesting that some of those may still be in the effector phase. Conversely, at early (30 dpi) and late (60 dpi) memory time points, only 10–15% of I-Ab HA91–107 tetramer+ and I-Ab NP306–322 tetramer+ NT CD4 TRM expressed Ki-67, indicating a low level of homeostatic proliferation (Fig. 1 J and Fig. S1 G). The importance of cognate antigen for the formation of TRM varies depending on tissue and T cell type; cognate recognition is a crucial factor for lung CD8 TRM formation (Takamura et al., 2016), but it is not required by NT CD8 TRM (Pizzolla et al., 2017a). Thus, we sought to assess the importance of cognate antigen maintenance for NT CD4 TRM formation. We generated effector tdTomato+ OT-II T cells in vitro and transferred them to C57BL/6 mice that were infected with either PR8 or PR8-OVA 2 days prior (Pizzolla et al., 2017a) (Fig. 1 K). In this model, even though effector OT-II cells can access the NT in both PR8 and PR8-OVA–infected groups, we found that OT-II CD4 TRM were only retained in the NT of the PR8-OVA–infected mice (Fig. 1, L and M). This was also observed for lung CD4 TRM (Fig. S1, H and I). Our results demonstrate that a cognate antigen, rather than only an inflammatory milieu, is required for the conversion of effector OT-II T cells to OT-II CD4 TRM in respiratory mucosal tissues, in contrast to NT CD8 TRM (Pizzolla et al., 2017a).
In summary, we found that IAV-specific CD4 TRM are generated and persist in the NT following infection.
NT CD4 TRM are multifunctional and produce cytokines upon antigen stimulation
A rapid and efficient response to viral reinfection is a key feature of memory T cells, including CD4 TRM. Upon recognition of their specific antigen, memory T cells are reactivated and produce cytokines, some of which have a direct impact on virus-infected cells, while others influence the recruitment of additional immune cells (Künzli and Masopust, 2023). To evaluate the IAV-specific response in NT CD4 TRM, we stimulated NT and lung cells, including CD4 TRM, from PR8-infected mice with an IAV-derived NP peptide pool. Compared with unstimulated controls or naïve NT CD4 TRM, a higher proportion of peptide-stimulated NT CD4 TRM produced IFN-γ and IL-2, while no change was observed for TNF (Fig. 2, A and B; and Fig. S2 A). Similarly, CD4 TRM sorted from either NT or lungs expressed higher amounts of intracellular IFN-γ when stimulated with splenocytes pulsed with the NP peptide pool compared with the SARS-CoV-2 nucleocapsid (SARS-CoV-2 N) peptide pool, ruling out a role for ongoing inflammation in IFN-γ production (Fig. 2, C and D). NT and lung CD4 TRM displayed a similar immunodominance profile with regard to different IAV-derived peptide stimulation (Fig. S2 B). In general, NT CD4 TRM harbored a lower frequency of cytokine-producing cells than lung CD4 TRM (Fig. 2, A–D; and Fig. S2 A). To exclude the possibility that this was due to the fact that NT CD4 TRM had a more exhausted phenotype, we measured PD-1 expression. CD4 TRM from both organs had a similar frequency of the PD-1+ CD4 TRM, indicating this was not the case (Fig. 2, E and F; and Fig. S2 C) but suggesting a role for PD-L1-PD1 axis in CD4 TRM regulation, similarly to what was described for IAV-specific CD8 TRM (Wang et al., 2019). Surprisingly, even though NT CD4 TRM had an overall lower frequency of cytokine-producing cells, ICOS, a T cell co-stimulatory molecule (Moguche et al., 2015; Peng et al., 2022), was expressed on a higher proportion of NT CD4 TRM in comparison with lung CD4 TRM (Fig. 2, E and F; and Fig. S2 C).
NT CD4 TRM provide protection upon heterologous IAV challenge
CD4 TRM promote viral clearance by recruiting and activating B cells, CD8 T cells, and innate cells and by exerting direct cytotoxic effects on virus-infected cells (Künzli and Masopust, 2023). Given that NT CD4 TRM displayed an IAV-specific response (Fig. 2, A–D and Fig. S2 A), we set out to elucidate their function upon heterosubtypic challenge. To test this, we infected mice with PR8 and challenged them with the heterologous X31 H3N2 virus on day 30 after primary infection. X31 has identical internal proteins to PR8 (including NP) but distinct surface glycoproteins (i.e., HA and neuraminidase). First, we verified whether CD4 TRM respond to and expand locally at 6 days after rechallenge (Fig. 3 A). To rule out any contribution of circulating lymphocytes, we treated the mice with FTY720 during the experiment (Brinkmann et al., 2002). Indeed, frequencies of I-Ab NP306–322 tetramer+, but not I-Ab HA91–107 tetramer+ CD4 TRM, sharply increased in both NT and lungs (Fig. 3, B and C; and Fig S3 A). The increase in I-Ab NP306–322 tetramer+ CD4 TRM was not due to expansion of naïve cells, as it was not observed upon primary X31 infection (Fig. S3 B).
To define a possible role in protection, we performed the same experiment as above but instead measured the viral titer in NT on day 3 after X31 infection. In addition to FTY720 treatment, we depleted either CD4 T cells, CD8 T cells, or both (Fig. 3 D). To effectively clear TRM from respiratory mucosal tissues, we injected mice with a high dose of depleting mAbs (Goplen et al., 2020; Son et al., 2021) (Fig. 3 E and Fig. S3, C and D). In the absence of any intervention, the secondary infection was most likely cleared from the NT by cross-reactive T cells (Fig. 3 F), as serum PR8 antibodies did not neutralize X31 (Fig. S3 E). Surprisingly, depletion of CD8 T cells did not affect the viral clearance in the NT (with or without FTY720 administration), bolstering the role of CD4 TRM in local protection. Conversely, CD4 T cell depletion severely impaired viral clearance, with viral titers similar to those of the mice that did not undergo a primary infection (Fig. 3 F). To further exclude any possible contribution of lung TRM, despite FTY720 administration, we used an intratracheal infection (i.t.) model for the primary IAV inoculation (Fig. 3 G). Upon i.t. infection, IAV-specific CD4 TRM formed only in the lungs but not in the NT (Fig. S3 F). Accordingly, heterologous i.n. X31 challenge, combined with FTY720, administration resulted in the expansion of I-Ab NP306–322 tetramer+ CD4 T cells only in lungs but not in NT (Fig. 3, H and I). When compared with local expansion after i.n. prime (Fig. 3 B), the results strongly suggest that TRM are the major players involved in the local response. In addition, we repeated the CD4 depletion experiment (Fig. 3 G) combined with i.n. or i.t. prime and X31 challenge (Fig. 3 J). As expected, CD4 depletion affected viral titers in lungs. Conversely, i.n. primed mice (both NT and lung CD4 TRM) had lower viral titers in NT when compared with i.t. primed mice (only lung TRM) and CD4 treatment impaired protection in NT only when local CD4 TRM were present (Fig. 3 J). Our overall results (Fig. 3, F–J) strongly support that NT CD4 TRM contribute to protection independently of lung CD4 TRM.
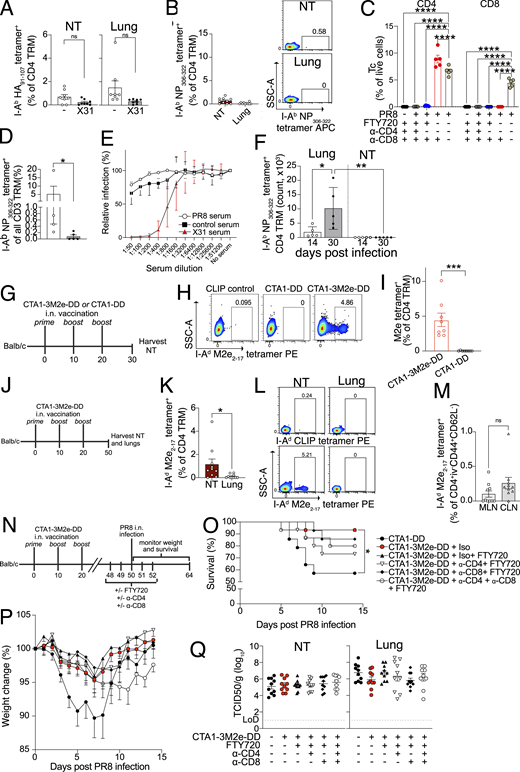
To verify whether NT CD4 TRM were also induced by vaccination, we established an i.n. vaccination model using CTA1-3M2e-DD as an immunogen. CTA1-3M2e-DD contains three tandem repeats of M2e linked to CTA1-DD adjuvant; M2e is highly conserved across IAVs, and immunization with CTA1-3M2e-DD has been shown to induce M2e-specific lung-resident CD4 T cells that protect against lethal IAV challenge in mice (Eliasson et al., 2008; Eliasson et al., 2018). For these experiments, we used BALB/c rather than C57BL/6 as CTA1-3M2e-DD induces poorer M2e-specific T cell responses in C57BL/6 (Eliasson et al., 2018). To maximize the formation of CD4 TRM in NT and not in the lung, we modified the immunization protocol and reduced the volume to 5 μl. We vaccinated mice with three i.n. doses of CTA1-3M2e-DD or CTA1-DD and evaluated the M2e-specific CD4 T cell (Tc) response on day 30 (day 10 after last vaccination) (Fig. S3 G). Like the response observed after infection, we found that I-Ad M2e2–17 tetramer+ CD4 Tc robustly formed in the NT (Fig. S3, H and I). Furthermore, I-Ad M2e2–17 tetramer+ CD4 TRM formed and persisted only in the NT and cLN on day 50 (day 30 after last vaccination) with almost none in the lungs and few in mediastinal LNs (MLN) (Fig. S3, J–M). To ascribe a role to M2e-specific CD4 TRM, we immunized mice with CTA1-DD or CTA1-3M2e-DD and challenged them at day 50 with a high dose of PR8 in low inoculum volume to restrict initial viral replication to the URT (Gailleton et al., 2025) (Fig. S3 N). This immunization strategy mainly generates M2e-specific CD4 TRM with no CD8 TRM (Eliasson et al., 2018). After challenge, 43% of the mice that were immunized with the control succumbed to the disease; however, immunization with CTA1-3M2e-DD protected against mortality and reduced weight loss (Fig. S3, O and P). Surprisingly, we could not detect differences in viral titers in either lungs or NT between the groups, suggesting that the immunization rely on other mechanisms for protection than viral control (Fig. S3 Q), as previously observed (Omokanye et al., 2022). Furthermore, while showing a trend, the depletion of CD4 TRM did not significantly decrease the survival of mice that were immunized with CTA1-3M2e-DD (Fig. S3, O and P), possibly due to the limited mortality of the reduced-volume infection model used herein.
Taken together, our findings revealed that NT CD4 TRM are important for protection during heterosubtypic infection.
CD4 TRM in the lungs and NT are heterogeneous, but Th17 cells are enriched in the NT
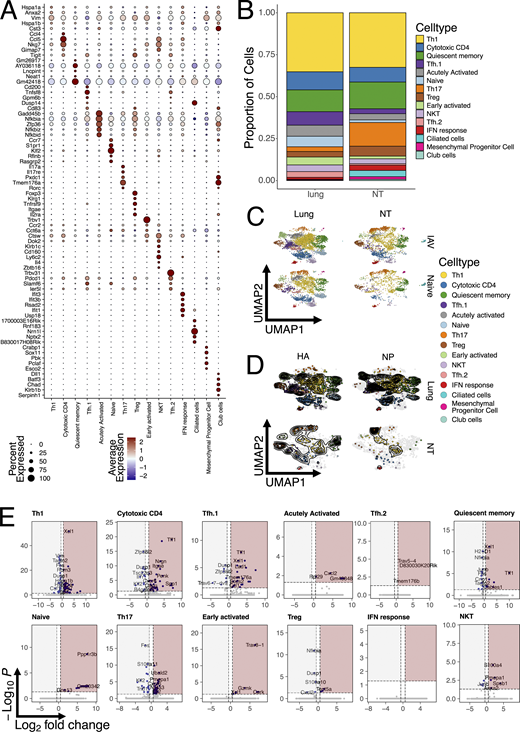
Having identified the dynamics and role of NT CD4 TRM, we wanted to investigate the transcriptional differences between total and antigen-specific CD4 TRM in NT versus lungs. Therefore, we performed scRNA-seq together with single-cell TCR sequencing (scTCR-seq) at 30 dpi. Mice were injected with CD4 antibody i.v. 5 min before sacrifice, and total CD4 TRM, I-Ab HA91–107 tetramer+, and I-Ab NP306–322 tetramer+ CD4 TRM were sorted from NT and lungs of mice (Fig. 4 A). Furthermore, we also sequenced a small proportion of all live cells, which were CD4−. To maximize the number of cells and samples that could be loaded simultaneously, different samples were labeled with hashtag antibodies. Data from all mice and organs were integrated, processed, and visualized on a two-dimensional UMAP (Fig. 4 B). T cell subpopulations were assigned based on well-known markers and gene signatures (Fig. 4, C and D; Fig. S4 A; and Table S1). The major T cell subpopulation was, as expected given the intracellular viral infection, Th1 cells (Fig. S4 B) defined by a well-established signature (Andreatta et al., 2022), including Ifng and Cxcr3. Cytotoxic CD4 T cells were present in both organs and defined by Nkg7, Fasl, Ccl4, Ccl5, and a CD4 cytotoxic signature (Hashimoto et al., 2019). T follicular helper (Tfh) cells were generally more prevalent within lungs (Fig. S4 B), consistent with a possible role in supporting ectopic germinal centers in iBALTs. Based on their transcriptional profile, they were separated into two clusters, named Tfh.1 and Tfh.2. Tfh.1 were Tfh cells with strong expression of Tnfsf8, Bcl6, and a signature previously associated with Tfh (Andreatta et al., 2022). Nevertheless, Tfh.2 had a higher expression of other Tfh-associated genes such as Tox, Slamf6, and Pdcd1. Quiescent memory cells were slightly more prevalent within NT and characterized by the expression of several long noncoding RNAs (including Malat1) (Dey et al., 2023; Plasek and Valadkhan, 2021) and low ribosomal gene expression. A small proportion of our CD4 TRM were naïve-like T cells expressing Ccr7, Sell, and Klf2 (Wang et al., 2022). Furthermore, we identified regulatory T cells (Tregs) by Foxp3 expression and Treg signature (Yang et al., 2021). Early activated cells were defined based on the expression of genes linked with migration within the tissue, such as Gpr183 (Li et al., 2016), and signaling receptors such as Ms4a4b (Yan et al., 2012), suggesting an overall movement within the tissue but also modulation of T cell activation. An “acutely activated” cluster expressed Cd40lg, several transcription factors and also had high expression of genes involved in regulation of NF-κB signaling (Nfkbia, Nfkbid, and Nfkbiz). This cluster has been previously reported and suggested to include cells that are poised to rapidly respond to secondary infections by producing cytokines (Kurd et al., 2020; Woodring et al., 2022). Natural killer T (NKT) cells and IFN-responsive cells were clearly identified by their distinctive signatures: the former expressed Klrd1, Klrk1, Klrb1c (Cohen et al., 2013; Hu et al., 2022), and other cytotoxic genes (Nkg7 and Fasl) (Ng et al., 2024; Tuomela et al., 2022), while the latter was enriched for IFN-stimulated genes (Isg15, Ifit3, and Ifit1), a signature similar to one previously identified in human CD4 (Szabo et al., 2019a). Strikingly, Th17 cells, expressing Rorgc, Il17a, Tmem176a, and Ccr6, were much more abundant within NT (Szabo et al., 2019a) (Fig. S4 B and Table S1). We were also able to identify several endothelial and epithelial airway cells clusters from the live cell fraction devoid of CD4 T cells.
Overall, our scRNA-seq analysis highlighted the diversity and variety of CD4 TRM subpopulations in both lungs and NT. While Th1 and cytotoxic CD4 T cells made up almost 50% of the total TRM in both lungs and NT, consistent with an intracellular viral infection, their relative proportions were similar between organs (Fig. S4 B). Interestingly, IAV infection did not appear to majorly reshape the CD4 TRM landscape in terms of the abundance of cell type (Fig. S4 C). Therefore, to confirm our results and limit our analysis to influenza-specific cells, we subsetted the CD4 cells to only include the tetramer+ cells. Indeed, this did not change the proportion of CD4 TRM, confirming an increased abundance of both HA+ and NP+ Th17 cells within the NT, while these cells were almost absent in lungs (Fig. 4 E and Fig. S4 D). By visualizing the distribution of HA+ and NP+ cells as density plots, we established that antigen-specific cells differentiated into almost all CD4 TRM subsets, except for Th17 in the lungs. However, the density distribution in NT was dramatically different between the two proteins, with NP+ cells being mostly Th17 and Th1, while HA+ cells also included many cytotoxic and Tfh CD4 TRM cells (Fig. 4 E and Fig. S4 D).
CD4 TRM within the NT exhibit a distinct transcriptional profile compared with lungs
The power of the single-cell approach is that it allows transcriptional differences to be compared between the same CD4 TRM subtypes, depending on the organ. Therefore, we performed differential gene expression (DEG) analysis for each cell subpopulation and visualized the results with volcano plots (Fig. S4 E and Table S1). As expected, the majority of CD4 TRM subpopulations exhibited similar gene expression between organs; however, Th1, cytotoxic CD4, Tfh.1, and Th17 clusters were the ones with the most organ-specific differences. For Th1, lung cells had higher expression of Ccr2, a chemokine receptor previously linked with pulmonary homing (Brownlie et al., 2022; Hoft et al., 2019), and Ctla4 but also Ifng and Nfkbia, suggesting a more active and productive phenotype compared with that of their NT counterpart, in line with what we observed above (Fig. 2, A and B). On the other hand, NT Th1 were enriched for other essential transcription factors such as Crem and Taf7, which are involved in cytokine production (Devaiah et al., 2010; Rauen et al., 2013), and Xcl1, which promotes Treg differentiation (Nguyen et al., 2008), suggesting an overall more immune-suppressive state. Cytotoxic CD4 cells had a similar pattern to Ccr2 and genes associated with a more active phenotype expressed in the lungs. Interestingly, NT CD4 TRM upregulated several chemokine receptors, including Cxcr6 and Ccr8, and many other factors previously associated with T cells at mucosal sites (such as Spp1 and Penk) (Kourepini et al., 2014; Mas-Orea et al., 2023). Remarkably, Cxcr6 was also significantly upregulated in NT Tfh.1 and Th17 clusters. In addition, Tfh.1 NT cells also shared Tff1 and Xcl1 upregulation with other clusters, while NT Th17 exhibited increased Crem and Nr4a2 expression.
In summary, several CD4 TRM subpopulations displayed DEG between lungs and NT, with the lung cells showing a more activated phenotype and higher Ccr2 expression, consistent with their localization. Conversely, NT CD4 TRM expressed genes commonly associated with T cells at mucosal sites and with an immune-dampening phenotype, with several clusters also expressing the chemokine receptor Cxcr6.
Expanded clonotypes have multiple fates and populate both lungs and NT
Having identified differences between CD4 TRM residing in different organs, we wanted to investigate whether T cell clones were compartmentalized or shared between them. A caveat of our experimental procedure is that we had to pool several mice to obtain sufficient numbers of antigen-specific cells, and, therefore, we were not able to define clonality at the single mouse level. However, a previous study suggested that CD4+ T cell responses are largely private even for a single viral-peptide specificity (Andreatta et al., 2022). Consistent with the notion of a largely private response, we did not detect any exaggeratedly expanded T cell clones, with only five families with >30 members. Expanded clones were distributed across the majority of CD4 TRM subpopulations within the lungs, except for the naïve, Treg, NKT, and Th17, which were composed mainly of single clones (Fig. 5 A). Conversely, NT exhibited a higher clonal expansion within Th17. When focusing only on antigen-specific clones, we detected a very similar pattern of clonal expansion (Fig. 5 B). To visualize the specificity, organ, and subpopulation distributions, we plotted the top 34 expanded clones (Fig. 5 C). This confirmed the pattern of expansion and that most of expanded clones were shared among several subpopulations but also between organs. Validating our tetramer sorting approach, clones were either HA- or NP-specific; however, in many cases, we did not sort all clones within the same family, as also expected. Substantiating the private nature of TCR responses was the fact that many of the NP expanded clones had similar, but not identical, CDR3 sequences (Fig. 5 C). This suggests that certain TCRs had higher affinity for the NP peptide–MHC complex, but stochastic variations, not altering binding, were present in different mice. Indeed, analysis of paired TCRa-TCRb usage demonstrated a strong bias in NP responses for TRAV6-7_TRBV3 for both lungs and NT (Fig. 5 D). By visualizing with a bubble plot, in which the size of the bubble is proportional to the clone size, we revealed that many of the more expanded, antigen-specific clones in the lungs were also shared with the NT (Fig. 5 E). This points toward a common origin within the same LNs and subsequent migration to tissues of residency. Finally, to determine whether certain clonotypes were more prone to direct CD4 TRM toward a certain fate, we utilized the “clonotype bias” measure introduced by Andreatta et al. (Andreatta et al., 2022) and implemented within the scRepertoire package (Borcherding et al., 2020). The analysis revealed that the majority of clonotypes were largely unbiased with few exceptions (Fig. 5 F). Interestingly, clonotype bias strongly correlated with being organ specific (Fig. 5, F and G). For instance, the third largest clone was present only in the lungs, and over 60% of its members belonged to the Tfh.2 cluster (Fig. 5, F and G). Likewise, the only large clone found exclusively in NT had just shy of 50% of its members belonging to the early activated CD4 TRM group (Fig. 5 G). However, the most expanded clones were generally not biased, despite having a strong prevalence of Th1 and Tfh.1 cells (Fig. 5 H). The tendency of the few biased clones to be organ specific could also suggest local proliferation and expansion.
Combined TCR analyses unveiled how most CD4 TRM clonotypes can differentiate into multiple fates and then reside in both lungs and NT. However, the few clones that were tissue specific tended to be biased toward one CD4 T cell subtype.
CXCR6–CXCL16 axis promotes CD4 TRM recruitment to the NT
Given that Cxcr6 was highly expressed in several NT clusters (Fig. S4 E), we further explored the mechanistic involvement of this chemokine receptor in the migration of effector CD4 T cells to the NT. First, we confirmed Cxcr6 to be the most interesting candidate, as it was more highly expressed in NT CD4 TRM than in lung CD4 TRM (Fig. 6, A–C). In particular, the clusters of cytotoxic CD4 T cells, Tfh.1, Th17, Treg, early activated, and Tfh.2 displayed higher expression of Cxcr6 in NT (Fig. 6 B). CXCR6 is known to be predominantly expressed in CD8 TRM rather than circulating CD8 T cells (Mabrouk et al., 2022). Similarly, we found high CXCR6 expression in CD4 TRM and low expression in CD4 effector memory T cells (TEM i.v.+) of IAV-infected mice (Fig. 6 D). While CXCR6 expression was greater in infected NT than lungs, its expression was similar to that of NT CD4 TRM in naïve mice (Fig. 6 D). This may seem surprising; however, by definition, TRM are not naïve cells, and therefore, their presence in NT has been triggered by a previous insult. Furthermore, we also found higher CXCR6 expression among antigen-specific OT-II TRM in NT versus lungs (Fig. 6, E and F).
Within the lungs, CXCR6 is essential for the positioning of CD8 TRM in the airways but not in the lung interstitium after IAV infection (Wein et al., 2019). To determine whether CXCR6 is required for the establishment of CD4 TRM in the NT, we generated WT-Cxcr6−/− bone marrow (BM) chimeras. To do this, we lethally irradiated congenic C57BL/6 (CD45.1+) (recipient) to deplete the host BM and reconstituted them with equal numbers of tdTomato+ WT BM (CD45.2+) and tdTomato−Cxcr6−/− BM (CD45.2+) (Fig. 6 G). Blood chimerism was verified on day 67 after BM transfer, with almost 90% of the leukocytes being donor derived. Interestingly, we detected a higher frequency of Cxcr6−/− leukocytes in the blood (Fig. S5 A). On day 70 after BM transfer, we infected the mice and analyzed organs at day 30 after infection. CXCR6 expression helped in lodging total and I-Ab NP306–322 tetramer+ CD4 TRM in both NT and lungs, as we observed a reduced frequency of Cxcr6−/− CD4 TRM in comparison with WT CD4 TRM in both compartments (Fig. 6, H–J). Surprisingly, we found that 6 out of 13 chimeric mice had too few I-Ab NP306–322 tetramer+ CD4 TRM in the NT for downstream analysis, from both WT and KO cells (Fig. 6 J). We speculate that this may be due to a generally low level of donor T cell chimerism achieved at the NT in comparison with that in the lungs (Fig. S5 B). The finding that NT CD4 TRM are not easily depleted or replenished, consolidate our data about the longevity of these cells, i.e., that once they are formed and reside in the NT, they will persist. Moreover, we detected an increased accumulation of Cxcr6−/− CD4 T cells in the blood, spleen, lung-draining MLN, and URT-draining cLN, confirming the hypothesis that Cxcr6−/− CD4 T cells have migratory defects and/or they are not retained in the tissue (Fig. 6 K).
CXCL16 is the only known ligand for CXCR6. It exists in both transmembrane and soluble forms and acts as an adhesion molecule and a chemoattractant for leukocytes (Koenen et al., 2017; Wilbanks et al., 2001). In murine lungs, CXCL16 colocalizes with Epcam, and its expression is restricted to the large airways lining the lungs (Wein et al., 2019). We investigated CXCL16 expression in NT by microscopy and demonstrated that CXCL16 is expressed in the NT of PR8-OVA–infected mice and some OT-II CD4 TRM localize near CXCL16 (Fig. 6 L and Fig. S5 C). Finally, to examine the contribution of CXCL16 to the early recruitment of effector CD4 T cells to the NT and lungs, we treated PR8-infected mice with α-CXCL16 antibody (Fig. 6 M). α-CXCL16 treatment significantly reduced the frequency and number of I-Ab NP306–322 tetramer+ CD4 TRM retained in the NT and, to a lesser extent, in the lungs (Fig. 6, N and O).
Altogether, we showed that CXCL16 production and CXCR6 expression on CD4 T cells are promoting the recruitment of CD4 effector memory T cells to respiratory mucosal tissues and the subsequent establishment of TRM.
Healthy humans harbor IAV-responsive CD4 TRM in the nasal mucosa
Given the importance of IAV-specific CD4 TRM in the NT of mice, we wanted to investigate whether a similar population exists in healthy human adults. As most individuals are exposed to IAV by the age of three (Bodewes et al., 2011), healthy adults must have undergone several symptomatic and asymptomatic infections, and we hypothesized that they should maintain IAV-specific CD4 TRM in their NT. The recruited subjects did not have any respiratory infections or symptoms in the 2 wk prior to the study to avoid contamination from recently activated CD4 T cells. We collected nasopharyngeal swabs (NT) and peripheral blood mononuclear cells (PBMCs) and analyzed T cells by flow cytometry (Thome et al., 2014) (Fig. 7 A and Fig. S5 D). While the frequency of naïve CD4 T cells (CD3+CD4+CD45RA+CCR7+) was higher in PBMCs, CD4 TCM (CD3+CD4+CD45RA−CCR7+) and CD4 terminally differentiated effector memory T cells re-expressing CD45RA did not show any difference (Fig. 7 B). Interestingly, CD4 TEM (CD3+CD4+CD45RA−CCR7−) were higher in NT with their subpopulation TRM (CD3+CD4+CD45RA−CCR7− CD69+) being exclusively present in NT of all subjects, albeit in varying numbers. When we analyzed both PBMCs and NT, we found that NT CD4 TRM included CD103 (18%) and CXCR6-expressing cells (23%) (Ramirez et al., 2024), while PBMC CD4 TEM barely expressed them, similar to our findings in mice (Fig. 7, C and D).
A longitudinal study on SARS-CoV-2 vaccines that had breakthrough infections showed functional NT T cells, specific for SARS-CoV-2 proteins, that persisted for ≥140 days (Lim et al., 2022). Given the relatively long lifespan of NT CD4 T cells, we wanted to examine whether healthy adults retained IAV-specific NT CD4 TRM, acquired by encountering IAV at some time point in life. To do that, we stimulated PBMC and NT cells with IAV peptide pools derived from the internal proteins NP and matrix protein 1 (M1) and assessed cytokine expression. We found that PBMC CD4 TEM from 79.5% of the subjects responded to stimulation by secretion of IFN-γ (Fig. 7 E). Conversely, we did not detect a significant change in IFN-γ response in NT CD4 TRM after stimulation. Furthermore, we found no detectable TNF response from NT or PBMC CD4 T cells (Fig. 7 E). As the scRNA-seq mouse experiment had revealed high frequencies of Th17 cells in NT (Fig. 4 E and Fig. S4 B), we also tested if IL-17a expression was induced in stimulated cells. Intriguingly, in NT CD4 TRM, the frequency of IL-17a–expressing cells increased after stimulation in 63.2% of the individuals. Little to no IL-17a expression was detected in PBMCs before or after stimulation (Fig. 7, E–G).
Overall, our data show that human NT CD4 TRM share important features with mouse CD4 TRM, such as the expression of CD103 and CXCR6. Moreover, NT of healthy individuals is populated by IAV-specific Th17 CD4 TRM.
NT Th17-CD4 TRM aid in viral clearance and dampen tissue damage
Given the abundance of Th17 CD4 TRM in the NT of mice and human, we wanted to further delineate their functional role. First, we confirmed that PR8-infected mice carried IAV-specific Th17 CD4 TRM in NT by stimulating lung and NT cells with a single immunodominant NP peptide (NP306–322) and assessing intracellular expression of IL-17a as a measure of the Th17 response. We used NP306–322, as this was the same peptide, loaded into tetramers, used to sort CD4 TRM for scRNA-seq. Consistent with all our data, we found that only NT, but not lung, CD4 TRM from PR8-infected mice expressed IL-17a. Surprisingly, IL-17a production was stimulation-independent although infection dependent (Fig. 8, A–C).
Th17 cells recruit neutrophils and B cells, promote the transport of secretory IgA to the mucosal sites, and protect the hosts in an antibody-independent manner during IAV infection (Eliasson et al., 2018; Jaffar et al., 2009; Laan et al., 1999; Wang et al., 2011). On the other hand, detrimental effect of Th17 was also reported as an i.n. immunization triggered Th17 population that caused increased morbidity in mice (Maroof et al., 2014). To examine whether NT Th17 CD4 TRM had a beneficial role, we utilized CD4CreRorcfl/fl (CD4-specific Th17 KO) and CD4CreRorcfl/wt (WT) mice (Gribonika et al., 2022) (Fig. S5 E). Mice deficient in the Rorc gene lack RORγt, a Th17 lineage-specific transcription factor that controls the activation of IL-17A and IL-17F genes, and do not differentiate to Th17 CD4 T cells (Ivanov et al., 2006). To verify the specific role of Th17 CD4 TRM, we infected CD4CreRorcfl/fl and CD4CreRorcfl/wt mice with PR8 and challenged them at day 30 after primary infection (Fig. 8 D). FTY720 treatment ruled out the contribution of circulating lymphocytes. CD4 Th17 KO mice had slightly higher viral titer in the NT than did WT mice on day 3 after secondary infection (Fig. 8 E). Furthermore, Th17 CD4 cells have been suggested to play a role in tissue repair by secreting anti-apoptotic cytokines that dampen tissue damage (Dutta et al., 2023; Konieczny et al., 2022). We performed a reinfection experiment in a similar way and enumerated apoptotic cells (TUNEL+) in the NT upon X31 challenge (Fig. 8 F). Indeed, CD4CreRorcfl/fl mice exhibited an increased number of apoptotic cells in the respiratory mucosa of the nasal septum in the NT (Fig 8, G and H; and Fig. S5 F). Of note the epithelium surrounding nasal septum is also a site of viral replication (Fig. 8 I) and of CD4 TRM residency (Fig. S1 B). However, depletion of IL-17 did not affect viral titer or apoptotic cell clearance in NT (Fig. S5, G and H), suggesting IL-17–independent mechanisms mediated by Th17 in the NT.
Altogether, we confirmed that Th17 CD4 TRM ameliorate IAV-related pathology by reducing viral replication and tissue damage.
Discussion
The current understanding of CD4 TRM during IAV infection relies on findings from lung CD4 TRM. Despite the URT being the first location where IAV encounters the immune system, little is known about CD4 TRM at this crucial immunological site. Here, we defined the phenotype and functional role of CD4 TRM residing in the NT region of the URT. We demonstrated that antigen-specific CD4 TRM vigorously respond to heterologous IAV challenges, promote direct viral control, and provide tissue protection in the URT.
Notably, we found that endogenous antigen-specific CD4 TRM are long-lived. The HA+ CD4 TRM in the NT decayed at a slower rate than CD4 TRM in the lungs, even though they were less abundant when quantified in absolute numbers. Furthermore, a small fraction of both NP+ and HA+ CD4 TRM expressed Ki-67 on day 60, suggesting that they may be maintained by homeostatic proliferation like lung CD4 and CD8 TRM (Takamura et al., 2016; Turner et al., 2014). We hypothesize that NALT (Bates, 2022) or tertiary lymphoid structures in the NT (Gailleton et al., 2025) could be the local sites for naïve CD4 T cell priming and expansion. Indeed, one recent study showed that i.n. vaccination established a CD4 T cell response within the interfollicular regions of the NALT, in turn promoting the homing of antibody-secreting plasma cells to the NT (Liu et al., 2024). Similarly, our recent work indicated that IAV infection and i.n. vaccination induce ectopic germinal center structures within the NT itself (Gailleton et al., 2025). Furthermore, it should be noted that, while expanded lung clonal families are also found within the NT, the opposite is not true, with all clonal families in the NT with more than eight members being organ specific (Fig. 5 E). However, we could not exclude the possibility of some degree of replenishment from local tertiary lymphoid structures or lymphoid organs via the blood. Overall, our data suggest that few cells traffic from lung-draining LNs to NT while the majority instead expand in situ.
Even though effector T cells traffic to the infection site due to inflammatory cues, their establishment as TRM may or may not require the presence of an antigen depending on the tissue. Antigen-dependent residency has been described for the lungs and brain (Khan et al., 2016; McMaster et al., 2018; Wakim et al., 2010; Zens et al., 2016), but not for genital tract or intestinal CD8 TRM, where the inflammatory milieu and chemokines are sufficient for their formation (Bergsbaken and Bevan, 2015; Sato et al., 2014). Even in lungs, antigen dependency can be bypassed by the introduction of adjuvants, such as zymosan, which evoke a specific inflammatory milieu (Caminschi et al., 2019). Here, we demonstrated that CD4 TRM in NT require the presence of a cognate antigen to be formed after IAV infection, which is the opposite of CD8 TRM (Pizzolla et al., 2017a). Furthermore, we showed that the same held true for lung CD4 TRM. The observation in the NT highlights an interesting discrepancy regarding the minimal cues needed by different T cell types to establish residency, even within the same tissue. Parabiosis is considered as the gold standard for establishing true tissue residency. While CD69 alone is not a definitive marker of durable tissue residency, the multiple lines of evidence presented in our study such as dependence on local antigen, viral control during rechallenge in the presence of FTY720, and viral control in NT by CD4 TRM independent of their lung counterparts (Fig. 3 J) support that NT CD4 cells identified herein are tissue resident and operate independently of the patrolling memory T cells.
Generally, CXCR6 is a chemokine receptor that is more highly expressed on CD8 T cells than on CD4 T cells (Di Pilato et al., 2021; Mabrouk et al., 2022) and is well described as an essential factor for CD8 TRM seeding to the lung airways but not the lung interstitium after IAV infection (Wein et al., 2019). Here, using BM chimeras and blocking experiments, we revealed that the CXCR6–CXCL16 axis is important for the formation of antigen-specific CD4 TRM in NT and lungs. Another study did not find a role for CXCR6 in the recruitment of antigen-specific CD4 T cells to the lungs of Mycobacterium tuberculosis–infected mice (Ashhurst et al., 2019); however, the establishment of TRM was not verified. Nevertheless, a recent sequencing-based study suggested the presence of CXCR6–CXCL16 interactions in the nasal mucosa of IAV-infected mice, using cell–cell communication analysis (Kazer et al., 2024). The authors showed CXCL16 to be expressed by monocyte-derived macrophages and KNIIFE cells (Krt13+ nasal immune-interacting floor epithelial) cells. Similarly, we found that cells in the NT express CXCL16; however, we did not pinpoint the specific cell population. We suggest two possible mechanisms by which CXCL16 could help recruit CD4 TEM to the NT; either as a soluble chemokine, by attracting CD4 TEM, or as membrane bound, by retaining the CD4 TRM after migration (Koenen et al., 2017; Wilbanks et al., 2001). Depletion with higher antibody dose, longer depletion experiments, or depletion at specific time windows will help cement the role of this chemokine. Still, given the homing of some CXCR6−/− CD4 to the NT, it is plausible that other factors may also be involved in the recruitment and maintenance of TRM.
Functionally, NT CD4 TRM produce cytokines such as IL-2 and IFN-γ upon antigen stimulation. IFN-γ is a cytokine that protects the host by inhibiting viral entry, blocking attachment and replication, and stimulating NK cells proliferation during the early phase of infection (Brass et al., 2009; Feeley et al., 2011; Fong et al., 2022; Weiss et al., 2010). The presence of TRM responding to environmental antigens may explain cytokine production in unstimulated cells. In addition, our scRNA-seq data highlighted the presence of diverse CD4 TRM subtypes, such as cytotoxic CD4, which has also been suggested to play a role in antiviral defense (Brown et al., 2012; McKinstry et al., 2012). In line with these mechanisms, we found that CD4 TRM are essential, while circulating memory CD4 cells are dispensable for local protection in the NT. The role of NT CD4 TRM in limiting viral growth was further validated by our i.t. versus i.n. priming model that established CD4 TRM in lungs only or in both lungs and NT, respectively. Our results agree with previous work demonstrating that mucosal lung CD4 TRM are better at producing IFN-γ and at protecting against IAV infection than are splenic memory CD4 T cells (Teijaro et al., 2011). CTA1-3M2e-DD vaccination provided protection against viral challenge; however, the effect size was limited as the URT-restricted infection was only lethal in 43% (6 of 14) of mice. In this context, it was hard to definitively ascribe an effect to CD4 T cells, even if CD4 depletion resulted in a trend toward decreased survival (4 of 15 mice died in CD4 depleted versus 1 of 15 in CTA1-3M2e-DD vaccinated). Overall, we demonstrated that NT CD4 TRM have coordinated antiviral and tissue repair functions, depending on antigen specificity. However, it remains to be determined how these distinct protective mechanisms are integrated during a physiological, polyclonal response to infection.
Interestingly, we detected a greater proportion of Th17 CD4 TRM in the NT as compared with lungs. Th17 enrichment was independent of both antigen specificity and infection status. In general, NT harbored a substantial proportion of CD4 memory T cells and very few naïve CD4 T cells. Th17 cells present at steady state could have formed in response to commensal microbiota of naïve mice; it is well known that commensal bacteria play an important role in mucosal immunity and tolerance induction (Liu et al., 2024; Pandiyan et al., 2019). We speculate that the Th17 CD4 TRM in the naïve mice might have an immunoregulatory function similar to that of commensal-specific Th17 in the intestine (Brockmann et al., 2023) or that they may have entered earlier in life, in response to other external stimuli. Importantly, lung Th17 cells have been previously reported to protect from IAV infection in an antibody-independent manner (Eliasson et al., 2018; Jaffar et al., 2009; Laan et al., 1999; Lin et al., 2009; McGeachy, 2011; Mills, 2023; Wang et al., 2011). In addition, NT Th17 cells have been shown to form during and protect against several i.n. bacterial infections (Borkner et al., 2021; Solans et al., 2018). All these studies took advantage of Il17a−/− mice, which totally lack IL-17a production, and thus have not been able to determine a definitive role for CD4 Th17 cells specifically. Here, we used a recently described conditional KO, CD4CreRorcfl/fl (Gribonika et al., 2022), which allowed us to exclusively dissect the contribution of Th17 CD4 T cells. We demonstrated that NT Th17 CD4 TRM play a role in reducing apoptosis and somewhat in viral clearance upon heterologous IAV infection, independently of IL-17. Th17 CD4 T cells are also well known to produce IL-22 (Liang et al., 2006), which in turn can stimulate the production of anti-apoptotic factors within airway epithelial cells and maintain a healthy tissue (Omokanye et al., 2022; Sonnenberg et al., 2010). Apoptotic cells were detected in the same areas of viral replication. In addition, we detected IAV-specific Th17 CD4 TRM in healthy human subjects, suggesting that these specific cells are formed and persist in the NT of healthy adults for years and can be reactivated upon reinfection, similar to what was shown for SARS-CoV-2 and Bordetella pertussis (Lim et al., 2022; McCarthy et al., 2024). A recent study reported that upper airways CD4 TRM in humans expressed IL26 (Ramirez et al., 2024); whether these also expressed IL22 or other antiviral cytokines should be investigated in future studies.
Finally, given that nasal respiratory epithelium plays a major role in the transmission of airborne viruses, it is essential to focus on vaccinations that can induce memory cells in the NT (Richard et al., 2020). Although the current intramuscular IAV vaccines are largely effective against IAV infection, they are poor at inducing mucosal immune responses and at protecting against heterologous viruses. Our findings that i.n. CTA1-3M2e-DD immunization can induce M2e-specific CD4 TRM, along with another recent study showing that i.n. Spike vaccination generates antigen-specific CD4 TRM in the NT support i.n. immunization as an effective tool for generating functional CD4 TRM in the NT (Diallo et al., 2023). Nevertheless, immunization strategies to overcome their gradual decay need to be developed.
Collectively, our data provide a comprehensive understanding of NT CD4 TRM that could be leveraged to design mucosal vaccines aimed at generating NT CD4 TRM, providing heterosubtypic protection.
Materials and methods
Mice
We conducted the experiments according to protocols approved by the regional animal ethics committee in Gothenburg (ethical permit numbers: 1666/19, 2230/19 3307/20, and 38/23). C57BL/6N and BALB/c mice were purchased from Janvier Labs, France, and Taconic Biosciences, Denmark. They were housed in the specific pathogen–free animal facility of the Experimental Biomedicine Unit (EBM) at the University of Gothenburg. B6.SJL-PtprcaPepcb/BoyJ (CD45.1+) and B6.129P2-Cxcr6tm1Litt/J (Cxcr6−/−) mice were purchased from the Jackson Laboratory, USA, and were bred at EBM. OVA-specific CD45.1+ OT-II, tdTomato+ OT-II mice TCR-transgenic, CD4CreRorcfl/wt, and CD4CreRorcfl/fl (Gribonika et al., 2022) (all B6 background) were bred at EBM at the University of Gothenburg. We used both female and male mice, which were 8–12 wk old, in the experiments.
Sampling of human subjects
Written consents were obtained from the study participants before obtaining the samples. An ethical permit (ethical permit number: 2023-07055-01) was obtained by the Swedish Ethical Review Authority to collect paired venous blood and nasopharyngeal swabs from healthy subjects aged between 18 and 60 years old. Both male and female volunteers were included. We excluded the subjects who fulfilled any of the following criteria: self-reported respiratory infection during the last 2 wk, allergic rhinitis, chronic obstructive pulmonary disease, asthma, regular nose bleeding, immunodeficiency, and IAV infection in the last 2 years (verified by PCR).
8 ml of venous blood was collected into lithium heparin-coated tubes and was subjected to density gradient centrifugation to obtain PBMCs using Lymphoprep (STEMCELL Technologies) according to the manufacturer’s instructions.
Nasopharyngeal cells (NT) were collected into 3 ml RPMI containing 2% fetal calf serum (FCS) and 1.5 mM dithiothreitol as previously described (Lim et al., 2022). For swab collection, the volunteers were seated, and sample were collected from both nostrils using the same ESwabs (#482C; Copan). Swabs were inserted along the floor of the nasal cavity and rotated in a circular motion. Either the posterior nasopharynx or the inferior nasal turbinates were sampled from different individuals. The cells were vortexed for 1 min and were incubated at 37°C for 30 min. The cells were spun at 700 × g for 8 min at 4°C, and the cell pellets were suspended in FACS buffer.
In total, 60 paired samples were analyzed, but data from 10 of them were excluded for the cytokine, CXCR6, and CD103 analysis because of low recovery of CD4 TRM from NT or PBMC.
Infection of mice
Mouse-adapted influenza A/Puerto Rico/8/34 (PR8) (molecular clone; H1N1), PR8-expressing mCherry protein (PR8 mCherry), and influenza A/HK/x31 (HKx31) (molecular clone; H3N2) were grown in 10-day-old embryonated chicken eggs. The allantoic fluid was harvested, and the viral titer was determined using TCID50 assay. PR8 containing OVA323–339 (PR8-OVA) (Thomas et al., 2006) was kindly gifted by Paul G. Thomas, St. Jude Children’s Research Hospital, Memphis, TN, USA. For sublethal total respiratory tract infection (TRT infection), mice were briefly anesthetized using isoflurane and inoculated i.n, with a sublethal dose (50 TCID50) of mouse-adapted influenza A/Puerto Rico/8/34 (PR8) (molecular clone; H1N1) diluted in HBSS containing 0.1% BSA (total volume of 25 μl). To enhance the amount of CD4 TRM in the NT, mice were infected i.n. with a high dose (105 TCID50) of PR8 or PR8-OVA in a volume of 5 μl (URT-restricted infection) 30 min after the TRT infection.
For the rechallenge experiment after PR8 infection, a lethal dose of 5,000 TCID50 of X31 strain was used. For the rechallenge experiment after CTA1-3M2e-DD immunization, mice were infected i.n. with a high dose (105 TCID50) of PR8 in a volume of 10 μl in a URT-restricted manner.
For i.t., 500 TCID50 of PR8 diluted in HBSS containing 0.1% BSA (total volume of 25 μl) is inoculated intratracheally after anesthetizing the mice.
Immunization of mice
5 µg CTA1-3M2e-DD fusion protein was diluted in 1× PBS to an end volume of 5 μl. BALB/c mice were immunized with CTA1-3M2e-DD i.n. and boosted twice with a 10-day interval. Control group was immunized with 5 µg CTA1-DD adjuvant similarly.
Blocking of CXCL16, IL17A/F, and depletion of T cells in vivo
Mouse CXCL16 antibody (MAB503) was purchased from R&D Systems and was dissolved in 1× PBS. The antibody was administered intraperitoneally at a dosage of 2.5 µg/g weight of mouse on days 1, 3, 6, and 9 after infection with PR8. Mice treated with equal amounts of InVivoMAb rat IgG2a isotype control and anti-trinitrophenol (BE0089; Bio X Cell) were used as controls. For depletion of IL-17A and IL-17 F, InVivoMAb anti-mouse IL-17A (BE0173) and InVivoMAb anti-mouse IL-17F (BE0303) were purchased from Bio X Cell and administered intraperitoneally. The mice received 400 μg of each antibody on day 29 and 200 μg of each antibody from day 30 to 32/33 after PR8 infection. The control group received an equal amount of InVivoMAb mouse IgG1 isotype control, unknown specificity (BE0083) (Bio X Cell).
InVivoMAb anti-mouse CD8α (BE0117), InVivoMAb anti-mouse CD4 (BE0003-1), and InVivoMAb rat IgG2b isotype control (BE0090) were purchased from Bio X Cell and were administered intraperitoneally at a daily dosage of 10 µg/g of mouse from day 28 to day 32 following PR8 infection.
FTY720 treatment
10 mg/ml stock of FTY720 (Cayman Chemicals) was prepared in DMSO. We administrated FTY720 at a dosage of 2.5 µg/g weight of mouse dissolved in sterile physiological saline solution by intraperitoneal injection on indicated days.
Adoptive transfer of antigen-specific T cells
For adoptive transfer of OT-II+ CD4 T cells, CD45.1+ OT-II or tdTomato+ OT-II mice were euthanized, and spleens were collected. Spleens were mashed and made into single-cell suspension by passing through a 70-µm strainer. Total CD4 T cells were isolated using EasySep mouse CD4+ T cell isolation kit (STEMCELL Technologies) according to the manufacturer’s instructions. A total of 2.5–5 × 106 OT-II+ CD4 T cells suspended in 1× PBS were administered by i.v. injection 4 h prior to infection with PR8-OVA.
For generation of effector OT-II T cells, we isolated CD4 T cells from splenocytes of tdTomato+ OT-II mice. OT-II T cells were stimulated by culturing with splenocytes pulsed with 1 μM OVA peptide ISQAVHAAHAEINEAGR in a 5:1 ratio, respectively, in RPMI media containing 10% FBS, 10 µg/ml gentamycin, 55 µM β-mercaptoethanol (Gibco), and 20 U/ml murine IL-2 (PeproTech) for 60 h in 5% CO2 cell culture incubator. The cells were washed and incubated with fluorochrome-conjugated antibodies against surface antigens CD3 PerCPCy5.5 (17A2), CD4 APC-Cy7 (RM4-4), and CD69 BV605 (H1.2F3) for 20 min at 4°C. To exclude dead cells, the cells were washed and stained with LIVE/DEAD Fixable Aqua Dead Cell Stain (cat. no: L34957; Invitrogen) according to the manufacturer’s instructions. Cells were suspended in FACS buffer, and live CD3+CD4+CD69− tdTomato+ OT-II effector T cells were collected after sorting in a BD FACSAria fusion (BD Biosciences). A total of 5 × 105 effector OT-II+ CD4 T cells suspended in 1× PBS were administrated by i.v. injection 60 h after infection with PR8 or PR8-OVA.
BM chimera
For myeloablation, CD45.1+ B6.SJL-PtprcaPepcb/BoyJ mice were irradiated with 11 Gy in equally split doses with a 4-h gap in a RS 2000 X-ray irradiator machine. The mice were rested for a day and transplanted with a mix of 2.5 × 106 tdTomato+ WT and 2.5 × 106 CD45.2+Cxcr6−/− BM cells by i.v. administration. The recipients with newly reconstituted BM (BM chimera) were infected with 50 TCID50 PR8 i.v. 70 days after transplantation.
Isolation and processing of blood, lungs, NALT, NT, and Peyer’s patches (PPs)
For flow cytometry experiments with cells from blood, lungs, NALT, and NT, mice were injected with CD4 FITC or CD4 APC-Cy7 (RM4-4) antibody i.v. to label the circulating CD4 T cells. The mice were completely anesthetized, and blood was withdrawn into EDTA-coated tubes from the heart after 5 min of in vivo antibody labelling. The lung was perfused with 1× PBS and processed into single-cell suspension using the mouse lung dissociation kit (Miltenyi Biotec). Blood was treated with RBC lysis buffer for 5 min, washed, and resuspended in FACS buffer. NT and NALT were isolated and processed immediately as previously described (Pizzolla et al., 2017a). For isolation of NT and NALT, the mouse head was collected and deskinned. Briefly, NALT was obtained by peeling the upper palate using forceps, and the cells were extracted by teasing the NALT between two frosted slides. The rest of the head was cut coronally to remove the brain, and the incisors were removed by scissors. The nasal septum, nasal turbinates, ethmoid turbinates, vomeronasal organs, olfactory recess, and MT, including cartilages and bones, were collected, which is referred to here as NT. NT was digested in 3 ml RPMI 1640 media supplemented with 2% FBS, 6 mg collagenase type 3 (Worthington Biochemical Corporation), and 600 µg DNase I, grade II, from bovine pancreas (Sigma-Aldrich) in a shaking incubator at 37°C for one h. The cells were made into single-cell suspension by straining through a 70-µm nylon mesh. The cells were washed and resuspended in FACS buffer or supplemented media for downstream processing.
For processing of cells from PPs, around three to five PPs were isolated from the small intestine and collected in HBSS media. The PPs were washed twice with HBSS supplemented with 1% FBS, 15 mM HEPES, and 5 mM EDTA by incubation at 37°C for 15 min with occasional shaking. The PPs were mashed, and the cells were made into single-cell suspension by straining through a 70-µm nylon mesh. The cells were washed and resuspended in FACS buffer for staining.
Stimulation of mouse cells with viral peptides
The cells obtained from the organs of naïve and infected mice were stained for extracellular markers prior to stimulation with peptides. The following IAV peptide array was obtained from BEI Resources, National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH): Influenza Virus A/New York/348/2003 (H1N1) NP (NR-50714), A/New York/444/2001 (H1N1) Nonstructural protein 1 (NS1), Influenza Virus A/Puerto Rico/8/1934 (H1N1) HA (NR-18973), Influenza Virus A/Puerto Rico/8/1934 (H1N1) Neuraminidase (NA) (NR-19257), and SARS-CoV-2 N (NR-52404). 1 µg each of all peptides from the peptide array were mixed to obtain the respective peptide pools. 10 million cells derived from lungs and NT were stimulated with IAV NP, NS1, HA, and NA peptide pools containing 1 µg of each peptide pool in RPMI 1640 supplemented with 10% FBS, 1% penicillin/streptomycin and protein transport inhibitor (containing brefeldin A) (diluted 1:1,000) (555029; BD). The cells were incubated for 4 h at 37°C and further washed with 1× PBS to proceed with intracellular cytokine staining.
For comparison of intracellular cytokines produced by CD4 TRM that are stimulated by IAV NP or SARS-CoV-2 N, live CD3+CD4iv−CD4 tissue+ CD44+CD62L−CD69+ CD4 TRM cells were sorted from lungs and NT 30-dpi. The CD4 TRM (CD45.2+) were co-cultured with congenic CD45.1+ splenocytes pulsed with IAV NP peptide pools containing 1 μg of each peptide from the peptide array (NR-50714; BEI Resources) or with SARS-CoV-2 N peptide pools containing 1 μg of each peptide from peptide array (NR-52404; BEI Resources) for 18 h at 37°C in RPMI 1640 supplemented with 10% FBS and 1% penicillin/streptomycin. The culture was supplemented with protein transport inhibitor (containing brefeldin A) (diluted 1:1,000) at the 12th hour. The cells were further washed with 1× PBS to proceed with intracellular cytokine staining.
Stimulation and flow cytometry of human PBMC and nasopharyngeal cells
PBMC and NT cells were incubated with Human TruStain FcX (422302; BioLegend) for 10 min at 4°C. The cells were then incubated with anti-human CD69 BV421 antibody (clone: FNXO) for 20 min at 4°C and washed with FACS buffer. The cells were finally resuspended in T cell stimulation media (RPMI 1640 supplemented with 10% FBS, 1% penicillin/streptomycin, protein transport inhibitor [containing brefeldin A]) (diluted 1:1,000) (555029; BD), monensin (420701; BioLegend), and anti-human CD28/CD49d (347690; BD) (Sekine et al., 2020). The cells were stimulated with peptide pools containing 2 μg each of all peptides from influenza virus A/New York/348/2003 (H1N1) NP (NR-50714) peptide array and influenza virus A/New York/348/2003 (H1N1) M1 (NR-2613) peptide array for 6 h at 37°C. Cells without the peptide pools (peptide diluent) were kept as unstimulated controls. After washing with FACS buffer, the cells were stained for extracellular antigens with the following fluorochrome-conjugated antibodies (clone used indicated in bracket): anti-CD3 BUV737 (UCHT1), anti-CD4 PE-Dazzle594 (OKT4), anti-CD45RA BV605 (HI100), anti-CCR7 APC-Cy7 (G043H7), anti-CD103 FITC (Ber-ACT8), and anti-CXCR6 (13B1E5). After incubation at 4°C for 20 min, the cells were washed and stained with LIVE/DEAD Fixable Aqua Dead Cell Stain (cat. no: L34957; Invitrogen) according to the manufacturer’s instructions to exclude dead cells. To detect intracellular cytokines, the cells were fixed with 4% paraformaldehyde for 20 min at room temperature (RT), washed with ice-cold 1× PBS, and permeabilized with permeabilization buffer (1× PBS containing 0.5% BSA and 0.5% saponin) for 10 min at RT. The cells were washed and resuspended in permeabilization buffer with the following fluorochrome-conjugated antibody cocktail: anti- IFN-γ PE (25723.11), anti-TNF BV650 (MAb11), and anti-IL-17a APC (BL168). After incubation at 4°C overnight, the cells were washed and suspended in FACS buffer for data acquisition at BD LSRFortessa X-20 or Sony ID7000 spectral analyzer.
Flow cytometry of murine cells
All fluorochrome-conjugated antibodies are titrated to determine the optimal concentration for flow cytometry. Single-cell suspension from organs were prepared and resuspended in FACS buffer as mentioned before. The following antibodies are used for mouse extracellular antigen staining: anti-CD3 PerCPCy5.5 (17A2), anti-CD4 FITC (RM4-4), anti-CD4 APC-Cy7 and BUV496 (RM4-5), anti-CD8 PE-Cy7 (53-6.7), anti-CD44 BV786 and PE-Dazzle 594 (IM7), anti-CD62L BV711 and APC (MEL-14), anti-CD69 BUV737 (H1.2F3), anti-CD11a PE-Cy7 (H155-78), anti-CD103 PE-Cy7, BV421 and BV711 (2E7), anti-CD45.1 APC (A20), anti-CD45.2 BUV395 (104), anti-CXCR6 BV421 (SA051D1), anti-ICOS BV421 (7E.17G9), anti-PD1 PE (J43), anti-CD19-Alexa Flour 700 (6D5), and anti-CD11b-Alexa Flour 700 (M1/70). The extracellular staining was performed by incubating the cells with the antibodies for 20 min at 4°C. For labelling antigen-specific cells with fluorochrome-conjugated tetramers, cells were incubated with the tetramers for 2 h 40 min at 4°C (I-Ab NP306–322 APC and I-Ab HA91–107 PE/BV421 tetramers) or 1 h at RT (I-Ad M2e2–17 PE), and the extracellular antibody mix was added on top and incubated for 20 min. The following tetramers, kindly provided by NIH Tetramer Core Facility at Emory University were used: I-Ab NP306–322 APC, I-Ad M2e2–17 PE, I-Ab HA91–107 PE, and I-Ab HA91–107 BV421. I-Ab human CLIP87–101 conjugated with APC, PE, or BV421 and I-Ad human CLIP 87–101 conjugated with PE were used as negative controls. To exclude dead cells, the cells were washed and stained with LIVE/DEAD Fixable Aqua Dead Cell Stain (cat. no: L34957; Invitrogen) according to the manufacturer’s instructions. The cells were fixed with IC fixation buffer (00-8222; eBioscience) for intracellular cytokine staining or fixed with Foxp3 Transcription Factor Fixation/Permeabilization buffer (00-5523; eBioscience) for transcription factor staining for 60 min at RT. The cells were washed and resuspended in 1× permeabilization buffer (00-8333; eBioscience) and incubated with the following antibodies: anti-IL-2 BV785 (JES6-5H4), anti-TNF BV421 (MP6-XT22), anti-IFN-γ BV605 (XMG1.2), anti-IL-17a PE or FITC (TC11-18H10), anti-RORγt PE-CF594 (Q31-378), and anti-Ki-67 V450 (B56). The cells were washed with 1× permeabilization buffer and resuspended in FACS buffer. The labelled cells were run, and the data were acquired on the BD LSRFortessa X-20 or BD LSR II (BD Biosciences) flow cytometer or Sony ID7000 Spectral cell analyzer and were analyzed using FlowJo software (Tree Star).
Immunofluorescence staining
Mice were euthanized, and the upper parts of mice heads were collected after removing the skin and eyeballs. The heads were fixed with 1.5% formaldehyde solution (wt/vol) (Thermo Fisher Scientific) solution at 4°C for 24 h. After removing the teeth and remaining muscle tissues, they were transferred into 0.5 M EDTA solution for decalcification for 7 days at 4°C. The samples were embedded into sequential dilutions of OCT paramount (HistoLab) and were snap-frozen for sectioning. 10–14-μm thick frontal sections were cut, and SuperFrost microscopy slides were stored at −80°C or were processed for staining. In the latter case, slides were baked for 1 h at 60°C. Antigen retrieval was performed on NT sections mounted on glass slides to enhance epitope accessibility for immunofluorescence staining. Slides were submerged in a sodium citrate retrieval buffer consisting of 10 mM sodium citrate and 0.05% Tween-20, adjusted to pH 6.0. Heat-induced epitope retrieval was carried out by placing the slides in the buffer and heating them in a microwave until the solution reached 100°C. Once the target temperature was reached, the slides were removed from heat and allowed to cool gradually to RT for ∼1 h. Following cooling, slides were fixed with ice-cold acetone until the samples were dried. The tissue was blocked with 1× PBS containing 10% FBS and 5% normal rat serum for 15 min at RT. After washing with 1× PBS, the tissues were stained with antibody cocktail overnight at 4°C in the dark. The following antibodies were used: anti-CXCL16 (Uncoupled Polyclonal, #bs-1441R-TR; Biosciences), anti-CD4 (FITC RM4-4, #116004; BioLegend), anti-CD44 (APC IM7, #103012; BioLegend), anti-CD45.1 (PE A20, #110707; BioLegend), and goat anti-rabbit IgG (Texas Red, #sc-2780; Santa Cruz Biotech). The sections were counterstained by incubating with Hoechst 33342 solution (Thermo Fisher Scientific) for 15 min at RT in the dark. The sections were mounted with a fluorescent mounting medium (Dako), acquired on an Axio Imager Z2 (Zeiss) and analyzed on TissueFACS Analyzer (version 7.1.120 Xylis).
TUNEL assay
Mice were euthanized, and the upper part of the mice heads were collected after removing the skin and eyeballs. The heads were fixed with 4% formaldehyde solution (wt/vol) (Thermo Fisher Scientific) solution at 4°C for 24 h. After removing the teeth and remaining muscle tissues, they were transferred into 14% EDTA solution (wt/vol) for decalcification for 14 days at 4°C. The tissues were sequentially dehydrated and embedded into paraffin wax in a cassette. 4-μm thick sections were cut from the paraffinized tissue blocks. TUNEL assay kit -HRP-DAB (ab206386; Abcam) was used to detect apoptotic cells according to the manufacturer’s instructions. The images were acquired on an Axio Imager Z2 (Zeiss) and analyzed on TissueFACS Analyzer (version 7.1.120 Xylis) and QuPath (version 0.5.1) software.
Neutralization assay
200,000 Madin–Darby canine kidney (MDCK) cells were plated in a 24-well plate and were incubated overnight at 37°C. Serum from IAV-infected mice or mice immunized with irrelevant protein (streptavidin or SARS-CoV-2 spike protein) was serially diluted (twofold) and incubated with 106 TCID50 X31 (MOI = 5) in 500 μl DMEM supplemented with 1 mM HEPES, 1 µg/ml TPCK trypsin, and 5 µg/ml gentamycin for 1 h at 37°C. The plate containing MDCK cells was washed twice with 1× PBS, and the virus/serum mixture was added onto the cells. The plate was incubated for 18 h and washed twice with 1× PBS. The cells were incubated with 5× trypsin for cell detachment, and the washed cells were fixed with fixation/permeabilization solution (eBioscience Foxp3/Transcription Factor Staining Buffer Set, 00-5523-00) overnight at 4°C. The cells were washed twice with FACS buffer containing 0.1% saponin. The infected cells were labelled by incubating cells with InVivoMAb anti-Influenza A virus NP (clone H16-L10-4R5 [HB65]) (Bio X Cell) labelled with Alexa Fluor 488 (Protein Labeling Kits for Alexa Fluor 488, A10235; Invitrogen) for 30 min at 4°C. The cells were washed twice with FACS buffer containing 0.1% saponin and resuspended in FACS buffer for flow cytometry. The average signals from cells incubated with virus in the absence of serum were considered 100% infectivity and are used to calculate the relative infectivity of cells in the presence of serum.
Viral titer determination
Lung and URT (NT and NALT) are harvested and suspended in 1 ml 1× PBS per 0.5 g of organ. The tissues were homogenized using a tissue homogenizer and spun at 1,200 × g for 10 min at 4°C. The supernatant was used for determining the viral titer using the standard TCID50 assay.
scRNA-seq
Mice were injected with CD4 FITC (RM4-4) antibody i.v. to label the circulating CD4 T cells (referred to as CD4 i.v.). The mice were completely anesthetized, and the lung was perfused with 1× PBS. NT and lungs were isolated, digested, and processed into single cells as described before. The samples were enriched for CD4 T cells using the EasySep mouse CD4+ T Cell isolation kit (STEMCELL Technologies), and the dead cells were removed using EasySep Dead Cell Removal (Annexin V) Kit (STEMCELL Technologies) according to the manufacturer’s instructions. Cells from PR8-infected mice were labelled with the following fluorochrome-conjugated MHC-II tetramers: I-Ab NP306–322 APC and I-Ab HA91–107 BV421 as described before. After washing with FACS buffer, cells were labelled with TotalSeq-C0987 anti-APC antibody (408007; BioLegend) to identify NP-specific CD4 TRM. Cells were washed and labelled with antibodies. The following fluorochrome-labelled antibodies were used: CD3 PerCPCy5.5 (17A2), CD4 APC-Cy7 (RM4-4) (for labelling tissue-resident CD4 Tc- referred to as CD4 tissue), CD69 BV605 (H1.2F3), CD44 PE-Dazzle 594 (IM7) and CD62L BV711 (MEL-14). Additionally, totalSeq-C0595 anti-mouse CD11a antibody (101131; BioLegend) and the following barcoded hashtag antibodies were used: hashtag antibodies 3,5–9 (155865, 155869, 155871, 155873, 155875, and 155877; BioLegend) for demultiplexing of samples during data processing and to detect cells expressing CD11a protein on the cell surface. Live total CD4 TRM (CD4 iv− CD4 tissue+CD44+CD62L−CD69+) were sorted from PR8-infected and naïve mice, while NP-specific CD4 TRM (NP306–322+ CD4 iv− CD4 tissue+CD44+CD62L−CD69+) and HA-specific CD4 TRM (HA91–107+ CD4 iv− CD4 tissue+CD44+CD62L−CD69+) were sorted from PR8-infected mice. In addition to CD4 T cells, we sorted total live cells devoid of CD4 T cells from the NT of naïve and PR8-infected mice. The sorted cells were collected in a BD FACSAria fusion (BD Biosciences) cell sorter and processed immediately. The total number of cells sorted from each group is the following. Lung total CD4 TRM from naïve mice: 8,000 cells, NT total CD4 TRM from naïve mice: 3,095 cells, lung NP-specific CD4 TRM: 4,002 cells, NT NP-specific CD4 TRM: 1,172 cells, lung HA-specific CD4 TRM: 4,073 cells, NT HA-specific CD4 TRM: 1,314 cells, lung total CD4 TRM from infected mice: 8,000 cells, NT total CD4 TRM from infected mice: 3,000 cells, and live cells except CD4 T cells from NT of naïve and infected mice: 10,000 cells each. The sorted cells were pooled and washed in the Laminar Wash Mini system (Curiox Biosystems).
A maximum of 20,000 cells were processed into single cells in a chromium controller (10x Genomics). The single-cell gene expression, TCR VDJ, and cell surface protein libraries were prepared using Chromium Next GEM Single Cell 5′ Reagent Kits version 2 (Dual Index) kit (PN-1000265), library construction kit (PN-1000190), 5′ Feature Barcode Kit (PN-1000256), Chromium Single Cell Mouse TCR Amplification Kit (PN-1000254), Chromium Next GEM Chip K Single Cell Kit (1000286), Dual Index Kit TT Set A (PN-1000215), and Dual Index Kit TN Set A (PN-1000250). The prepared cDNA libraries were quantified using Qubit Fluorometer (Invitrogen), and quality was assessed using Agilent TapeStation System. The libraries were sequenced on Illumina NovaSeq 6000 by SNP&SEQ Technology Platform, Science for Life Laboratory (Uppsala Biomedical Centre, Uppsala University, Sweden), according to sequencing instructions provided by 10x Genomics.
scRNA-seq data processing
Raw fastq files were processed through the 10x Cell Ranger pipeline using the multi command and default parameters with reference genome GRCm38-mm10. Raw UMI count matrices generated from the Cell Ranger 10x pipeline were loaded and merged into a single Seurat object (Seurat version 4) (Hao et al., 2021; Hao et al., 2024; Satija et al., 2015). Cells were discarded if they met any of the following criteria: percentage of mitochondrial counts was >15%; number of unique features or total counts was in the bottom or top 0.5% of all cells; and number of unique features was <250. Gene counts were normalized to the same total counts per cell and were natural log transformed (after the addition of a pseudocount of 1). The normalized counts in each cell were mean-centered and scaled by their SD, and the following variables were regressed out: percentage of mitochondrial counts, percentage of ribosomal counts, G2M and S phase scores, and TCR gene expression scores. Hashtags and APC-surface antibody were normalized by a centered log-ratio normalization, and binding was assigned using HTODemux function. Data integration across cells originating from different samples was done using the Anchor method within Seurat. Selection of the number of components for the nearest-neighbor network computation was based on their visualization in an elbow plot. UMAP (Becht et al., 2018) was performed for the spatial visualization of the single-cell data set after features and cells were clustered using the Louvain algorithm. Clusters were manually curated. TCR data were processed using scRepertoire package (Borcherding et al., 2020) with standard setting. T cell clonality was based on identical nucleotide sequence in the CDR3 region. All TCR information was integrated with the scRNA-seq data by aligning and merging the data with the metadata slot in the processed RNA-seq Seurat object. Differentially expressed genes between different clusters were identified using the FindAllMarkers function from Seurat using default settings (Wilcoxon test and Bonferroni P value correction). Significant genes with average log fold change >0.25 and expressed in >25% of cells in that group were ranked according to fold change and are reported in Table S1. Cell signatures were based on gene lists indicated in the figure legends and their expression score computed using Mann-Whitney U statistic via the UCell package (Andreatta and Carmona, 2021). For comparison between two groups, the FindMarkers function was used, and either the top and bottom 20 genes were represented by a heatmap using the DoHeatmap function of Seurat or all genes were represented on a VolcanoPlot using the EnhancedVolcano R package (Blighe et al., 2024). Data underlying the volcano plot are in Table S1. Gene signatures were added to the Seurat object using the AddModuleScore function.
Statistical analysis
The data were analyzed using GraphPad Prism software. The experiments were performed and analyzed in a non-blinded fashion. Both male and female mice were included in the experiments. For statistical analysis, unpaired two-tailed t test, one-way ANOVA with Dunnett’s multiple comparison test, two-way ANOVA, Tukey’s multiple comparison test, or two-sided Wilcoxon matched-pairs signed-rank test is used and is indicated in the figure legends. Normality and lognormality were tested using GraphPad prism. Differences were considered significant when the P value was <0.05. Outliers were identified and removed using Grubbs’ method (alpha = 0.05) when indicated in the figure legend. For weight loss analysis, we used a linear mixed effects analysis of the body weight data (normalized to the initial weight of each individual animal) in R (https://www.r-project.org/). As fixed effects, we used the treatment and the day (with an interaction term between those fixed effects). As random effects, we had intercepts for the individual animals. Thereafter, we compared the weight curves across treatment groups (averaged over time) using estimated marginal means and pairwise comparisons with Tukey’s honest significant difference adjustment. To assess treatment group differences at each time point, we used the same linear mixed-effects model described above. Estimated marginal means were computed for each treatment group at each individual day (0–14). Pairwise comparisons between treatment groups were performed at each time point, and P values were adjusted for multiple testing using Holm’s method.
Online supplemental material
Fig. S1 is related to Fig. 1 and contains the gating strategy for CD4 TRM, kinetics for the frequency of antigen-specific CD4 TRM, and data showing the requirement of cognate antigen for establishment of CD4 TRM in the lungs. Fig. S2 is related to Fig. 2 and compares the frequency of cytokine-expressing CD4 TRM in the lungs and NT upon peptide stimulation, flow cytometry plots of stimulated and unstimulated controls, and MFI for PD1 and ICOS. Fig. S3 is related to Fig. 3 and shows frequency of antigen-specific CD4 TRM in NT and lungs on day 6 following primary infection or secondary heterologous infection, frequency of T cells in the blood after T cell depletion, neutralization assay, and data on CTA1-DD M2e immunization experiments. Fig. S4 is related to Fig. 4 and contains scRNA-seq analysis data. Fig. S5 is related to Fig. 6, Fig. 7, and Fig. 8 and contains percentage of donor and recipient cells in the BM chimera, controls for CXCL16 staining, gating strategy to identify different cell populations in human PBMC, flow cytometry plots for RORγt+CD4+ T cells in the PPs, magnified microscopic images for TUNEL+ cells, and data on viral titer and TUNEL+ cells upon IL-17a/f antibody treatment. Table S1 is related to scRNA-seq data and shows differentially expressed genes across clusters and all data underlying Fig. S4 E. Table S2 is related to Fig. S3, N–P and shows different statistical comparisons across groups.
Data availability
scRNA-seq and TCR-seq data have been deposited in the NCBI GEO database under accession number GSE270932. All other data are present in the article and supplementary information.
Acknowledgments
We would like to thank the staff at the Experimental Biomedicine core facility at the University of Gothenburg for animal management; Paul G. Thomas (St. Jude Children’s Hospital, Memphis, TN, USA) for PR8-OVA virus; Linda M Wakim, Doherty Institute, for providing the protocol for processing nasal tissue; Carina Karlsson, University of Gothenburg, for providing access to microtome; Alvar Almstedt, Bioinformatics and Data Centre, Core Facilities, University of Gothenburg, for data preprocessing; the Genomics core facility at Sahlgrenska Hospital for providing access to the Qubit Fluorometer and TapeStation; and study participants for providing samples. MHC-II tetramers were obtained from the NIH tetramer core facility at Emory University. The following reagents were obtained through BEI Resources, NIAID, NIH: Peptide Array, Influenza Virus A/New York/348/2003 (H1N1) Nucleocapsid Protein, NR-5071; Peptide Array, Influenza Virus A/New York/348/2003 (H1N1) Matrix protein 1 (M1) (NR-2613); and Peptide Array, SARS-Related Coronavirus 2 Nucleocapsid (N) Protein (NR-52404). Sequencing was performed by the SNP&SEQ Technology Platform in Uppsala. The facility is part of the National Genomics Infrastructure Sweden and Science for Life Laboratory. The SNP&SEQ Platform is also supported by the Swedish Research Council and the Knut and Alice Wallenberg Foundation. The graphical abstract and the experiment schemes are created with Biorender.com.
The project was supported by grants from the European Research Council (ERC-StG, B-DOMINANCE, grant no. 850638 to D. Angeletti), the Swedish Research Council (grant nos. 2021-01164 and 2021-01165 to D. Angeletti), the Knut and Alice Wallenberg Foundation (grant nos. 2021.0033 and 2024.0097 to D. Angeletti), and the Sahlgrenska Academy (GU2020/2376-JNR2020 to D. Angeletti). N.R. Mathew is supported by a Svenska Sällskapet för Medicinsk Forskning post-doctoral grant (grant no. PD20-0017), the Elisabeth Bollan Linden Stipend, Stiftelsen Lars Hiertas Minne, and Stiftelsen Wilhelm och Martina Lundgrens Vetenskapsfond. K. Tang is supported by grants from Svenska Sällskapet för Medicinsk Forskning (S21-0083). Open Access funding is provided by Gothenburg University.
Author contributions: Nimitha R. Mathew: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, software, validation, visualization, and writing—original draft, review, and editing. Romain Gailleton: data curation, formal analysis, investigation, methodology, validation, visualization, and writing—review and editing. Lydia Scharf: formal analysis, methodology, and writing—review and editing. Karin Schon: investigation. Josue Enriquez: formal analysis, investigation, and writing—review and editing. Hannes Axelsson: formal analysis, investigation, and writing—review and editing. Anneli Stromberg: methodology and resources. Nils Lycke: formal analysis, resources, and supervision. Mats Bemark: formal analysis, investigation, methodology, resources, supervision, and writing—review and editing. Ka-Wei Tang: formal analysis, investigation, methodology, supervision, and writing—review and editing. Davide Angeletti: conceptualization, formal analysis, funding acquisition, investigation, methodology, project administration, software, supervision, visualization, and writing—original draft, review, and editing.
References
Author notes
Disclosures: The authors declare no competing interests exist.