Complex organ shapes arise from the coordinate actions of individual cells. The Drosophila egg chamber is an organ-like structure that lengthens along its anterior–posterior axis as it grows. This morphogenesis depends on an unusual form of planar polarity in the organ’s outer epithelial layer, the follicle cells. Interestingly, this epithelium also undergoes a directed migration that causes the egg chamber to rotate around its anterior–posterior axis. However, the functional relationship between planar polarity and migration in this tissue is unknown. We have previously reported that mutations in the Misshapen kinase disrupt follicle cell planar polarity. Here we show that Misshapen’s primary role in this system is to promote individual cell motility. Misshapen decreases integrin levels at the basal surface, which may facilitate detachment of each cell’s trailing edge. These data provide mechanistic insight into Misshapen’s conserved role in cell migration and suggest that follicle cell planar polarity may be an emergent property of individual cell migratory behaviors within the epithelium.
Introduction
During development, changes in organ shape require the precise coordination of individual cell behaviors across tissues. The Drosophila egg chamber provides a highly tractable system to investigate the cellular control of organ morphogenesis. Egg chambers are simple, multicellular structures within fly ovaries that will each give rise to a single egg. They consist of a central germ cell cluster surrounded by an epithelial layer of follicle cells. The apical epithelial surface faces the germ cells, whereas the basal surface contacts a basement membrane (BM) extracellular matrix (ECM; Fig. 1, A and B). Though initially spherical, each egg chamber lengthens along its anterior–posterior (AP) axis as it grows. The elongation depends on an unusual form of planar polarity at the basal epithelial surface, in which linear bundles of actin filaments and fibril-like structures in the BM both align perpendicular to the AP axis (Gutzeit, 1990; Gutzeit et al., 1991). The resulting circumferential arrangement of structural molecules, which is first evident at the onset of elongation, is thought to act as a “molecular corselet” that directionally biases egg chamber growth (Gutzeit et al., 1991; He et al., 2010; Haigo and Bilder, 2011). Follicle cell planar polarity is independent of the Frizzled/Strabismus and Fat/Dachsous planar cell polarity pathways (Viktorinová et al., 2009). Instead, this system largely relies on cell–BM interactions, as mutations that block these processes disrupt planar polarity and produce round eggs (Bateman et al., 2001; Frydman and Spradling, 2001; Conder et al., 2007).
Msn functions cell-autonomously to promote follicle cell migration. (A) Overview of egg chamber structure. (B) Schematic for visualization of follicle cell migration. (C and D) msn-RNAi epithelia (n = 15) show migratory defects compared with control epithelia (n = 19). All imaging was performed on stage 6–7 egg chambers. (C) Stills from a time lapse (Video 1) of stage 6 control and msn-RNAi epithelia, in which a single follicle cell is monitored over time (blue and yellow dots). (D) Maximum intensity kymograph drawn through control or msn-RNAi follicle cells (dotted lines). (E) Maximum intensity kymograph drawn through wild-type (blue) and msn172 (yellow) cells in a mosaic epithelium (Video 2). (F) Stills from a time lapse (Video 3) of an msn-RNAi flipout clone (GFP). Bars: (C, E, and F; egg chamber images) 20 µm; (D and E; kymographs) 5 µm.
Msn functions cell-autonomously to promote follicle cell migration. (A) Overview of egg chamber structure. (B) Schematic for visualization of follicle cell migration. (C and D) msn-RNAi epithelia (n = 15) show migratory defects compared with control epithelia (n = 19). All imaging was performed on stage 6–7 egg chambers. (C) Stills from a time lapse (Video 1) of stage 6 control and msn-RNAi epithelia, in which a single follicle cell is monitored over time (blue and yellow dots). (D) Maximum intensity kymograph drawn through control or msn-RNAi follicle cells (dotted lines). (E) Maximum intensity kymograph drawn through wild-type (blue) and msn172 (yellow) cells in a mosaic epithelium (Video 2). (F) Stills from a time lapse (Video 3) of an msn-RNAi flipout clone (GFP). Bars: (C, E, and F; egg chamber images) 20 µm; (D and E; kymographs) 5 µm.
New insight has come from the recent discovery that egg chamber elongation is also linked to an unexpected epithelial migration. Coincident with planar polarization of the epithelium, the basal follicle cell surfaces migrate on the inside of the BM, such that the migration path is oriented perpendicular to the AP axis. This process causes the entire egg chamber to rotate within the BM, which remains largely stationary. The current model is that follicle cell migration creates the fibril-like structures in the BM, which contribute to the constrictive corselet (Haigo and Bilder, 2011). This migration is unusual, in that the follicle cells form a continuous epithelium with no leading edge. It is therefore possible that the planar polarity in this tissue coordinately aligns the cells’ front–rear axes to set the direction for collective movement. However, the functional relationship between follicle cell planar polarity and tissue migration is unknown.
We have previously reported that mutations in the Ste20 kinase Misshapen (Msn) disrupt follicle cell planar polarity (Horne-Badovinac et al., 2012). Here we show that Msn functions autonomously within each follicle cell to promote its motility. Msn decreases integrin levels at the basal epithelial surface, which appears to facilitate the detachment of each cell’s trailing edge. These findings provide mechanistic insight into Msn’s conserved role in cell migration. They also suggest that follicle cell planar polarity may, in fact, be an emergent property of individual cell migratory behaviors within the epithelium.
Results and discussion
To investigate Msn’s role in egg chamber elongation, we depleted the protein from the follicle cells by RNAi using TubP-Gal4 (Zhu and Stein, 2004), and confirmed that this condition produces round eggs (Fig. S1 A). Although control epithelia migrated normally, Msn-depleted epithelia remained completely stationary (Fig. 1, C and D; and Video 1). Importantly, msn-RNAi egg chambers also become significantly rounder than controls, concurrent with the onset of follicle cell migration at stage 6 (Fig. S1 B). These data suggest that a key role for Msn during egg chamber elongation is to promote follicle cell migration.
Msn promotes follicle cell migration. Live imaging of control and msn-RNAi egg chambers (Fig. 1 C). Samples were analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64. Frames were taken every 30 s for 22.5 min. Playback rate is 300× real time.
Msn promotes follicle cell migration. Live imaging of control and msn-RNAi egg chambers (Fig. 1 C). Samples were analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64. Frames were taken every 30 s for 22.5 min. Playback rate is 300× real time.
We have previously reported that loss of Msn in large clones leads to a global defect in follicle cell planar polarity during migration stages (Horne-Badovinac et al., 2012). We therefore hypothesized that the motility defect exhibited by msn-RNAi epithelia might occur at the tissue level, reflecting an inability of the follicle cells to coordinately orient their front–rear axes. Surprisingly, however, visualization of migration dynamics in mosaic epithelia revealed that Msn is autonomously required for individual cell motility. Although large msn mutant clones block egg chamber rotation, the wild-type cells still show what appears to be a very slow, directed migration. In contrast, the mutant cells are completely still (Fig. 1 E and Video 2). To gain further insight into this defect, we produced small clones of 2–3 cells expressing msn-RNAi. Here, the wild-type cells migrate relatively normally (0.42 µm/min), and the egg chamber rotates as in controls (Fig. 1 F and Video 3). Interestingly, the Msn-depleted cells now also migrate, but at a much slower rate (0.14 µm/min), likely due to incomplete knockdown. Unlike normal follicle cell migration, where individual cells rarely change neighbors (Haigo and Bilder, 2011), the wild-type cells easily slide past the slower moving clone, showing that variations in migration speed across the tissue can induce unexpected changes in cell–cell contacts. These data show that, although large msn clones can inhibit the migration of the entire epithelium, Msn primarily functions cell-autonomously to promote individual follicle cell motility.
A large msn mutant clone blocks global follicle cell migration.msn172 cells (yellow dots) are stationary, while neighboring wild-type cells attempt to move past them (Fig. 1 D). The msn mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64. Frames were taken every 30 s for 61 min. Playback rate is 300× real time.
A large msn mutant clone blocks global follicle cell migration.msn172 cells (yellow dots) are stationary, while neighboring wild-type cells attempt to move past them (Fig. 1 D). The msn mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64. Frames were taken every 30 s for 61 min. Playback rate is 300× real time.
msn-RNAi mosaic egg chambers exhibit a cell-autonomous defect in follicle cell migration. The cells in an msn-RNAi flipout clone (GFP) move very little while neighboring wild-type cells efficiently migrate past them (Fig. 1 F). The mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64 (red). Frames were taken every 30 s for 32 min. Playback rate is 300× real time.
msn-RNAi mosaic egg chambers exhibit a cell-autonomous defect in follicle cell migration. The cells in an msn-RNAi flipout clone (GFP) move very little while neighboring wild-type cells efficiently migrate past them (Fig. 1 F). The mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64 (red). Frames were taken every 30 s for 32 min. Playback rate is 300× real time.
During these mosaic analyses, we observed a novel phenotype that further highlights Msn’s cell-autonomous function. When a clone is confined to one of the egg chamber’s poles, the wild-type cells at the clone boundary break contact with their mutant neighbors and invade the egg chamber’s interior, sometimes bisecting the germ cell cluster (Fig. 2 A). Under these conditions, planar polarization of the basal actin filaments is maintained (Fig. 2 B), and the egg chambers elongate (Fig. 2, A–C). Unlike genetic perturbations that cause mutant follicle cells to invade the germ cells (Goode and Perrimon, 1997; Bilder et al., 2000; Lu and Bilder, 2005), the wild-type cells maintain their cuboidal morphology and contact with a BM (Fig. S1 C). Depletion of Msn specifically in the anterior follicle cells using C306-Gal4 also causes invasion (Fig. 2 C).
Terminal msn clones induce a novel invasion phenotype. (A) Transverse section showing that a posterior terminal msn clone causes neighboring wild-type cells (GFP) to invade the germ cells (arrowheads). (B) An invasion event (arrowheads) does not disrupt planar polarization of the basal actin filaments. (C) Expressing msn-RNAi exclusively in the anterior follicle cells (GFP) also causes invasion (arrowheads). (D) Initial and final frames of a time lapse (Video 4) taken of an msn172 mosaic egg chamber with an anterior terminal clone (bracket). Wild-type cells (GFP) adjacent to the mutant clone invade the germ cells (arrowheads). (E) Stills from the same time lapse acquired at the basal surface show that the noninvading wild-type cells continue to migrate. Bars are as indicated in the figure. (F) Model for how the invasion phenotype develops over time.
Terminal msn clones induce a novel invasion phenotype. (A) Transverse section showing that a posterior terminal msn clone causes neighboring wild-type cells (GFP) to invade the germ cells (arrowheads). (B) An invasion event (arrowheads) does not disrupt planar polarization of the basal actin filaments. (C) Expressing msn-RNAi exclusively in the anterior follicle cells (GFP) also causes invasion (arrowheads). (D) Initial and final frames of a time lapse (Video 4) taken of an msn172 mosaic egg chamber with an anterior terminal clone (bracket). Wild-type cells (GFP) adjacent to the mutant clone invade the germ cells (arrowheads). (E) Stills from the same time lapse acquired at the basal surface show that the noninvading wild-type cells continue to migrate. Bars are as indicated in the figure. (F) Model for how the invasion phenotype develops over time.
We have observed how this surprising phenotype develops by performing live imaging on an egg chamber with a small anterior clone. At the start of the experiment, the wild-type cells at the clone boundary had just begun to invade (Fig. 2 D, 0 min). Imaging at the basal surface of the same egg chamber revealed that the noninvading wild-type cells migrated normally, and the egg chamber rotated (Fig. 2 E and Video 4). Returning to the transverse plane, the invading wild-type cells had continued to move toward one another through the germ cell cluster (Fig. 2 D, 90 min). These data suggest that the wild-type cells undergo a wound healing–like response to exclude the nonmigratory mutant cells from the epithelium and preserve the ability of the remaining cells to migrate (schematized in Fig. 2 F). Thus, the invasion phenotype appears to be an extreme example of the cell-autonomous nature of the msn migration defect that also induces a novel behavior in the wild-type cells.
The presence of an msn172 mutant clone at the anterior leads to invasion of neighboring wild-type cells (GFP) into the germ cell cluster. The time-lapse begins at a transverse section, then goes up to a more basal plane before returning back to the transverse section (Fig. 2, D and E). The mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64 (red). Frames were taken every 30 s for 90 min. Playback rate is 300× real time
The presence of an msn172 mutant clone at the anterior leads to invasion of neighboring wild-type cells (GFP) into the germ cell cluster. The time-lapse begins at a transverse section, then goes up to a more basal plane before returning back to the transverse section (Fig. 2, D and E). The mosaic egg chamber was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Cell membranes are labeled with FM4-64 (red). Frames were taken every 30 s for 90 min. Playback rate is 300× real time
Previous work had demonstrated that clones of immotile follicle cells can be carried along the migration path by their wild-type neighbors (Haigo and Bilder, 2011). Why then do msn-null mutant clones remain stationary? Msn homologues in other organisms have been implicated in regulating integrin-based adhesion (Becker et al., 2000; Poinat et al., 2002; Wright et al., 2003; Martynovsky et al., 2012). Therefore, we next investigated whether Msn performs a similar function in this context. The predominant integrin heterodimer expressed by the migrating follicle cells contains the α-PS1 and β-PS subunits (Delon and Brown, 2009). Immunofluorescence studies of mosaic epithelia revealed a cell-autonomous increase in both subunits at the basal surface of msn cells (Fig. 3 A). In contrast, expression of an activated, membrane-tethered form of Msn (Myr-Msn; Kaneko et al., 2011) decreased integrin levels (Fig. 3 B). In some tissues, Msn activates the c-Jun N-terminal kinase (JNK) signaling pathway (Su et al., 1998; Paricio et al., 1999); however, loss of the JNK kinase Basket in follicle cell clones does not affect integrin levels (Fig. S2 A). These results indicate that Msn functions in a JNK-independent manner to decrease integrins at the basal follicle cell surface.
Msn reduces integrin-based adhesion. (A) Basal views of msn mosaic epithelia stained with antibodies against α-PS1 (maximum projection) or β-PS show increased integrin levels at the basal surfaces of msn cells. GFP marks wild-type cells. (B) A clone of cells expressing an activated form of Msn (green) shows reduced integrins at the basal surface. (C) Maximum projection of Vkg-GFP in the BM. Loss of Msn disrupts BM structure in a cell-autonomous manner, and BM structure is restored by introducing one copy of a loss-of-function β-PS mutation (mysXG43/+; msn-RNAi). Bars are as indicated in the figure. (D) Removing one copy of β-PS also rescues the defect in follicle cell migration seen in msn-RNAi, whereas removing one copy of β-PS alone or overexpressing Msn (UAS-Msn) increases the migration rate. Graph shows migration rates of individual stage 6–7 egg chambers for each condition. Numbers indicate fraction of egg chambers that showed follicle cell migration. The data shown are from a single representative experiment out of two repeats. Each experimental condition involves egg chambers dissected from >10 female flies, imaged over multiple days.
Msn reduces integrin-based adhesion. (A) Basal views of msn mosaic epithelia stained with antibodies against α-PS1 (maximum projection) or β-PS show increased integrin levels at the basal surfaces of msn cells. GFP marks wild-type cells. (B) A clone of cells expressing an activated form of Msn (green) shows reduced integrins at the basal surface. (C) Maximum projection of Vkg-GFP in the BM. Loss of Msn disrupts BM structure in a cell-autonomous manner, and BM structure is restored by introducing one copy of a loss-of-function β-PS mutation (mysXG43/+; msn-RNAi). Bars are as indicated in the figure. (D) Removing one copy of β-PS also rescues the defect in follicle cell migration seen in msn-RNAi, whereas removing one copy of β-PS alone or overexpressing Msn (UAS-Msn) increases the migration rate. Graph shows migration rates of individual stage 6–7 egg chambers for each condition. Numbers indicate fraction of egg chambers that showed follicle cell migration. The data shown are from a single representative experiment out of two repeats. Each experimental condition involves egg chambers dissected from >10 female flies, imaged over multiple days.
During their migration, the follicle cells create fibril-like structures in the BM (Haigo and Bilder, 2011), a phenomenon best observed with a GFP protein trap of one of the type IV collagen subunits, encoded by the viking gene (Vkg-GFP, Fig. 3 C; Buszczak et al., 2007). Consistent with the migration defect, msn-RNAi causes a severe disruption in the formation of BM fibrils. However, an even more striking defect is seen in msn mosaic epithelia. Here the Vkg-GFP contacting mutant cells becomes tightly associated with basal cell edges. This phenotype can also be seen to a lesser extent in the msn-RNAi condition. This cell-autonomous deformation of the BM suggests that the additional integrins at the basal surface may participate in active focal adhesions that increase BM contact and reduce cell movement.
If increased integrin levels produce the msn migration defect, reducing integrins in the msn-RNAi background should restore cell motility. By depleting Msn with a weaker follicle cell driver, traffic jam-Gal4 (tj-Gal4), we found that 46% (6/13) of msn-RNAi epithelia now undergo very slow migration, providing a sensitized condition for phenotypic rescue (Fig. 3 D). We then introduced one copy of a null β-PS integrin mutation, myospheroidXG43 (mysXG43), into this background. Strikingly, 100% (13/13) of these epithelia are motile (Fig. 3 D). Reducing integrin levels in msn-RNAi epithelia also rescues follicle cell planar polarity and Vkg-GFP polarization (Figs. S2 B and 3 C), suggesting that increased integrin-based adhesion may produce these phenotypes as well.
We were surprised to find, however, that reducing integrin levels alone increases the average rate of follicle cell migration (Fig. 3 D; control, 0.76 ± 0.17 µm/min; mysXG43/+, 0.93 ± 0.19 µm/min). This observation is consistent with previous reports showing that changes in ECM adhesion strength can affect migration speed (Palecek et al., 1997; Gupton and Waterman-Storer, 2006), but it also raises the possibility that the rescue we observed of the msn-RNAi migration phenotype is simply an additive effect of combining faster migrating and slower migrating states. Motivated by this unexpected result, we overexpressed Msn in the follicle cells and found that this condition also increases epithelial migration rates (Fig. 3 D; 0.95 ± 0.20 µm/min). All together, our results support a model in which Msn decreases the integrin-based adhesion of the follicle cells to the BM.
To further investigate the mechanism by which Msn regulates cell motility, we used a fluorescent protein trap in the msn gene (Msn-YFP) to reveal this protein’s localization. During follicle cell migration, Msn-YFP shows a clear planar polarized distribution at the basal epithelial surface (Fig. 4 A). Previous reports showed that the follicle cells also extend filopodia from their basal surfaces in a planar polarized pattern (Gutzeit, 1991). Live imaging has revealed that these structures mark the leading edge of each migrating cell (unpublished data). Using mitotic recombination to create isolated clones of Msn-YFP cells, we found that the heightened Msn localization always occurs on the opposite side of the cell from the filopodia, showing that Msn is enriched at each cell’s trailing edge (Fig. 4 B). β-PS integrin shows a complementary localization pattern, being slightly enriched at the front of each migrating cell (Fig. 4 C). This polarization is lost in clones of msn cells (Fig. 4 C). Together, these data suggest that Msn may reduce integrin-based adhesion at the cell’s trailing edge.
Msn is enriched at the trailing edge of each migrating cell. (A) Msn-YFP shows a planar polarized distribution at the basal surface. (B) Follicle cell clones expressing Msn-YFP show that this protein is enriched at the back (blue arrowhead) of each migrating cell. Dots indicate filopodial protrusions at the front of the cell. (C) Basal views of control and msn mosaic epithelia stained with antibodies against β-PS. There is a slight enrichment of integrins at the leading edge of each follicle cell, which is disrupted in a clone of msn cells. All images are maximum intensity projections of the basal surface.
Msn is enriched at the trailing edge of each migrating cell. (A) Msn-YFP shows a planar polarized distribution at the basal surface. (B) Follicle cell clones expressing Msn-YFP show that this protein is enriched at the back (blue arrowhead) of each migrating cell. Dots indicate filopodial protrusions at the front of the cell. (C) Basal views of control and msn mosaic epithelia stained with antibodies against β-PS. There is a slight enrichment of integrins at the leading edge of each follicle cell, which is disrupted in a clone of msn cells. All images are maximum intensity projections of the basal surface.
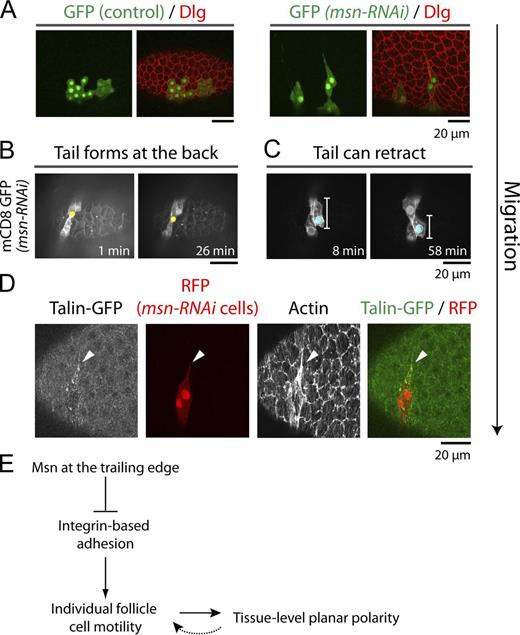
To test this idea, we returned to the small msn-RNAi clones that maintain the ability to migrate, albeit at a slower rate (Fig. 1 F). Interestingly, these cells are often elongated with tail-like extensions, a phenotype never observed in wild-type clones (Fig. 5 A). Live imaging confirmed that the tails correspond to the cell’s trailing edge (Fig. 5 B and Video 5), and that they can eventually retract as the cell moves forward (Fig. 5 C and Video 6). Interestingly, we have also observed prominent foci of the focal adhesion protein Talin/Rhea within the small msn-RNAi clones, which are often enriched in the tail (Fig. 5 D). These data support an increased requirement for Msn at the cell’s trailing edge.
“Tails” form at the back of individual msn-RNAi cells in a small clone (mCD8-GFP, Fig. 5 B). An egg chamber containing a clone of cells expressing UAS-msn-RNAi and UAS-mCD8-GFP was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Frames were taken every 30 s for 28 min. Playback rate is 300× real time.
“Tails” form at the back of individual msn-RNAi cells in a small clone (mCD8-GFP, Fig. 5 B). An egg chamber containing a clone of cells expressing UAS-msn-RNAi and UAS-mCD8-GFP was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Frames were taken every 30 s for 28 min. Playback rate is 300× real time.
“Tails” can retract as the msn-RNAi follicle cells migrate forward (mCD8-GFP, Fig. 5 C). An egg chamber containing a clone of cells expressing UAS-msn-RNAi and UAS-mCD8-GFP was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Frames were taken every 30 s for 60 min. Playback rate is 300× real time.
“Tails” can retract as the msn-RNAi follicle cells migrate forward (mCD8-GFP, Fig. 5 C). An egg chamber containing a clone of cells expressing UAS-msn-RNAi and UAS-mCD8-GFP was analyzed by time-lapse microscopy using a spinning disk confocal microscope (Axiovert 200M; Carl Zeiss) with a spinning disk unit (model CSU-10; Yokogawa Corporation of America). Frames were taken every 30 s for 60 min. Playback rate is 300× real time.
Msn appears to promote detachment of the cell’s trailing edge. (A) Control and msn-RNAi flipout clones (GFP) show that Msn depletion causes cell stretching, often forming a “tail” at one end. (B and C) Stills from two time-lapse movies of egg chambers with clones of cells expressing msn-RNAi and mCD8-GFP (Videos 5 and 6), which show that the “tails” form at the trailing edge of the migrating msn-RNAi cells (B), and can retract as the cells move forward (C). (D) Maximum projection of the basal surface in an egg chamber expressing Talin-GFP with msn-RNAi flipout clones (RFP). The migration direction for all panels is down (arrow). (E) Schematic for Msn’s role in follicle cell migration and planar polarity.
Msn appears to promote detachment of the cell’s trailing edge. (A) Control and msn-RNAi flipout clones (GFP) show that Msn depletion causes cell stretching, often forming a “tail” at one end. (B and C) Stills from two time-lapse movies of egg chambers with clones of cells expressing msn-RNAi and mCD8-GFP (Videos 5 and 6), which show that the “tails” form at the trailing edge of the migrating msn-RNAi cells (B), and can retract as the cells move forward (C). (D) Maximum projection of the basal surface in an egg chamber expressing Talin-GFP with msn-RNAi flipout clones (RFP). The migration direction for all panels is down (arrow). (E) Schematic for Msn’s role in follicle cell migration and planar polarity.
Here we have shown that Msn’s primary function during egg chamber elongation is to promote follicle cell motility. This finding is consistent with previous work establishing Msn and its homologues as pro-migratory kinases (Poinat et al., 2002; Collins et al., 2006; Chapman et al., 2008; Cobreros-Reguera et al., 2010; Teulière et al., 2011; Martynovsky et al., 2012). However, it is important to note that the mechanistic role that individual family members play in cell movement can vary with cell type. In the follicle cells, our data are consistent with a model in which Msn reduces integrin-based adhesion to the BM. Msn homologues in mice and Caenorhabditis elegans directly bind the β-integrin tail, suggesting that Msn could play a proximal role in focal adhesion turnover (Poinat et al., 2002). Msn’s enrichment at the basal follicle cell surface and its known association with the focal adhesion component Dreadlocks/NCK also support such a mechanism (Ruan et al., 1999; Buday et al., 2002). However, it is possible that Msn controls focal adhesion dynamics indirectly through modulation of the actin cytoskeleton (Fu et al., 1999; Ruan et al., 2002; Baumgartner et al., 2006; Köppen et al., 2006; Shakir et al., 2006). Our data further suggest a greater requirement for Msn at the cell’s trailing edge. This aspect of Msn’s function may also be conserved, as the C. elegans homologue MIG-15 limits cell-protrusive activity at the back of migrating growth cones (Teulière et al., 2011). Thus, we propose that Msn regulates follicle cell motility, at least in part, by inducing cell detachment at the trailing edge.
This work also sheds light on the mechanisms by which epithelial cells migrate at the tissue level. Consistent with in vitro studies (Palecek et al., 1997; Gupton and Waterman-Storer, 2006), we have shown that increasing or decreasing follicle cell–BM adhesion strength can alter epithelial migration rates in vivo. Importantly, our studies of msn mosaic epithelia demonstrate the robust nature of this tissue’s migratory behavior. Because Msn-deficient cells adhere more tightly to the BM, small clones cannot be carried along the migration path by their wild-type neighbors. Consequently, wild-type cells at the clone boundary modify cell–cell adhesion to either increase neighbor exchange or fully exclude the msn clone, thus preserving tissue-level migration and the morphogenetic program for egg chamber elongation.
We have previously shown that Msn is required for planar polarization of the follicle cell epithelium (Horne-Badovinac et al., 2012). Our current finding that Msn functions autonomously within each follicle cell to promote its motility now raises the intriguing possibility that tissue-level planar polarity may, in fact, arise from individual cell migratory behavior. Recent studies have shown that forces exerted by epithelial morphogenesis or by tension from neighboring tissues can align the Frizzled/Stabismus planar cell polarity system (Aigouy et al., 2010; Olguín et al., 2011; Lee et al., 2012). We propose that follicle cell planar polarity has a similar morphogenetic basis (Fig. 5 E). Our studies of msn mosaic epithelia show a positive correlation between tissue-level migration and planar polarity that is dependent on clone size. Similarly, loss of the atypical cadherin Fat2 from less than 50% of the follicle cells has no effect on planar polarity, whereas larger clones cause global disruptions (Viktorinová et al., 2009, 2011). We envision that this binary phenotype could also result from the success or failure of tissue migration. If enough wild-type cells are present to drive bulk epithelial movement, this would be sufficient to align the basal actin filaments and BM fibrils across the entire tissue. In this way, individual cell migratory behaviors can be translated into the tissue-level polarity required for organ morphogenesis.
Materials and methods
Drosophila genetics
Full genotypes for each experiment are in Table S1. Most crosses that involved clone generation were performed at 25°C, with the exception of RNAi experimental crosses that were raised at 18°C and shifted to higher temperatures as indicated in Table S2. All msn follicle cell clones were produced using FRT80B, and e22c-Gal4 to drive expression of the FLP recombinase in the follicle stem cells. bskDf(2L)flp147E clones were generated using FRT40A and the FLP recombinase under the control of a heat shock promoter. For flipout clones, UAS lines were crossed to flies containing the FLP recombinase under a heat shock promoter as well as the act5c>>Gal4, UAS-GFP (or UAS-RFP) flipout cassette. Most lines were obtained from the Bloomington Drosophila Stock Center (Bloomington, IN), with exceptions listed here. UAS-msn-RNAi (v101517 KK) is from the Vienna Drosophila RNAi Center (Vienna, Austria). tj-Gal4 (104055) and msn-YFP (115454) are from the Drosophila Genetic Resource Center (Kyoto Institute of Technology, Kyoto, Japan; Rees et al., 2011). vkg-GFP (CC00791) is from the Carnegie Protein Trap Library (Buszczak et al., 2007). UAS-HA-myr-msn is from Kaneko et al. (2011). UAS-msn is from Su et al. (2000). UAS-mCD8-GFP is from Pfeiffer et al. (2010). msnP23-5, FRT80/TM3 is from Horne-Badovinac et al. (2012). The genetic lesion in mysXG43, FRT101/FM6 is described in Jannuzi et al. (2002). Talin(rhea)-GFP/TM6b is from Venken et al. (2011). msn102 and msn172 are available from the Bloomington Drosophila Stock Center, but were first described in Treisman et al. (1997). w1118 served as a wild-type control. For experiments involving heat shock, females were placed on yeast in an EchoTherm programmable incubator (Torrey Pines Scientific), heat shocked twice daily for 2 d, and then dissected 2–3 d later. Each heat shock consisted of a 1-h period at 37°C, with a 1-h recovery at 25°C, followed by a second hour at 37°C.
Live imaging
A highly detailed protocol for egg chamber live imaging can be found in Prasad et al. (2007). For this study, female flies were fed yeast to increase egg production. Ovaries were then dissected from the females in live imaging media, which consists of Schneider’s Drosophila medium (Invitrogen) supplemented with 200 µg/ml insulin (Sigma-Aldrich), 15% (vol/vol) fetal bovine serum, and 0.6× streptomycin/penicillin (Invitrogen) at pH 6.95–7.0. Ovaries were removed from the female by using one set of forceps to submerge her in the media and the other set to pull on the posterior-most part of the abdomen, revealing the pair of ovaries. Individual ovarioles were then isolated from the muscle sheath by gently pulling on the germarium with forceps. Egg chambers were transferred into live imaging media with 0.4% NuSieve GTG low melt agarose (Lonza) and 6 µM FM4-64 (Invitrogen) to label cell membranes, and mounted in an aluminum slide (76 cm × 26 cm) containing a circular hole (12-cm diameter) with a coverslip attached to the bottom using melted parafilm. The agarose was allowed to solidify for ∼10 min, and then egg chambers were imaged with a 63×/1.4 NA oil-immersion objective on a microscope (Axiovert 200M; Carl Zeiss) equipped with a spinning disk unit (model CSU-10; Yokogawa Corporation of America) and illuminated with 50-mW 473-nm (GFP) and 25-mW 561-nm (mCherry) diode pump solid-state lasers. Images were acquired with an EM-CCD camera (Cascade 512B; Photometrics) controlled by MetaMorph software (Molecular Devices).
Immunohistochemistry and microscopy
Ovaries were dissected in S2 medium and fixed for 15 min in PBS + 0.1% Triton + 4% EM-grade formaldehyde (Polysciences). Antibody stains were performed in PBS + 0.1–0.3% Triton + 0.1% BSA. Alexa Fluor 488– or 555–conjugated secondary antibodies were used (1:200; Invitrogen). Rhodamine (1:200; Sigma-Aldrich) or Alexa Fluor 647 Phalloidin (1:50; Invitrogen) was used to label F-actin. Samples were mounted in SlowFade Antifade (Invitrogen) and imaged at room temperature using a 40×/1.3 NA EC Plan-NeoFluar oil objective on a laser-scanning confocal microscope (LSM510; Carl Zeiss) controlled by the LSM acquisition software. Maximum intensity projections were generated using ImageJ (National Institutes of Health) as indicated, and images were compiled using Photoshop CS4 (Adobe). Commercial antibodies include: anti-HA (1:200; rabbit; Rockland), and anti-Myc (1:200; mouse; Cell Signaling Technology). The following antibodies were obtained from the Developmental Studies Hybridoma Bank, developed under the auspices of the National Institute of Child Health and Human Development and maintained by The University of Iowa, Department of Biology (Iowa City, IA): Dlg (4F3; 1:10 supernatant), α-PS1 (DK.1A4; 1:50 concentrate), and β-PS (COMPARE6G11; 1:10 supernatant). Anti-laminin (rabbit, 1:50) was a generous gift from Lisa Fessler (UCLA, Los Angeles, CA).
Online supplemental material
Fig. S1 shows examples of the msn round egg phenotype and characterizes the effect of msn-RNAi on egg chamber elongation between stages 5 and 10. It also shows an example of msn mosaic egg chamber displaying the invasion phenotype, which has been stained for laminin. Fig. S2 shows that defects in JNK signaling do not affect integrin levels, and that reducing integrin levels rescues the defect in basal actin organization in msn-RNAi egg chambers. Supplemental videos show examples of egg chambers described in Fig. 1 C (Video 1), Fig. 1 E (Video 2), Fig. 1 F (Video 3), Fig. 2, D and E (Video 4), Fig. 5 B (Video 5), and Fig. 5 C (Video 6). Table S1 lists experimental genotypes used in each figure panel. Table S2 lists details for the temperatures at which the flies were maintained. Additional data are available in the JCB DataViewer at http://dx.doi.org/10.1083/jcb.201209129.dv.
Acknowledgments
Hugo Bellen, Lisa Fessler, Barret Pfeiffer, and Lan Xu generously provided reagents. We are also grateful to Julie Canman, Lisa Gallegos, Kirstin Sherrard, and members of the Horne-Badovinac laboratory for insightful comments on the manuscript; to Darcy McCoy for technical assistance; and to Nick Badovinac for illustrations.
Lindsay Lewellyn was supported by a Postdoctoral Fellowship (PF-12-135-01-CSM) from the American Cancer Society and an award from the American Heart Association. This work was further supported by grants from the Edward Mallinckrodt, Jr. Foundation and National Institutes of Health (R01 GM094276) to Sally Horne-Badovinac.