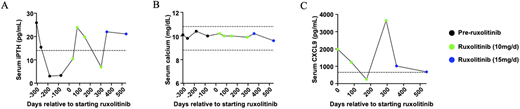
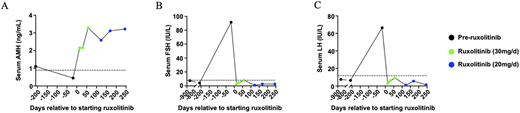
Autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy (APECED/APS-1) is a monogenic disorder of failed central tolerance caused by deleterious variants in the autoimmune regulator (AIRE) gene. Interferon-γ-driven inflammation in APECED underlies multiorgan autoimmunity and chronic mucocutaneous candidiasis and is targetable with JAK1/2 inhibition. Yet, whether early cytokine blockade can avert irreversible endocrine failure is currently unknown. Here, we describe two individuals with APECED who developed early autoimmune hypoparathyroidism and hypergonadotropic hypogonadism and in whom JAK1/2 inhibition halted progression and reversed biochemical and clinical abnormalities, preserving parathyroid and gonadal function. Specifically, patient 1 at the age of 2 years developed progressively declining intact parathyroid hormone (iPTH) levels that became undetectable within four months, while calcium and phosphorus levels remained normal, consistent with early, subclinical hypoparathyroidism. Ruxolitinib normalized iPTH levels and preserved calcium levels (Figure 1 A and B). Serum CXCL9 levels dropped and then increased, associated with a transient decline in iPTH levels; when the ruxolitinib dose was increased, iPTH increased, and CXCL9 dropped again and has remained normal after 18 months of treatment (Figure 1 C), suggesting that CXCL9 may be a potential biomarker of ruxolitinib efficacy in APECED. Ruxolitinib also remitted mycophenolate-refractory autoimmune hepatitis and recurrent oral candidiasis, which had occurred despite the absence of autoantibodies against IL-17A or IL-17F. Patient 2, at the age of 18 years, developed clinical and biochemical features of early hypergonadotropic hypogonadism. Screening labs showed declining anti-Müllerian hormone (AMH) and elevated follicle-stimulating hormone (FSH) and luteinizing hormone (LH). She reported severe hot flashes and oligomenorrhea with only three menses over five months, a reduction from her prior regular 28-day cycles. After initiation of ruxolitinib, hot flashes resolved while FSH, LH, and AMH normalized, and regular 28-day menstrual cycles were restored (Figure 2 A–C). Ruxolitinib caused no hematopoietic, hepatic, or renal toxicity in either patient. These findings provide, to our knowledge, the first clinical evidence that timely JAK–STAT pathway inhibition can intercept endocrine autoimmunity in APECED. More broadly, they advance a disease-interception paradigm in which early, pathway-directed cytokine blockade may alter the natural history of autoimmune endocrinopathies.
Ruxolitinib reverses early hypoparathyroidism in patient 1. Data points in black represent time points before ruxolitinib initiation, while those in green and blue represent time points while receiving ruxolitinib at the indicated dose. A) Serum iPTH over time relative to ruxolitinib initiation. The dashed horizontal line represents the lower limit of normal for this assay. B) Total calcium levels over time relative to ruxolitinib initiation. C) Serum CXCL9 levels over time relative to ruxolitinib initiation. The dashed horizontal line marks the upper limit of normal for this assay.
Ruxolitinib reverses early hypoparathyroidism in patient 1. Data points in black represent time points before ruxolitinib initiation, while those in green and blue represent time points while receiving ruxolitinib at the indicated dose. A) Serum iPTH over time relative to ruxolitinib initiation. The dashed horizontal line represents the lower limit of normal for this assay. B) Total calcium levels over time relative to ruxolitinib initiation. C) Serum CXCL9 levels over time relative to ruxolitinib initiation. The dashed horizontal line marks the upper limit of normal for this assay.
Ruxolitinib reverses premature ovarian failure in patient 2. Data points in black represent time points before ruxolitinib initiation, while those in green and blue represent time points while receiving ruxolitinib at the indicated dose. Serum AMH (A), FSH (B), and LH (C) levels over time relative to ruxolitinib initiation. The dashed lines represent the lower (A) or upper (B and C) limits of normal for these assays.
Ruxolitinib reverses premature ovarian failure in patient 2. Data points in black represent time points before ruxolitinib initiation, while those in green and blue represent time points while receiving ruxolitinib at the indicated dose. Serum AMH (A), FSH (B), and LH (C) levels over time relative to ruxolitinib initiation. The dashed lines represent the lower (A) or upper (B and C) limits of normal for these assays.