Injuries that heal by fibrosis can compromise organ function and increase patient morbidity. The oral mucosal barrier has a high regenerative capacity with minimal scarring, but the cellular mechanisms remain elusive. Here, we identify distinct postnatal paired-related homeobox-1+ (Prx1+) cells as a critical fibroblast subpopulation that expedites mucosal healing by facilitating early immune response. Using transplantation and genetic ablation model in mice, we show that oral mucosa enriched with Prx1+ cells heals faster than those that lack Prx1+ cells. Lineage tracing and scRNA-seq reveal that Prx1+ fibroblasts exhibit progenitor signatures in physiologic and injured conditions. Mechanistically, Prx1+ progenitors accelerate wound healing by differentiating into immunomodulatory SCA1+ fibroblasts, which prime macrophage recruitment through CCL2 as a key part of pro-wound healing response. Furthermore, human Prx1+ fibroblasts share similar gene and spatial profiles compared to their murine counterpart. Thus, our data suggest that Prx1+ fibroblasts may provide a valuable source in regenerative procedures for the treatment of corneal wounds and enteropathic fibrosis.
Introduction
Fibroblasts are ubiquitous stromal cells known for their canonical role in connective tissue wound healing by differentiating into contractile, matrisome-secretory myofibroblasts (Marshall et al., 2018; Chen et al., 2021). It is now recognized that fibroblasts are exceptionally plastic and heterogeneous within and across various organs, giving rise to specialized mesenchymal cell types in steady-state and perturbed conditions (Plikus et al., 2021; Lynch and Watt, 2018). In skin, papillary fibroblast progenitors differentiate into dermal papilla to support hair follicle regeneration (Phan et al., 2020; Zhang et al., 2009), whereas in arthritic synovial joints, fibroblasts take on an immuno-pathological phenotype to induce joint destruction (Croft et al., 2019; Mizoguchi et al., 2018). There may exist distinct fibroblast progenitors that promote wound healing responses by shifting the response from repair to regeneration (Buechler et al., 2021). However, there is limited in vivo evidence for this concept since most organs heal by scar formation in adult tissues.
Wound healing is accelerated at the oropharyngeal and gastroenteric mucosal interface (Thornton and Barbul, 1997), amongst which oral mucosal healing is best studied. Oral mucosa is subject to continuous mechanical insults from mastication, yet most injuries in the oral cavity heal by regeneration over repair (Wong et al., 2009; Griffin et al., 2021). Oral mucosal healing contrasts with cutaneous repair by shorter healing time, reduced inflammation, and minimal scar formation (Szpaderska et al., 2003; Mak et al., 2009), which closely mirrors fetal wound regeneration (Larson et al., 2010; Glim et al., 2014). Oral fibroblasts are largely derived from the neural crest in contrast to mesodermal origin of skin fibroblasts (Xu et al., 2013; Kou et al., 2018). Thus, they may be functionally distinct and possess stem cell–like properties to promote rapid and regenerative mucosal healing. However, most investigations rely on ex vivo expansion of oral fibroblasts in cultured conditions, which can alter fibroblast phenotype and mask true in vivo function and identity of these cells (Walmsley et al., 2015; Kim et al., 2018). In contrast to dermal wound repair, which is well-established (Driskell et al., 2013; Rinkevich et al., 2015; Leavitt et al., 2020), studies investigating distinct oral fibroblasts and their role in expedited wound regeneration in vivo are comparatively lacking.
Paired-related homeobox-1 gene (Prx1) expression is highly upregulated during embryonic limb development and for injury-responsive limb regeneration in salamanders (Logan et al., 2002; Gerber et al., 2018). In adult mice, abundant postnatal Prx1+ cells are found in regenerative organs such as axial bones, cranial sutures, and periodontal ligament, where they function as mesenchymal progenitors upon injury (Bassir et al., 2019; Wilk et al., 2017; Ko et al., 2019). Fibroblasts with postnatally active 2.4 kb Prx1 enhancer element (Prx1enh+) are found minimally at below 0.5% frequency and expand rapidly to differentiate into papillary fibroblasts and adipocytes upon injury in skin (Currie et al., 2019), but the mechanism by which they contribute to wound repair has not been explored. Moreover, it is not known whether these cells reside at the mucosal interface and contribute to expedited mucosal healing.
Recent studies of human oral mucosae by single-cell RNA sequencing (scRNA-seq) have unveiled a remarkable stromal transcriptomic heterogeneity (Williams et al., 2021; Caetano et al., 2021). To investigate how select oral fibroblast subsets contribute to expedited wound healing, we took advantage of regenerative oral wound healing model and identified genetically traceable Prx1enh+ fibroblast progenitors in masticatory mucosa, also known as gingiva. We show that these cells facilitate wound healing by differentiating into highly chemotactic SCA1+ (stem cell antigen-1)+ fibroblasts. Our study implicates priming of macrophage trafficking via SCA1+ fibroblast-derived C-C motif chemokine ligand 2 (CCL2) as a mechanism for rapid mucosal wound healing and uncovers a direct immunomodulatory role by specific oral fibroblasts for a prompt innate immune response and subsequent resolution of inflammation.
Results
Expedited wound healing in Prx1enh+ cell–enriched gingivae
We first tested the hypothesis that Prx1+ cells reside in the oral mucosa and that they may be responsible for rapid wound healing. Using Prx1CreERT:R26RtdTomato reporter mice that express Cre recombinase under the 2.4 kb Prx1 promoter activity (Ouyang et al., 2014), we found that postnatal Prx1+ cells were found exclusively in the lamina propria within anterior rugae of the hard palate (rugae 1–2, or R1–R2), whereas the posterior hard palate mucosa lacked Prx1+ cells in rugae 3–8 (R3–R8; Fig. 1, A and B). Immunofluorescence with an antibody against Prx1 protein showed significantly higher numbers of immunopositive Prx1+ cells in the R2 compared to R4 (Fig. S1 A). Approximately 93% of cells that express GFP driven by Prx1 promoter activity (Prx1CreERT-eGFP) faithfully expressed Prx1 protein; however, 24% of Prx1-immunopositive cells did not express GFP (Fig. S1 B). To distinguish Prx1-immunopositive cells and Prx1+ cells with active 2.4 kb promoter element, we hereafter refer to our target Prx1+ cells as Prx1enh+ cells. Prx1enh+ cells accounted for ∼10% of cell population, exhibited spindle-shape morphology, co-expressed fibroblast marker vimentin, and were seldom associated with CD31+ blood vessels (Fig. S1, C and D). Moreover, Prx1enh+ cells were also found in the buccal mucosa and intestinal stroma (Fig. S1 E), suggesting that these fibroblastic cells are generally found in mucosal tissues but exhibit differential spatial density as shown in the palatal gingiva.
Expedited wound healing in Prx1 + cell – enriched gingiva. (A) Left, macrophotograph of mouse palatal rugae numbered from anterior to posterior direction. Right, whole tissue imaging for red fluorescence in Prx1CreERT:R26RtdTomato mice that received tamoxifen injection for 5 d. Scale bar, 1 mm. N = 3 mice from two independent experiments. (B) Representative immunofluorescent images showing tdTomato+ signals in sagittal sections of R1–R3 (green box; magnified view is shown in inset, orange box) and R3–R6 (blue box). Animals were euthanized 2 wk after last tamoxifen injection. Scale bar, 250 μm. N = 3 mice from two independent experiments. (C) Macrophotograph of 1 mm palatal wounds created in Prx1+-enriched R2 or Prx1-deficient R4 after 2, 4, and 6 d of healing. Scale bar, 1 mm; M1, first molar tooth. Dashed line demarcates clinical wound outline. N = 7 mice for each time point, three independent experiments. (D) Masson trichrome-stained section images of R2 and R4 wounds. Scale bar, 0.5 mm. N = 7 mice for each time point, three independent experiments. (E and F) Quantification of open wound area percent (E) and epithelial gap closure distance (F) during wound healing. Each dot represents one wound location. Data represent mean ± SEM (N = 7 mice for each time point for a total of 21 mice, three independent experiments). Student’s t test; **P < 0.01, ***P < 0.001. (G) Quantification of new collagen formation by measuring blue-stained area in the wound bed from trichrome sections. Each dot represents one wound location. Data represent mean ± SEM (N = 7 mice for each time point for a total of 21 mice, three independent experiments). Student’s t test; *P < 0.05, ***P < 0.001. (H) Schematic diagram of gingival tissue engraftment from R2 to R4 to enrich Prx1+ cells or from R4 to R4 as control group in Prx1CreERT-eGFP mice. Representative immunofluorescent images validating incorporation of GFP+ cells. Scale bar, 0.5 mm. N = 3 mice each from two independent experiments. (I) Representative trichrome-stained images of day 2 wounds after R2 or R4 engraftment. N = 7–8 mice each from three independent experiments. (J and K) Quantification of granulation tissue (J) and new collagen formation areas (K) in wounds created after R2 or R4 autograft. Each dot represents one mouse, and data represent mean ± SEM; N = 7–8 mice per group for a total of 15 mice, three independent experiments. Student’s t test; **P < 0.01. (L) Macrophotograph of day 4 wounds in experimental Prx1CreERT:R26RDTA/+ (DTAPrx1) or control littermate mice (Prx1CreERT). Scale bar, 1 mm; dashed line demarcates clinical wound outline. N = 6–7 mice each from three independent experiments. (M and N) Representative images of COL3+ stained area within the lamina propria (demarcated with dashed line) in control and DTAPrx1 mice (M), and quantification of stromal healing by measuring COL3+ area in the wound bed at day 4 (N). Scale bar, 1 mm. Each dot represents one mouse, and data represent mean ± SEM; N = 6–7 mice per group for a total of 13 mice, three independent experiments. Student’s t-test; ***P < 0.001.
Expedited wound healing in Prx1 + cell – enriched gingiva. (A) Left, macrophotograph of mouse palatal rugae numbered from anterior to posterior direction. Right, whole tissue imaging for red fluorescence in Prx1CreERT:R26RtdTomato mice that received tamoxifen injection for 5 d. Scale bar, 1 mm. N = 3 mice from two independent experiments. (B) Representative immunofluorescent images showing tdTomato+ signals in sagittal sections of R1–R3 (green box; magnified view is shown in inset, orange box) and R3–R6 (blue box). Animals were euthanized 2 wk after last tamoxifen injection. Scale bar, 250 μm. N = 3 mice from two independent experiments. (C) Macrophotograph of 1 mm palatal wounds created in Prx1+-enriched R2 or Prx1-deficient R4 after 2, 4, and 6 d of healing. Scale bar, 1 mm; M1, first molar tooth. Dashed line demarcates clinical wound outline. N = 7 mice for each time point, three independent experiments. (D) Masson trichrome-stained section images of R2 and R4 wounds. Scale bar, 0.5 mm. N = 7 mice for each time point, three independent experiments. (E and F) Quantification of open wound area percent (E) and epithelial gap closure distance (F) during wound healing. Each dot represents one wound location. Data represent mean ± SEM (N = 7 mice for each time point for a total of 21 mice, three independent experiments). Student’s t test; **P < 0.01, ***P < 0.001. (G) Quantification of new collagen formation by measuring blue-stained area in the wound bed from trichrome sections. Each dot represents one wound location. Data represent mean ± SEM (N = 7 mice for each time point for a total of 21 mice, three independent experiments). Student’s t test; *P < 0.05, ***P < 0.001. (H) Schematic diagram of gingival tissue engraftment from R2 to R4 to enrich Prx1+ cells or from R4 to R4 as control group in Prx1CreERT-eGFP mice. Representative immunofluorescent images validating incorporation of GFP+ cells. Scale bar, 0.5 mm. N = 3 mice each from two independent experiments. (I) Representative trichrome-stained images of day 2 wounds after R2 or R4 engraftment. N = 7–8 mice each from three independent experiments. (J and K) Quantification of granulation tissue (J) and new collagen formation areas (K) in wounds created after R2 or R4 autograft. Each dot represents one mouse, and data represent mean ± SEM; N = 7–8 mice per group for a total of 15 mice, three independent experiments. Student’s t test; **P < 0.01. (L) Macrophotograph of day 4 wounds in experimental Prx1CreERT:R26RDTA/+ (DTAPrx1) or control littermate mice (Prx1CreERT). Scale bar, 1 mm; dashed line demarcates clinical wound outline. N = 6–7 mice each from three independent experiments. (M and N) Representative images of COL3+ stained area within the lamina propria (demarcated with dashed line) in control and DTAPrx1 mice (M), and quantification of stromal healing by measuring COL3+ area in the wound bed at day 4 (N). Scale bar, 1 mm. Each dot represents one mouse, and data represent mean ± SEM; N = 6–7 mice per group for a total of 13 mice, three independent experiments. Student’s t-test; ***P < 0.001.
Characterization of postnatal Prx1 enh+ cells and validation in DTA Prx1 mice. (A) Left, immunofluorescence experiments using PRRX1 antibody to detect immunopositive cells in R2 and R4 of wildtype C57BL/6 (B6) mice. Scale bar, 100 μm. Right, quantification of PRRX1+ cells in R2 and R4 of B6 mice. Each dot represents one wound location, and data represent mean ± SEM. N = 6 mice, three independent experiments. Student’s t test; ***P < 0.001. (B) PRRX1 immunopositive cells detected by immunofluorescence in R2 of Prx1CreERT-eGFP mice. Arrows point to double-positive PRRX1+GFP+ cells whereas arrowheads point to single PRRX1+ cells. Scale bar, 100 μm. Right, quantification of double PRRX1+GFP+, single PRRX1+, and single GFP+ cell percentage, normalized per total PRRX1+ cell count or per total GFP+ cell count. N = 3 mice, two independent experiments. (C) Left, graphical diagram of mouse palate from which anterior (R1–3) or posterior (R3–8) gingivae were collected for flow cytometry. Right, GFP+ cell percentage in single cell suspension prepared from anterior or posterior palate tissue. N = 3 mice, two independent experiments. (D) Left, paraffin-embedded sections from palatal gingiva corresponding to second rugae of Prx1CreERT:R26RtdTomato mice. Tissues were stained with RFP antibody and vimentin (green, top) or CD31 (green, bottom) antibody. Scale bar, 50 μm. Right, quantification of tdTomato+ cells that co-express vimentin or CD31. Each dot represents one mouse, and data represent mean ± SEM. N = 4 mice, two independent experiments. (E) Representative immunofluorescence images in buccal mucosa, jejunum, and colon from Prx1eGFP mice. Cryosections were stained with antibody against GFP to detect postnatal Prx1+ cells and anti-EpCam antibody to distinguish intestinal epithelium in jejunum and colon specimens. Dotted lines demarcate epithelial–lamina propria–muscularis propria borders in buccal mucosa. Scale bar, 100 μm. N = 3 mice, two independent experiments. (F–I) Immunofluorescence experiments validating Prx1+ fibroblast ablation in experimental Prx1CreERT:R26RDTA/+ (DTAPrx1) mice by examining unwounded palatal rugae. (F) Quantification of GFP+ Prx1+ cells in Prx1CreERT-GFP or Prx1CreERT-eGFP:R26RDTA that received tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test; ***P < 0.001. (G) Quantification of CD45+ leukocytes (red) in epithelial (K14+, green) or lamina propria compartments. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (H) Quantification of CD31+ endothelial cells (red) and the number of blood vessels. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (I) Quantification of αSMA+ pericytes (green). Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (J) Immunofluorescent signal for COL3 in littermate controls (Prx1CreERT) and experimental DTAPrx1 mice in day 4 wounds of the R4 area. Scale bar, 1 mm; dashed line marks COL3-immunopositive area in wound bed. N = 6 mice per group, three independent experiments. (K) Quantification of epithelial gap closure (left) and COL3+ area (right) in day 4 wounds. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test.
Characterization of postnatal Prx1 enh+ cells and validation in DTA Prx1 mice. (A) Left, immunofluorescence experiments using PRRX1 antibody to detect immunopositive cells in R2 and R4 of wildtype C57BL/6 (B6) mice. Scale bar, 100 μm. Right, quantification of PRRX1+ cells in R2 and R4 of B6 mice. Each dot represents one wound location, and data represent mean ± SEM. N = 6 mice, three independent experiments. Student’s t test; ***P < 0.001. (B) PRRX1 immunopositive cells detected by immunofluorescence in R2 of Prx1CreERT-eGFP mice. Arrows point to double-positive PRRX1+GFP+ cells whereas arrowheads point to single PRRX1+ cells. Scale bar, 100 μm. Right, quantification of double PRRX1+GFP+, single PRRX1+, and single GFP+ cell percentage, normalized per total PRRX1+ cell count or per total GFP+ cell count. N = 3 mice, two independent experiments. (C) Left, graphical diagram of mouse palate from which anterior (R1–3) or posterior (R3–8) gingivae were collected for flow cytometry. Right, GFP+ cell percentage in single cell suspension prepared from anterior or posterior palate tissue. N = 3 mice, two independent experiments. (D) Left, paraffin-embedded sections from palatal gingiva corresponding to second rugae of Prx1CreERT:R26RtdTomato mice. Tissues were stained with RFP antibody and vimentin (green, top) or CD31 (green, bottom) antibody. Scale bar, 50 μm. Right, quantification of tdTomato+ cells that co-express vimentin or CD31. Each dot represents one mouse, and data represent mean ± SEM. N = 4 mice, two independent experiments. (E) Representative immunofluorescence images in buccal mucosa, jejunum, and colon from Prx1eGFP mice. Cryosections were stained with antibody against GFP to detect postnatal Prx1+ cells and anti-EpCam antibody to distinguish intestinal epithelium in jejunum and colon specimens. Dotted lines demarcate epithelial–lamina propria–muscularis propria borders in buccal mucosa. Scale bar, 100 μm. N = 3 mice, two independent experiments. (F–I) Immunofluorescence experiments validating Prx1+ fibroblast ablation in experimental Prx1CreERT:R26RDTA/+ (DTAPrx1) mice by examining unwounded palatal rugae. (F) Quantification of GFP+ Prx1+ cells in Prx1CreERT-GFP or Prx1CreERT-eGFP:R26RDTA that received tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test; ***P < 0.001. (G) Quantification of CD45+ leukocytes (red) in epithelial (K14+, green) or lamina propria compartments. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (H) Quantification of CD31+ endothelial cells (red) and the number of blood vessels. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (I) Quantification of αSMA+ pericytes (green). Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test. (J) Immunofluorescent signal for COL3 in littermate controls (Prx1CreERT) and experimental DTAPrx1 mice in day 4 wounds of the R4 area. Scale bar, 1 mm; dashed line marks COL3-immunopositive area in wound bed. N = 6 mice per group, three independent experiments. (K) Quantification of epithelial gap closure (left) and COL3+ area (right) in day 4 wounds. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test.
We next investigated the functional significance of Prx1enh+ fibroblasts for gingival injury response by contrasting wound healing in Prx1enh+-enriched gingiva (rugae 2, R2) and Prx1-deficient gingiva (rugae 4, R4). 1-mm wounds created in the Prx1enh+-enriched R2 site exhibited complete re-epithelialization and wound gap closure by day 4, whereas healing in the Prx1-deficient R4 site was significantly delayed and did not fully close until day 6 after wounding (Fig. 1, C–F). Moreover, there was a dramatic difference in new collagen formation between R2 and R4 wounds through 6 d after injury (Fig. 1 G). To rule out differential healing response due to anatomical location and masticatory stresses (Byrd et al., 2019), we transplanted Prx1enh+-enriched R2 gingiva to the Prx1-deficient R4 site and examined wound healing. This approach successfully incorporated GFP+Prx1enh+ fibroblasts transplanted from the R2 to the R4 area, whereas control autografts did not (Fig. 1 H). After wounding and healing for 2 d, the recipient site with transplanted Prx1enh+ fibroblasts exhibited significantly increased new collagen formation with lower granulation tissue formation compared to control sites with transplanted Prx1-poor tissue (P < 0.01; Fig. 1, I–K). To further determine whether Prx1enh+ fibroblasts are necessary for proper stromal healing in mucosal wounds, we conditionally ablated postnatal Prx1enh+ cells by tamoxifen-induced diphtheria toxin expression in Prx1CreERT:R26RDTA mice (DTAPrx1) and examined R2 wounds. This approach effectively reduced GFP+ Prx1enh+ fibroblasts in the mucosa without affecting the number of pericytes, endothelial cells, or leukocytes (Fig. S1, F–I). We found that induced Prx1enh+ fibroblast ablation in adult mice led to a drastic reduction in type 3 collagen (COL3) expression in the R2 wound bed (Fig. 1, L–N), demonstrating the critical role that these cells play in stromal healing despite the general presence of other fibroblast types. As expected, ablation of Prx1enh+ cells had no effect on epithelial gap closure or COL3+ area in the Prx1-deficient R4 wounds (Fig. S1, J and K). Together, the gain-of-function by transplantation and loss-of-function by cell ablation experiments clearly demonstrate that Prx1enh+ fibroblasts are critical for robust oral mucosal wound healing.
Prx1+ cells are functionally distinct from other oral fibroblast clusters
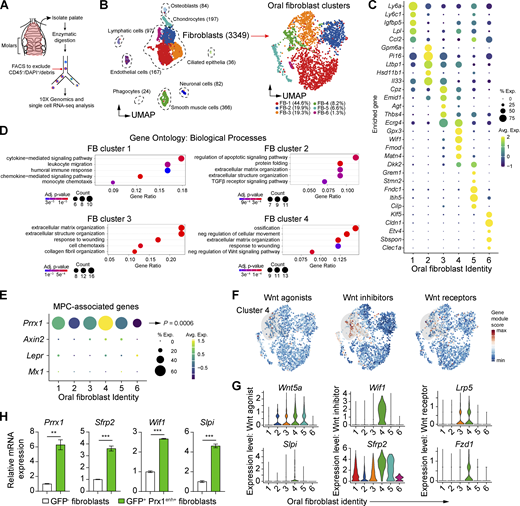
To examine oral fibroblast subtypes and how Prx1enh+ cells differ from them, we performed scRNA-seq analysis of sorted CD45− non-hematopoietic cells isolated from the mouse palate (Fig. 2 A). After quality assessment, filtering, and cell type assignment (Fig. S2, A–C), we focused on fibroblasts that expressed common mesenchymal markers such as Col3a1 and Dcn (Fig. 2 B). This approach identified six fibroblast clusters that had distinct upregulated gene signatures, in which the two largest oral fibroblast clusters were enriched with Ly6a (encoding SCA1 protein) and Pi16 transcripts, respectively (Fig. 2, B and C; and Table S1). To assess biological significance of these fibroblast clusters, we performed gene ontology (GO) analysis using differentially upregulated gene sets from each fibroblast group. We found cluster 1 to be implicated in chemotaxis and immune response, cluster 2 in TGFβ receptor signaling pathway, cluster 3 to in extracellular matrix and collagen fibril organization, and cluster 4 in ossification, wound healing, and Wnt (Wingless and Int-1) signaling pathway (Fig. 2 D and Table S2).
scRNA-seq analysis of oral fibroblasts and Prx1+cells in steady state. (A) Experimental diagram for scRNA-seq analysis of oral mucosa derived from the hard palate of C57BL/6 mice. (B) Left, UMAP plot of CD45− gingival cells isolated from 8-wk-old mice. Right, UMAP plot of oral fibroblast clusters, selected based on putative extracellular matrix gene expression. (C) Dot plot showing enriched expression of top five genes from each six oral fibroblast clusters. (D) GO analysis for fibroblast clusters 1–4, truncated to five biological processes. Full list is provided in Table S1. (E) Dot plot showing enrichment of mesenchymal progenitor cell–associated genes Prrx1, Mx1, Lepr, and Axin2 in oral fibroblast clusters. (F) Gene module scores for Wnt activator, inhibitor, and receptor genes. (G) Violin plots of Wnt-associated genes significantly upregulated in cluster 4, as determined by Wilcoxon rank-sum analysis. (H) Real-time qPCR of cluster 4–specific genes (Prrx1, Sfrp2, Wif1, Slpi) comparing GFP+ Prx1+ fibroblasts versus GFP− fibroblasts sorted from the anterior palate of Prx1eGFP mice. Data represent mean ± SEM. Fold changes in mRNA expression (N = 3 mice) from three independent experiments are shown. Student’s t test; **P < 0.01, ***P < 0.001.
scRNA-seq analysis of oral fibroblasts and Prx1+cells in steady state. (A) Experimental diagram for scRNA-seq analysis of oral mucosa derived from the hard palate of C57BL/6 mice. (B) Left, UMAP plot of CD45− gingival cells isolated from 8-wk-old mice. Right, UMAP plot of oral fibroblast clusters, selected based on putative extracellular matrix gene expression. (C) Dot plot showing enriched expression of top five genes from each six oral fibroblast clusters. (D) GO analysis for fibroblast clusters 1–4, truncated to five biological processes. Full list is provided in Table S1. (E) Dot plot showing enrichment of mesenchymal progenitor cell–associated genes Prrx1, Mx1, Lepr, and Axin2 in oral fibroblast clusters. (F) Gene module scores for Wnt activator, inhibitor, and receptor genes. (G) Violin plots of Wnt-associated genes significantly upregulated in cluster 4, as determined by Wilcoxon rank-sum analysis. (H) Real-time qPCR of cluster 4–specific genes (Prrx1, Sfrp2, Wif1, Slpi) comparing GFP+ Prx1+ fibroblasts versus GFP− fibroblasts sorted from the anterior palate of Prx1eGFP mice. Data represent mean ± SEM. Fold changes in mRNA expression (N = 3 mice) from three independent experiments are shown. Student’s t test; **P < 0.01, ***P < 0.001.
scRNA-seq analysis of murine CD45 − gingival cells. (A) Left, schematic diagram for scRNA-seq data quality control measurements. Right, mitochondrial percent, number of features, and UMI counts to exclude cell counts with outlier values (>threefold from median value). (B) Heatmap of differentially expressed genes from each cluster. Upregulated genes are indicated on the y axis. (C) Violin plots for genes known to be putatively expressed for specific cell type (Col3a1, Dcn: fibroblasts, Acta2: smooth muscle/pericytes, Acan: chondrocytes, Pecam1: endothelium, Lyve1: lymphatics, Bglap2: osteoblasts, Mpz: neuronal, Sntn: ciliated epithelia, H2-Ab1: phagocytic). (D) Immunophenotyping of lineage-negative (Lin−: CD45−CD31−Epcam−Ter119−) Prx1+ cells for the expression of pan-fibroblast marker (PDGFRA+) and mesenchymal progenitor markers (CD90, CD73, and CD105). Isotype IgG was used as negative control. Right, percent positive for each tested marker within GFP+ cell population. Each dot represents one mouse, N = 4 mice from two independent experiments are shown.
scRNA-seq analysis of murine CD45 − gingival cells. (A) Left, schematic diagram for scRNA-seq data quality control measurements. Right, mitochondrial percent, number of features, and UMI counts to exclude cell counts with outlier values (>threefold from median value). (B) Heatmap of differentially expressed genes from each cluster. Upregulated genes are indicated on the y axis. (C) Violin plots for genes known to be putatively expressed for specific cell type (Col3a1, Dcn: fibroblasts, Acta2: smooth muscle/pericytes, Acan: chondrocytes, Pecam1: endothelium, Lyve1: lymphatics, Bglap2: osteoblasts, Mpz: neuronal, Sntn: ciliated epithelia, H2-Ab1: phagocytic). (D) Immunophenotyping of lineage-negative (Lin−: CD45−CD31−Epcam−Ter119−) Prx1+ cells for the expression of pan-fibroblast marker (PDGFRA+) and mesenchymal progenitor markers (CD90, CD73, and CD105). Isotype IgG was used as negative control. Right, percent positive for each tested marker within GFP+ cell population. Each dot represents one mouse, N = 4 mice from two independent experiments are shown.
To characterize the Prx1enh+ fibroblast cluster, we further queried the scRNA-seq dataset and focused on Prrx1 expression and other mesenchymal progenitor cell markers that share similar functional profiles with Prx1enh+ cells in skeletal niche such as Mx1, Axin2, and Lepr (Zhou et al., 2014; Ortinau et al., 2019; Di Pietro et al., 2020; Ouyang et al., 2014). We found Prrx1 to be most significantly expressed in fibroblast cluster 4 (P = 0.0006), whereas Mx1, Axin2, and Lepr expression was minimal (Fig. 2 E). Cluster 4 was also enriched in Wnt inhibitor genes (Fig. 2 F), consistent with GO analysis results. Specifically, cluster 4 had upregulated gene expression of Wnt agonists such as Wnt5a and Slpi ([Morimoto et al., 2021]), Wnt inhibitors such as Wnt-inhibitory factor 1 (Wif1) and secreted frizzled-related protein 2 (Sfrp2), and Wnt receptor genes, Frizzled-1 (Fzd1) and Lrp5 (Fig. 2 G). We next confirmed the expression of selected genes in cluster 4 by real-time quantitative PCR (qPCR) using sorted Prx1enh+ cells that express GFP+. We found that Prrx1, Sfrp2, Wif1, and Slpi were upregulated in GFP+ Prx1enh+ cells when compared to GFP− oral fibroblasts (Fig. 2 H), demonstrating that postnatal Prx1enh+ cells in vivo correspond to Prrx1high cluster 4 group from our scRNA-seq data. These results implicate Prx1enh+ cells as a distinct fibroblast group with a cellular function closely associated with inhibition of the Wnt pathway.
Prx1+ cells function as fibroblast progenitors during homeostasis and injury response
We hypothesized that Prx1enh+ cells may function as fibroblast progenitors in the oral mucosa for the following reasons: (1) Prx1enh+ fibroblast-enriched genes such as Sfrp2 and Wif1 are highly upregulated in mesenchymal precursors (Ambrosi et al., 2021; de Castro et al., 2021; Tikhonova et al., 2019); (2) inhibition of Wnt signaling is critical for progenitor cell differentiation (Sharon et al., 2019; Mirotsou et al., 2007); and (3) postnatal Prx1enh+ cells give rise to multiple mesenchymal lineages during bone regeneration (Ouyang et al., 2014). To first infer lineage progression of Prx1enh+ fibroblasts, we performed monocle-based trajectory analysis (Cao et al., 2019) by setting seed in Prrx1high cluster (Fig. 3 A). We found that Prrx1 expression gradually decreased along pseudotime towards Ly6a+, Pi16+, or Acan+ enrichment in three major trajectories identified (Fig. 3 B), suggesting that Prx1enh+ progenitors differentiate along multiple fibroblast lineages. To determine whether mucosal Prx1enh+ fibroblasts exhibit self-renewal properties in vivo, a key feature of progenitors, we generated adult Prx1CreERT-eGFP:R26RtdTomato mice and traced Prx1-lineage cells in adult mice for 1, 4, and 16 wk. This approach labeled postnatal Prx1enh+ expressing cells with double-positive GFP+tdTomato+ and Prx1-derived lineage cells with single tdTomato+ (i.e., differentiated cells that lost Prx1enh+ expression; Fig. 3 C). We found that Prx1-lineage cells emerged by the 4th and 16th wk, but not at the 1st wk (Fig. 3 D). The ratio of tdTomato+ to GFP+ cells increased by ≥twofold over this tracing period without reducing the number of GFP+ cells (Fig. 3, E and F). This indicated that the Prx1enh+ fibroblasts population is expanding and replenishing its lineage cells, as the percentage of GFP+ cells actively expressing Prx1 is stable over 16 wk. To next characterize the fibroblast identity of Prx1-lineage cells during homeostasis, we traced Prx1enh+ cells for 4 wk and performed flow cytometry to specifically examine single tdTomato+ cells (Fig. 3 G). We found that Prx1-lineage cells formed distinct cell populations including CD146+ pericytes and SCA1+ fibroblasts, with ∼75% of tdTomato+ cells consisting of SCA1+ fibroblasts (Fig. 3, G and H). Additional phenotyping analysis in adult Prx1eGFP mice showed that Prx1enh+ cells expressed pan-fibroblast marker PDGFRA and common mesenchymal progenitor markers such as CD105, CD90, and CD73 (Pittenger et al., 2019; Fig. S2 D). Together, our data support progenitor identity of Prx1enh+ fibroblasts, which may have significant implications for improved oral wound healing.
Prx1 + fibroblasts function as fibroblast progenitors during homeostasis. (A) Pseudotime analysis of three major trajectories with Prrx1high cluster seeded as starting root. (B)Ly6a, Pi16, or Acan expression paired with Prrx1 expression along calculated pseudotime of each trajectory. (C) Experimental scheme for in vivo lineage tracing of postnatal Prx1+ cells. (D) Immunofluorescent images in Prx1CreERT-eGFP:R26RtdTomato mice chased for 1, 4, and 16 wk after last tamoxifen (TAM) injection. Scale bar, 250 μm; inset scale bar, 50 μm. N = 5 mice per each endpoint for a total of 15 mice, three independent experiments per endpoint. (E) Quantification of tdTomato+ to GFP+ cell number ratio. Each dot represents one mouse, and data represent mean ± SEM; N = 5 mice per each end point for a total of 15 mice, three independent experiments. One-way ANOVA and Tukey’s post-hoc test; **P < 0.01, ***P < 0.001. (F) GFP+ cell number normalized to total nucleated cells in mice traced for 1, 4, and 16 wk after tamoxifen. Each dot represents one mouse, and data represent mean ± SEM; N = 5 mice per each end point for a total of 15 mice, three independent experiments. One-way ANOVA and Tukey’s post-hoc test. (G) Flow cytometry analysis of Prx1-lineage cells from Prx1CreERT:R26RtdTomato mice traced for 4 wk. N = 6 mice, three independent experiments. (H) Pie chart quantification of Prx1-lineage cell identity by the expression of SCA1, PDGFRA, and/or CD146. Average percent of tdTomato+ numbers ± SEM is shown from N = 6, three independent experiments.
Prx1 + fibroblasts function as fibroblast progenitors during homeostasis. (A) Pseudotime analysis of three major trajectories with Prrx1high cluster seeded as starting root. (B)Ly6a, Pi16, or Acan expression paired with Prrx1 expression along calculated pseudotime of each trajectory. (C) Experimental scheme for in vivo lineage tracing of postnatal Prx1+ cells. (D) Immunofluorescent images in Prx1CreERT-eGFP:R26RtdTomato mice chased for 1, 4, and 16 wk after last tamoxifen (TAM) injection. Scale bar, 250 μm; inset scale bar, 50 μm. N = 5 mice per each endpoint for a total of 15 mice, three independent experiments per endpoint. (E) Quantification of tdTomato+ to GFP+ cell number ratio. Each dot represents one mouse, and data represent mean ± SEM; N = 5 mice per each end point for a total of 15 mice, three independent experiments. One-way ANOVA and Tukey’s post-hoc test; **P < 0.01, ***P < 0.001. (F) GFP+ cell number normalized to total nucleated cells in mice traced for 1, 4, and 16 wk after tamoxifen. Each dot represents one mouse, and data represent mean ± SEM; N = 5 mice per each end point for a total of 15 mice, three independent experiments. One-way ANOVA and Tukey’s post-hoc test. (G) Flow cytometry analysis of Prx1-lineage cells from Prx1CreERT:R26RtdTomato mice traced for 4 wk. N = 6 mice, three independent experiments. (H) Pie chart quantification of Prx1-lineage cell identity by the expression of SCA1, PDGFRA, and/or CD146. Average percent of tdTomato+ numbers ± SEM is shown from N = 6, three independent experiments.
We next examined Prx1enh+ progenitors in wounded conditions that could explain expedited healing in Prx1enh+-enriched gingiva. We first assessed proliferation of Prx1enh+ cells by examining Ki67+ expression in healing oral wounds and found that Prx1enh+ fibroblasts were significantly more proliferative than Prx1− fibroblasts (P < 0.05; Fig. 4 A). We further carried out in vivo EdU incorporation assay and observed abundant Prx1-lineage cells at the R2 wound edge expressing EdU, indicating active DNA synthesis (Fig. 4 B). Oral wounds that were created in R3 and R4 had little to no tdTomato+ cells, suggesting minimal migration of Prx1-lineage cells from R2 (Fig. 4 C). To next determine the cell fate of Prx1enh+ fibroblasts during mucosal healing, we examined single tdTomato+ Prx1-lineage cell populations from day 4 wounds and found that nearly 80% of the Prx1-derived cells expressed SCA1+ (Fig. 4 D). Immunofluorescence experiments confirmed that most of Prx1-lineage cells expressing tdTomato were also SCA1+ (Fig. 4 E). Notably, Prx1-lineage cells minimally expressed α-smooth muscle actin (αSMA; Fig. 4 F), suggesting other oral fibroblast populations as a major source for myofibroblast differentiation in oral wounds. In autograft experiments described above (Fig. 1 H), transplantation of Prx1enh+ cell–enriched tissue resulted in increased pan-fibroblast and SCA1+ fibroblast numbers in healing wounds compared to Prx1-deficient autograft wounds (Fig. 4, G and H). In contrast, the transplantation of Prx1enh+ cell–enriched tissue did not increase PI16+ fibroblast and pericyte numbers. Therefore, Prx1enh+ progenitors divide rapidly upon injury and differentiate mostly into SCA1+ fibroblasts in oral wounds.
Preferential differentiation of Prx1 + progenitors toward SCA1 + oral fibroblasts during wound healing. (A) Flow cytometry analysis and quantification of GFP+ Prx1+ cells expressing proliferation marker Ki67 compared to GFP− PDGFRA+ pan-fibroblasts. Each dot represents pooled cells from two mice, and data represent mean ± SEM; N = 4 data points from a total of eight mice, two independent experiments. Student’s t test; *P < 0.05. (B) Representative images for Prx1-lineage cells (red) and EdU incorporation (green) in day 4 wound from Prx1CreERT:R26RtdTomato mice. N = 3 mice from two independent experiments. Scale bar, 1 mm. Inset, arrows point to some EdU+tdTomato+ cells, dashed line demarcates epithelia–lamina propria border. (C) Quantification of tdTomato+ cells per mm2 in day 4 wounds created in R2, R3, or R4 from Prx1CreERT:R26RtdTomato mice. Data represent mean ± SEM; N = 3 mice from two independent experiments. (D) Flow cytometry analysis of Prx1-derived lineage cells 4 d after wounding. Right, quantification of Prx1-lineage cell identity during wound healing by the expression of SCA1, PI16, PDGFRA, and/or CD146. FB, fibroblast; neg, negative. Each dot represents one mouse, and data represent mean ± SEM; N = 4 mice from two independent experiments. (E) Representative immunofluorescent images of day 4 wound bed showing SCA1+tdTomato+ fibroblasts (white arrowheads) and some SCA1−tdTomato+ cells (orange arrowheads). Scale bar, 50 μm; N = 3 mice from two independent experiments. (F) Flow cytometry analysis of myofibroblasts (lineage−αSMA+PDGFRA+) in day 4 wounds. Right, quantification of percent myofibroblasts that are tdTomato+ or tdTomato−. Each dot represents one mouse, and data represent mean ± SEM; N = 8 mice from three independent experiments. Student’s t test; ***P < 0.001. (G) Representative immunofluorescent images of day 2 wounds after R2 or R4 graft incorporation, stained with SCA1 and vimentin antibodies. Arrows point to initial wound edges. Scale bar, 0.5 mm. Inset, arrowheads point to SCA1+vimentin+ cells. N = 5–6 each from two independent experiments. (H) Quantification of cell numbers per mm2 for pan-fibroblasts (pan-FB; vimentin+ spindle-shaped cells), SCA1+ FB (SCA1+vimentin+), PI16+ (PI16+vimentin+), and pericytes (CD146+CD31−). Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11 mice from two independent experiments. Student’s t test; *P < 0.05, **P < 0.01.
Preferential differentiation of Prx1 + progenitors toward SCA1 + oral fibroblasts during wound healing. (A) Flow cytometry analysis and quantification of GFP+ Prx1+ cells expressing proliferation marker Ki67 compared to GFP− PDGFRA+ pan-fibroblasts. Each dot represents pooled cells from two mice, and data represent mean ± SEM; N = 4 data points from a total of eight mice, two independent experiments. Student’s t test; *P < 0.05. (B) Representative images for Prx1-lineage cells (red) and EdU incorporation (green) in day 4 wound from Prx1CreERT:R26RtdTomato mice. N = 3 mice from two independent experiments. Scale bar, 1 mm. Inset, arrows point to some EdU+tdTomato+ cells, dashed line demarcates epithelia–lamina propria border. (C) Quantification of tdTomato+ cells per mm2 in day 4 wounds created in R2, R3, or R4 from Prx1CreERT:R26RtdTomato mice. Data represent mean ± SEM; N = 3 mice from two independent experiments. (D) Flow cytometry analysis of Prx1-derived lineage cells 4 d after wounding. Right, quantification of Prx1-lineage cell identity during wound healing by the expression of SCA1, PI16, PDGFRA, and/or CD146. FB, fibroblast; neg, negative. Each dot represents one mouse, and data represent mean ± SEM; N = 4 mice from two independent experiments. (E) Representative immunofluorescent images of day 4 wound bed showing SCA1+tdTomato+ fibroblasts (white arrowheads) and some SCA1−tdTomato+ cells (orange arrowheads). Scale bar, 50 μm; N = 3 mice from two independent experiments. (F) Flow cytometry analysis of myofibroblasts (lineage−αSMA+PDGFRA+) in day 4 wounds. Right, quantification of percent myofibroblasts that are tdTomato+ or tdTomato−. Each dot represents one mouse, and data represent mean ± SEM; N = 8 mice from three independent experiments. Student’s t test; ***P < 0.001. (G) Representative immunofluorescent images of day 2 wounds after R2 or R4 graft incorporation, stained with SCA1 and vimentin antibodies. Arrows point to initial wound edges. Scale bar, 0.5 mm. Inset, arrowheads point to SCA1+vimentin+ cells. N = 5–6 each from two independent experiments. (H) Quantification of cell numbers per mm2 for pan-fibroblasts (pan-FB; vimentin+ spindle-shaped cells), SCA1+ FB (SCA1+vimentin+), PI16+ (PI16+vimentin+), and pericytes (CD146+CD31−). Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11 mice from two independent experiments. Student’s t test; *P < 0.05, **P < 0.01.
Identification of highly chemotactic Ly6a/SCA1+ oral fibroblasts
Next, we evaluated a potential impact of Prx1-derived SCA1+ on expedited oral wound healing. We first examined the single-cell gene expression profile of Ly6a/SCA1+ cluster and found that this cluster had increased expression of key chemotaxis genes such as Ccl2, Cxcl1, and Ccl7, suggesting a role involving leukocyte recruitment (Fig. 5 A). When unwounded gingiva was examined, SCA1+ expression was largely localized to the submucosal compartment, indicating their submucosal fibroblast identity (Fig. S3, A–C). This was distinct from the reticular layer that was populated by PI16+ fibroblasts (Fig. S3, D and E). In line with our scRNA-seq analysis, in vivo validation with CCL2-mCherry reporter mice demonstrated intense CCL2 expression in the gingival submucosa where SCA1+ oral fibroblasts reside (Fig. S3 F). Thus, SCA1+ oral fibroblasts highly express chemokines during steady state, which may be important for priming a rapid healing response.
Identification of highly chemotactic Ly6a/SCA1 + oral fibroblasts. (A) Single-cell expression of Ly6a, Ccl2, Ccl7, and Cxcl1 in oral fibroblast subsets of the mouse palate. (B) Flow cytometry analysis of CCL2 expression in SCA1− and SCA1+ fibroblasts (CD31−CD45−Epcam−Ter119−PDGFRA+) from unwounded and day 2 wounds in C57BL/6 mice. Right, quantification of fibroblast (FB) percent positive for CCL2. Each dot represents one mouse, and data represent mean ± SEM. N = 5 mice per endpoint for a total of 10 mice from three independent experiments. Two-way ANOVA and Tukey’s post-hoc test; ***P < 0.001. (C) Pie chart of cell percentages that express CCL2. Average percent ± SEM is shown from N = 5 mice for each endpoint, a total of 10 mice from three independent experiments. (D) Flow cytometry plot gated for live CD45+CD11b+ cells to quantify F4/80+ (macrophages) and Ly6g+ (neutrophils) cells in R2 and R4 wounds 2 d after wounding. N = 6–8 mice per endpoint from three independent experiments for each endpoint. (E and F) Quantification of neutrophil (E) and macrophage (F) infiltration to the R2 and R4 wounds during wound healing. Each dot represents one wound location. Data represent mean ± SEM (N = 6–8 mice for each time point for a total of 20 mice, three independent experiments). Student’s t test; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. (G–J) Representative flow cytometry plots (G and I) and quantification of pro-inflammatory macrophages co-expressing IL1β (H) and pro-resolving macrophages co-expressing TGFβ1 (J) in healing R2 and R4 wounds. Each dot represents pooled cells from the designated wound location of one mouse. Data represent mean ± SEM (N = 6–8 mice for each time point for a total of 20 mice, three independent experiments). Student’s t test; *P < 0.05, ***P < 0.001.
Identification of highly chemotactic Ly6a/SCA1 + oral fibroblasts. (A) Single-cell expression of Ly6a, Ccl2, Ccl7, and Cxcl1 in oral fibroblast subsets of the mouse palate. (B) Flow cytometry analysis of CCL2 expression in SCA1− and SCA1+ fibroblasts (CD31−CD45−Epcam−Ter119−PDGFRA+) from unwounded and day 2 wounds in C57BL/6 mice. Right, quantification of fibroblast (FB) percent positive for CCL2. Each dot represents one mouse, and data represent mean ± SEM. N = 5 mice per endpoint for a total of 10 mice from three independent experiments. Two-way ANOVA and Tukey’s post-hoc test; ***P < 0.001. (C) Pie chart of cell percentages that express CCL2. Average percent ± SEM is shown from N = 5 mice for each endpoint, a total of 10 mice from three independent experiments. (D) Flow cytometry plot gated for live CD45+CD11b+ cells to quantify F4/80+ (macrophages) and Ly6g+ (neutrophils) cells in R2 and R4 wounds 2 d after wounding. N = 6–8 mice per endpoint from three independent experiments for each endpoint. (E and F) Quantification of neutrophil (E) and macrophage (F) infiltration to the R2 and R4 wounds during wound healing. Each dot represents one wound location. Data represent mean ± SEM (N = 6–8 mice for each time point for a total of 20 mice, three independent experiments). Student’s t test; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. (G–J) Representative flow cytometry plots (G and I) and quantification of pro-inflammatory macrophages co-expressing IL1β (H) and pro-resolving macrophages co-expressing TGFβ1 (J) in healing R2 and R4 wounds. Each dot represents pooled cells from the designated wound location of one mouse. Data represent mean ± SEM (N = 6–8 mice for each time point for a total of 20 mice, three independent experiments). Student’s t test; *P < 0.05, ***P < 0.001.
Validation of oral fibroblast heterogeneity in mouse palate. (A) H&E staining of mouse palatal rugae. (B) Immunofluorescence experiment with SCA1 (red) and PDGFRA (green) antibodies. N = 3 mice, two independent experiments. Scale bar, 100 μm; dashed lines demarcate borders between epithelium, papillary and reticular lamina propria, and submucosa. (C) Quantification of SCA1 mean fluorescent intensity (MFI) in the papillary, reticular, and submucosal layers. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. One-way ANOVA and Tukey’s post-hoc test; **P < 0.01. (D) Immunofluorescence experiment with PI16 (red) and PDGFRA (green) antibodies. N = 3 mice, two independent experiments. Scale bar, 100 μm; dashed lines demarcate borders between epithelium, papillary and reticular lamina propria, and submucosa. (E) Quantification of PI16 MFI in the papillary, reticular, and submucosal layers. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. One-way ANOVA and Tukey’s post-hoc test; ***P < 0.001. (F) Immunofluorescence experiments with anti-RFP (to detect mCherry, red) and K14 (epithelial marker, green) antibodies within the palatal gingiva of CCL2mCherry mice. Representative image of N = 3 mice from two independent experiments is shown. Scale bar, 100 μm; dashed line demarcates submucosal layer. (G) Left, representative images of immunofluorescence stained with CXCL1 (green) and SCA1 (red) antibodies. Scale bar, 100 μm; dashed lines demarcate epithelial–lamina propria border. Right, CXCL1 MFI in epithelium and lamina propria. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. Student’s test; ***P < 0.001. (H) Flow cytometry analysis and gating strategy for CCL7+ expressing gingival cells. N = 4 mice from two independent experiments. (I) Left, percent of CCL7+ cells that are PDGFRA+ (fibroblast identity) or PDGFRA− (non-fibroblast); right, percent of CCL7+ fibroblasts that are SCA1+ or SCA1−. Each dot represents one mouse, and data represent mean ± SEM. N = 4 mice, two independent experiments. Student’s test; **P < 0.01, ***P < 0.001. Epi, epithelium; PL, papillary layer; RL, reticular layer; SM, submucosa; AU, arbitrary unit.
Validation of oral fibroblast heterogeneity in mouse palate. (A) H&E staining of mouse palatal rugae. (B) Immunofluorescence experiment with SCA1 (red) and PDGFRA (green) antibodies. N = 3 mice, two independent experiments. Scale bar, 100 μm; dashed lines demarcate borders between epithelium, papillary and reticular lamina propria, and submucosa. (C) Quantification of SCA1 mean fluorescent intensity (MFI) in the papillary, reticular, and submucosal layers. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. One-way ANOVA and Tukey’s post-hoc test; **P < 0.01. (D) Immunofluorescence experiment with PI16 (red) and PDGFRA (green) antibodies. N = 3 mice, two independent experiments. Scale bar, 100 μm; dashed lines demarcate borders between epithelium, papillary and reticular lamina propria, and submucosa. (E) Quantification of PI16 MFI in the papillary, reticular, and submucosal layers. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. One-way ANOVA and Tukey’s post-hoc test; ***P < 0.001. (F) Immunofluorescence experiments with anti-RFP (to detect mCherry, red) and K14 (epithelial marker, green) antibodies within the palatal gingiva of CCL2mCherry mice. Representative image of N = 3 mice from two independent experiments is shown. Scale bar, 100 μm; dashed line demarcates submucosal layer. (G) Left, representative images of immunofluorescence stained with CXCL1 (green) and SCA1 (red) antibodies. Scale bar, 100 μm; dashed lines demarcate epithelial–lamina propria border. Right, CXCL1 MFI in epithelium and lamina propria. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice, two independent experiments. Student’s test; ***P < 0.001. (H) Flow cytometry analysis and gating strategy for CCL7+ expressing gingival cells. N = 4 mice from two independent experiments. (I) Left, percent of CCL7+ cells that are PDGFRA+ (fibroblast identity) or PDGFRA− (non-fibroblast); right, percent of CCL7+ fibroblasts that are SCA1+ or SCA1−. Each dot represents one mouse, and data represent mean ± SEM. N = 4 mice, two independent experiments. Student’s test; **P < 0.01, ***P < 0.001. Epi, epithelium; PL, papillary layer; RL, reticular layer; SM, submucosa; AU, arbitrary unit.
Given their chemotactic gene signature, we asked whether Prx1-derived SCA1+ fibroblasts accelerate wound healing by modulating the immune response. We first examined chemokine expression by the SCA1+ fibroblasts and found that CXCL1 and CCL7 were mostly expressed by cells other than fibroblasts (Fig. S3, G–I), whereas CCL2 was largely expressed by SCA1+ oral fibroblasts in both physiologic and wounded conditions (Fig. 5, B and C). We further examined inflammatory cell infiltration in Prx1+-enriched and deficient wounds at different healing time points. In Prx1enh+-enriched (R2) gingival wound, Ly6g+ neutrophil infiltration was subdued compared to the Prx1-poor (R4) gingiva (Fig. 5, D and E). In contrast, F4/80+ macrophage infiltration was swiftly upregulated by day 2 and reduced over time in the R2 wound, whereas the R4 wound had a delayed and sustained macrophage recruitment to the wound site (Fig. 5 F). The macrophages found in R4 wound exhibited an inflammatory phenotype as determined by their co-expression of IL1β (Fig. 5, G and H), whereas macrophages in R2 wound quickly progressed to a pro-resolving and healing phenotype, expressing TGFβ1 (Fig. 5, I and J). As CCL2 is a potent chemokine for monocyte/macrophage recruitment (Gschwandtner et al., 2019), our data suggest a cellular mechanism by which Prx1-derived SCA1+ fibroblasts facilitate rapid macrophage response to accelerate wound healing.
Heightened inflammation and delayed healing by Prx1+ cell ablation or CCL2 deletion
We next sought to establish a mechanistic link between Prx1enh+ cells and a pro-healing innate immune response in vivo. We first confirmed that ablation of Prx1enh+ cells in DTAPrx1 mice effectively reduced the number of SCA1+ fibroblasts by ∼75% in R2 wound bed (Fig. 6 A). This had a significant impact on myeloid cell infiltration by preventing early recruitment of F4/80+ macrophages in days 2 and 4 after injury, whereas the number of Ly6g+ neutrophils was significantly elevated compared to control groups (Fig. 6 B). Moreover, wounds with Prx1enh+ cell deletion had significantly higher percentage of pro-inflammatory macrophages whereas that of pro-resolving macrophages was reduced at day 2 and 6 after wounding, respectively (Fig. 6, C and D), demonstrating a similar shift in macrophage phenotype as seen in Prx1-deficient R4 wound.
Heightened inflammation and delayed healing by Prx1 + cell ablation or CCL2 deletion. (A) Left, representative immunofluorescent images of SCA1 (green) and vimentin (red) staining in day 4 wound of DTAPrx1 mice and control littermates. Arrowheads point to SCA1+vimentin+-immunopositive spindle-shaped cells. Right, quantification of SCA1+vimentin+ spindle-shaped cells in the wound bed per mm2 area. Scale bar, 100 μm. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test; ***P < 0.001. (B) F4/80+ macrophages (left) and Ly6g+ neutrophils (right) per live cells during wound healing in control and DTAPrx1 mice. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group and time point for a total of 38 mice (14 for day 2, 13 for day 4, and 11 for day 6), three independent experiments. Student’s t test; *P < 0.05. (C and D) Percentage of macrophages that are pro-inflammatory in day 2 wound (C) or that are pro-resolving in day 6 wound (D) in control and DTAPrx1 mice. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group for a total of 14 for day 2 (C) and 11 for day 6 (D), three independent experiments. Student’s t test; *P < 0.05, **P < 0.01. (E) Quantification of SCA1+ fibroblast percent that express CCL2 in Prx1CreERT:Ikbkbf/f littermate mice that received vehicle (corn oil, +veh) or tamoxifen (+tam). Analysis in day 2 wound is shown. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group for a total of 13 mice, three independent experiments. Student’s t test; ***P < 0.001. (F) F4/80+ macrophage infiltration to healing wounds in Prx1CreERT:Ikbkbf/f mice that received vehicle or tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group and time point for a total of 26 mice (14 for day 2 and 12 for day 6), three independent experiments. Student’s t test; **P < 0.01. (G and H) Quantification of pro-inflammatory macrophage percent in day 2 wound (G) and pro-resolving macrophages in day 6 wound (H) in Ikbkb-deleted mice. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group for a total of 14 mice for day 2 (G) and 12 for day 6 (H), three independent experiments. Student’s t test; *P < 0.05, ***P < 0.001. (I) Quantification of epithelial gap closure in day 2 wound (left) and COL3+ immunopositive area in day 4 wound bed (right) in Prx1CreERT:Ikbkbf/f mice that received vehicle or tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group for a total of 24 mice (12 for day 2 and 12 for day 4 wound), three independent experiments. Student’s t test; ***P < 0.001. (J) CCL2 expression in unwounded gingiva (R1) from control CCL2f/f-mCherry littermates and experimental Prx1CreERT:CCL2f/f-mCherry (△CCL2Prx1) mice 2 wk after tamoxifen injection (left). Right, quantification of CCL2+ fibroblasts in day 4 wounds by flow cytometry. Scale bar, 50 μm; SM, submucosa. CCL2 was visualized by staining with primary anti-RFP antibody followed by secondary antibody conjugated to AlexaFluor 647. N = 3–4 mice per group for a total of seven mice, two independent experiments. (K) Flow cytometry analysis of F4/80+ macrophage infiltration in day 2 wounds of control and △CCL2Prx1 mice. Right, quantification of macrophages per total live cell. Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11, three independent experiments. Student’s t test; *P < 0.05. (L) Quantification of percent pro-inflammatory macrophages (CD45+CD11b+F4/80+IL1β+), normalized to total macrophage cell numbers in △CCL2Prx1 mice in day 2 wounds. Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11, three independent experiments. Student’s t test; *P < 0.05. (M) Quantification of epithelial gap closure in day 2 wounds and COL3+ area in day 4 wounds in control, △CCL2Prx1 mice, and △CCL2Prx1 mice that received recombinant CCL2 (+rCCL2). Each dot represents one mouse, and data represent mean ± SEM. N = 5–11 mice per group for a total of 16 for day 2 wound and 27 for day 4 wound, three independent experiments. Student’s t test; *P < 0.05, **P < 0.01, ***P < 0.001.
Heightened inflammation and delayed healing by Prx1 + cell ablation or CCL2 deletion. (A) Left, representative immunofluorescent images of SCA1 (green) and vimentin (red) staining in day 4 wound of DTAPrx1 mice and control littermates. Arrowheads point to SCA1+vimentin+-immunopositive spindle-shaped cells. Right, quantification of SCA1+vimentin+ spindle-shaped cells in the wound bed per mm2 area. Scale bar, 100 μm. Each dot represents one mouse, and data represent mean ± SEM. N = 6 mice per group for a total of 12 mice, three independent experiments. Student’s t test; ***P < 0.001. (B) F4/80+ macrophages (left) and Ly6g+ neutrophils (right) per live cells during wound healing in control and DTAPrx1 mice. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group and time point for a total of 38 mice (14 for day 2, 13 for day 4, and 11 for day 6), three independent experiments. Student’s t test; *P < 0.05. (C and D) Percentage of macrophages that are pro-inflammatory in day 2 wound (C) or that are pro-resolving in day 6 wound (D) in control and DTAPrx1 mice. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group for a total of 14 for day 2 (C) and 11 for day 6 (D), three independent experiments. Student’s t test; *P < 0.05, **P < 0.01. (E) Quantification of SCA1+ fibroblast percent that express CCL2 in Prx1CreERT:Ikbkbf/f littermate mice that received vehicle (corn oil, +veh) or tamoxifen (+tam). Analysis in day 2 wound is shown. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group for a total of 13 mice, three independent experiments. Student’s t test; ***P < 0.001. (F) F4/80+ macrophage infiltration to healing wounds in Prx1CreERT:Ikbkbf/f mice that received vehicle or tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group and time point for a total of 26 mice (14 for day 2 and 12 for day 6), three independent experiments. Student’s t test; **P < 0.01. (G and H) Quantification of pro-inflammatory macrophage percent in day 2 wound (G) and pro-resolving macrophages in day 6 wound (H) in Ikbkb-deleted mice. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per group for a total of 14 mice for day 2 (G) and 12 for day 6 (H), three independent experiments. Student’s t test; *P < 0.05, ***P < 0.001. (I) Quantification of epithelial gap closure in day 2 wound (left) and COL3+ immunopositive area in day 4 wound bed (right) in Prx1CreERT:Ikbkbf/f mice that received vehicle or tamoxifen. Each dot represents one mouse, and data represent mean ± SEM. N = 5–7 mice per group for a total of 24 mice (12 for day 2 and 12 for day 4 wound), three independent experiments. Student’s t test; ***P < 0.001. (J) CCL2 expression in unwounded gingiva (R1) from control CCL2f/f-mCherry littermates and experimental Prx1CreERT:CCL2f/f-mCherry (△CCL2Prx1) mice 2 wk after tamoxifen injection (left). Right, quantification of CCL2+ fibroblasts in day 4 wounds by flow cytometry. Scale bar, 50 μm; SM, submucosa. CCL2 was visualized by staining with primary anti-RFP antibody followed by secondary antibody conjugated to AlexaFluor 647. N = 3–4 mice per group for a total of seven mice, two independent experiments. (K) Flow cytometry analysis of F4/80+ macrophage infiltration in day 2 wounds of control and △CCL2Prx1 mice. Right, quantification of macrophages per total live cell. Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11, three independent experiments. Student’s t test; *P < 0.05. (L) Quantification of percent pro-inflammatory macrophages (CD45+CD11b+F4/80+IL1β+), normalized to total macrophage cell numbers in △CCL2Prx1 mice in day 2 wounds. Each dot represents one mouse, and data represent mean ± SEM. N = 5–6 mice per group for a total of 11, three independent experiments. Student’s t test; *P < 0.05. (M) Quantification of epithelial gap closure in day 2 wounds and COL3+ area in day 4 wounds in control, △CCL2Prx1 mice, and △CCL2Prx1 mice that received recombinant CCL2 (+rCCL2). Each dot represents one mouse, and data represent mean ± SEM. N = 5–11 mice per group for a total of 16 for day 2 wound and 27 for day 4 wound, three independent experiments. Student’s t test; *P < 0.05, **P < 0.01, ***P < 0.001.
To next examine whether the knockdown of chemotactic function in Prx1-lineage fibroblasts impairs oral wound healing, we generated Prx1CreERT:Ikbkbf/f mice to selectively delete inhibitor of NF-κB kinase β (Ikbkb), an activator kinase for NF-κB that is responsible for an array of chemokine transcription including CCL2 (Richmond, 2002). As expected, deletion of Ikbkb markedly reduced CCL2 expression by the SCA1+ fibroblasts in healing wounds (Fig. 6 E). Concurrently, Ikbkb deletion delayed rapid recruitment of F4/80+ macrophages, promoted an inflammatory macrophage phenotype and reduced a pro-resolving phenotype (Fig. 6, F–H). Importantly, this led to delayed epithelial gap closure and significant reduction in stromal healing as demonstrated by the decrease in type 3 collagen-stained signal in the wound bed (Fig. 6 I), though the effect was moderate compared to complete Prx1enh+ cell ablation (Fig. 1 N).
To further determine whether CCL2 is the primary chemokine responsible for altered macrophage recruitment and mucosal healing, we examined mice that had CCL2 deleted specifically in Prx1-lineage fibroblasts (Prx1CreERT:CCL2f/f or △CCL2Prx1). This approach effectively reduced CCL2 expression by the oral fibroblasts in healing wounds (Fig. 6 J). The loss of CCL2 significantly reduced the rapid recruitment of F4/80+ macrophages (P < 0.05; Fig. 6 K) and increased the pro-inflammatory phenotype within the macrophage population (P < 0.05; Fig. 6 L). This change had a negative effect on epithelial gap closure and stromal healing, reducing COL3+ expression in the wound of △CCL2Prx1 mice compared to control groups (Fig. 6 M). Injecting recombinant CCL2 to wounded areas in △CCL2Prx1 mice partially rescued these wound healing parameters (Fig. 6 M). Collectively, the data suggest that CCL2 expression by the Prx1-lineage SCA1+ fibroblasts through NF-κB activity is required for rapid mucosal healing, which involves timely resolution of inflammation by the macrophages.
Improved skin wound healing by adoptive Prx1enh+ cell transfer
Cutaneous wounds that heal slowly by scar formation may heal more effectively following adoptive transfer of oral Prx1enh+ cells. To test this, we first sorted GFP+tdTomato+ Prx1enh+ cells from oral mucosae of Prx1CreERT:R26RtdTomato donor mice and injected them to scalp skin of recipient Prx1CreERT:R26RtdTomato mice (Fig. 7 A). In control Prx1CreERT:R26RtdTomato mice that had vehicle injected to the scalp, there were minimal tdTomato+ cells in both unwounded and wounded skin (Fig. 7 B), similar to previously reported findings (Currie et al., 2019). In contrast, Prx1enh+ cell injection prior to wounding substantially increased the number of tdTomato+ cells in unwounded skin and day 4 wound bed (Fig. 7, B and C), indicating that the injected cells remained viable during wound healing. We next examined cutaneous wound healing process in C57BL/6 recipient mice that received oral Prx1enh+ cells from Prx1CreERT-eGFP donor mice. Mice that received Prx1enh+ cells exhibited significantly accelerated wound closure on days 2 and 4 after injury compared to vehicle-injected control groups (Fig. 7, D and E). Histological examination of healed wounds revealed that Prx1enh+ transplantation facilitated more complete healing with hair follicle regeneration in the wound bed, which was limited in control mice (Fig. 7 F). Importantly, wounds that had Prx1enh+ supplementation showed significant decrease in αSMA expression compared to vehicle-injected wounds (Fig. 7 G), suggesting reduced cutaneous scarring. When inflammatory infiltrates were examined, we found that Prx1enh+ transfer significantly reduced the number of neutrophils while increasing the number of macrophages with a pro-resolving phenotype (Fig. 7, H–J), consistent with immunomodulatory function of Prx1enh+-derived fibroblasts in oral wounds (Fig. 5). Thus, Prx1enh+ cell transplantation in skin shifts its wound healing trajectory from scarring to regeneration that is associated with rapid resolution of inflammation.
Improved skin wound healing by adoptive Prx1 enh+ cell transfer. (A) Schematic experimental approach using Prx1CreERT:R26RtdTomato mice as donor and recipient groups after tamoxifen administration. (B) Representative immunofluorescent images of unwounded and day 4 scalp skin wound in Prx1CreERT:R26RtdTomato mice that received saline (+vehicle) or Prx1enh+ cell grafting from littermate Prx1CreERT:R26RtdTomato mice. N = 3 mice per treatment and endpoint for a total of 12 mice, two independent experiments. (C) Quantification of Prx1+-lineage tdTomato+ cells in unwounded and wounded scalp skin. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice per treatment and endpoint for a total of six for unwounded and six for day 2 wound, two independent experiments. (D) Representative microphotographs of 1 mm scalp wound in C57BL/6 mice that received either vehicle or Prx1enh+ cells prior to wounding. Donor cells were derived from Prx1CreERT-eGFP mice. Scale bar, 1 mm. N = 6–7 mice per treatment for a total of 13, three independent experiments. (E) Longitudinal quantification of open skin wound area during wound healing process. Each dot represents one mouse within time point, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01, ***P < 0.001. (F) Left, H&E-stained sections from day 6 skin wounds that received either vehicle (+veh) or cell transfer (+Prx1enh+). Arrows designate original 1 mm wound length; scale bar, 0.5 mm. Right, quantification of hair follicle numbers per 1 mm length within wound bed. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01. (G) Left, representative immunofluorescent images of skin wounds that were stained with antibody against αSMA. Dashed line demarcates epithelium–dermis border. Scale bar, 100 μm. Right, quantification of αSMA-immunopositive area in mm2. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01. (H) Representative flow cytometry plots pre-gated for live CD45+CD11b+ cell populations in day 6 skin wounds that received vehicle or Prx1enh+ cells. N = 6–7 mice per treatment for a total of 13, three independent experiments. (I) Quantification of neutrophils (PMN, CD45+CD11b+Ly6g+) and macrophages (CD45+CD11b+F4/80+) in day 6 wounds, normalized to total CD45+ leukocyte population. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; *P < 0.05. (J) Quantification of pro-resolving macrophages that express TGFβ1 in day 6 wounds of vehicle or Prx1enh+-treated mice, normalized to total macrophage cell numbers. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; *P < 0.05.
Improved skin wound healing by adoptive Prx1 enh+ cell transfer. (A) Schematic experimental approach using Prx1CreERT:R26RtdTomato mice as donor and recipient groups after tamoxifen administration. (B) Representative immunofluorescent images of unwounded and day 4 scalp skin wound in Prx1CreERT:R26RtdTomato mice that received saline (+vehicle) or Prx1enh+ cell grafting from littermate Prx1CreERT:R26RtdTomato mice. N = 3 mice per treatment and endpoint for a total of 12 mice, two independent experiments. (C) Quantification of Prx1+-lineage tdTomato+ cells in unwounded and wounded scalp skin. Each dot represents one mouse, and data represent mean ± SEM. N = 3 mice per treatment and endpoint for a total of six for unwounded and six for day 2 wound, two independent experiments. (D) Representative microphotographs of 1 mm scalp wound in C57BL/6 mice that received either vehicle or Prx1enh+ cells prior to wounding. Donor cells were derived from Prx1CreERT-eGFP mice. Scale bar, 1 mm. N = 6–7 mice per treatment for a total of 13, three independent experiments. (E) Longitudinal quantification of open skin wound area during wound healing process. Each dot represents one mouse within time point, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01, ***P < 0.001. (F) Left, H&E-stained sections from day 6 skin wounds that received either vehicle (+veh) or cell transfer (+Prx1enh+). Arrows designate original 1 mm wound length; scale bar, 0.5 mm. Right, quantification of hair follicle numbers per 1 mm length within wound bed. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01. (G) Left, representative immunofluorescent images of skin wounds that were stained with antibody against αSMA. Dashed line demarcates epithelium–dermis border. Scale bar, 100 μm. Right, quantification of αSMA-immunopositive area in mm2. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; **P < 0.01. (H) Representative flow cytometry plots pre-gated for live CD45+CD11b+ cell populations in day 6 skin wounds that received vehicle or Prx1enh+ cells. N = 6–7 mice per treatment for a total of 13, three independent experiments. (I) Quantification of neutrophils (PMN, CD45+CD11b+Ly6g+) and macrophages (CD45+CD11b+F4/80+) in day 6 wounds, normalized to total CD45+ leukocyte population. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; *P < 0.05. (J) Quantification of pro-resolving macrophages that express TGFβ1 in day 6 wounds of vehicle or Prx1enh+-treated mice, normalized to total macrophage cell numbers. Each dot represents one mouse, and data represent mean ± SEM. N = 6–7 mice per treatment for a total of 13 mice, three independent experiments. Student’s t test; *P < 0.05.
Transcriptomic profile and spatial distribution of human Prx1+ oral fibroblasts
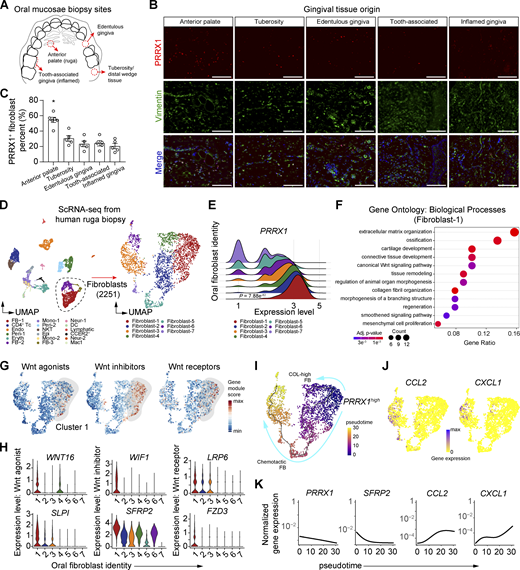
We next investigated whether similar Prx1enh+ cell populations are found in the human oral cavity. We examined oral biopsies collected from various anatomical sites and found that while Prx1+ immuno-positive oral fibroblasts were present in all gingival tissues examined, anterior palate rugae had the highest frequency of these cells (Fig. 8, A–C), consistent with specific geographical identity observed in mice (Fig. 1, A and B). To determine whether the human Prx1+ cells exhibit transcriptomic similarities to murine Prx1enh+ cells, we isolated cells from anterior palate and performed scRNA-seq to analyze fibroblast population as determined by their matrisome expression (Fig. 8 D and Fig. S4 A). As expected, PRRX1 expression was highly enriched and represented as the most abundant fibroblast population as cluster 1 (Fig. 8 E and Table S3). GO analysis of differentially upregulated transcripts from this Prx1+ cluster revealed gene enrichment in ossification, chondrogenesis, regeneration and inhibition of Wnt signaling pathway (Fig. 8 F and Table S4), suggesting a progenitor identity and pro-healing function. Similar to murine Prx1enh+ fibroblasts, the human Prx1+ cell cluster was highly enriched with Wnt inhibitor transcripts, with moderate expression of Wnt agonist and receptor genes (Fig. 8 G). The significantly upregulated genes from Prx1+ cluster included WNT16 and SLPI, WIF1 and SFRP2, and LRP6 and FZD3, for activation, inhibition, and receptor function of Wnt signaling, respectively (Fig. 8 H).
Transcriptomic profile and spatial distribution of human Prx1+oral fibroblasts. (A) Graphical diagram of human oral biopsy locations from the occlusal view of maxilla. (B) Representative immunofluorescent images of each oral biopsy specimen stained with PRRX1 (red) and vimentin (green) antibodies. Scale bar, 100 μm. N = 5–6 human specimen per group for a total of 26 specimens, three independent experiments. (C) Quantification of PRRX1+vimentin+ spindle-shaped cell percentage per total spindle-shaped fibroblasts. Each dot represents one human specimen, and data represent mean ± SEM. N = 5–6 specimens for a total of 26 samples, three independent experiments. One-way ANOVA and Tukey’s post-hoc test; *P < 0.05. (D) UMAP of single cells derived from the anterior rugae of human oral biopsy (left) and fibroblast sub-clusters (right). (E) Ridge plot of PRRX1 gene expression from oral fibroblast subclusters. (F) GO analysis for biological processes of PRRX1-enriched fibroblast cluster 1. Full list is provided in Table S4. (G) Gene module scores for Wnt activator, inhibitor, and receptor genes. (H) Violin plots of Wnt-associated genes significantly upregulated in cluster 1, as determined by Wilcoxon rank-sum analysis. (I) Monocle-based trajectory and pseudotime calculation of fibroblast subclusters with PRRX1high selected as a starting node. (J) Feature plot of single-cell expression of CCL2 and CXCL1 in oral fibroblast clusters. (K) Normalized gene expression of PRRX1, SFRP2, CCL2, and CXCL1 along calculated pseudotime.
Transcriptomic profile and spatial distribution of human Prx1+oral fibroblasts. (A) Graphical diagram of human oral biopsy locations from the occlusal view of maxilla. (B) Representative immunofluorescent images of each oral biopsy specimen stained with PRRX1 (red) and vimentin (green) antibodies. Scale bar, 100 μm. N = 5–6 human specimen per group for a total of 26 specimens, three independent experiments. (C) Quantification of PRRX1+vimentin+ spindle-shaped cell percentage per total spindle-shaped fibroblasts. Each dot represents one human specimen, and data represent mean ± SEM. N = 5–6 specimens for a total of 26 samples, three independent experiments. One-way ANOVA and Tukey’s post-hoc test; *P < 0.05. (D) UMAP of single cells derived from the anterior rugae of human oral biopsy (left) and fibroblast sub-clusters (right). (E) Ridge plot of PRRX1 gene expression from oral fibroblast subclusters. (F) GO analysis for biological processes of PRRX1-enriched fibroblast cluster 1. Full list is provided in Table S4. (G) Gene module scores for Wnt activator, inhibitor, and receptor genes. (H) Violin plots of Wnt-associated genes significantly upregulated in cluster 1, as determined by Wilcoxon rank-sum analysis. (I) Monocle-based trajectory and pseudotime calculation of fibroblast subclusters with PRRX1high selected as a starting node. (J) Feature plot of single-cell expression of CCL2 and CXCL1 in oral fibroblast clusters. (K) Normalized gene expression of PRRX1, SFRP2, CCL2, and CXCL1 along calculated pseudotime.
scRNA-seq analysis of human anterior palatal gingiva. (A) UMAP of putative genes highly expressed in each cluster identified. FB, fibroblast. (B) UMAP of myeloid and fibroblast subsets. Mono, monocyte; DC, dendritic cells; Mac1, inflammatory macrophage. (C) CellChat analysis of single cells for CCL and CXCL signaling networks between fibroblast and myeloid subsets. (D) Relative contribution of CCL (left) and CXCL (right) ligand to known receptor interaction among fibroblast-myeloid cell subsets. (E) Violin plots of ligand and receptor genes (CCL2, ACKR1, CXCL8, and CXCR2) implicated as the highest contributors of CCL and CXCL signaling interactions.
scRNA-seq analysis of human anterior palatal gingiva. (A) UMAP of putative genes highly expressed in each cluster identified. FB, fibroblast. (B) UMAP of myeloid and fibroblast subsets. Mono, monocyte; DC, dendritic cells; Mac1, inflammatory macrophage. (C) CellChat analysis of single cells for CCL and CXCL signaling networks between fibroblast and myeloid subsets. (D) Relative contribution of CCL (left) and CXCL (right) ligand to known receptor interaction among fibroblast-myeloid cell subsets. (E) Violin plots of ligand and receptor genes (CCL2, ACKR1, CXCL8, and CXCR2) implicated as the highest contributors of CCL and CXCL signaling interactions.
Finally, we asked whether the human Prx1+ fibroblasts follow a differentiation pattern toward chemotactic fibroblast cluster, similar to that observed in mouse palate. Monocle-based trajectory analysis of human PRRX1high fibroblasts generated two branches of trajectory, one toward high collagen-content fibroblasts and another toward chemotactic fibroblasts (Fig. 8 I). Although humans do not express orthologous gene to murine Ly6a, similarly chemotactic fibroblast cluster 3 was found to co-express CCL2 and CXCL1 (Fig. 8 J), whereas CCL7 was not detected. Consistent with murine trajectory analysis (Fig. 3 B), the expression of PRRX1 and SFRP2 was inversely correlated to that of CCL2 and CXCL1 along calculated pseudotime (Fig. 8 K). To gain further insight into oral stromal-immune cell crosstalk during homeostasis, we selected fibroblast and myeloid cell populations from the scRNA-seq data and examined cell–cell communication network using CellChat package (Jin et al., 2021). When CCL and CXCL signaling networks were examined, fibroblast clusters were found to be a prominent source of CCL ligands, among which CCL2 contributed the most for communication with ACKR1+ myeloid cell population (Fig. S4, B–E). In contrast, myeloid cells were the major source of CXCL ligand, among which CXCL8 to CXCR2+ dendritic cell-signaling network was most pronounced (Fig. S4, B–E). Taken together, the scRNA-seq data strongly implicate a potential role of Prx1-lineage oral fibroblast in regulating immune response via CCL2 and enhancing wound healing in humans, which aligns with our findings in a mouse model.
Discussion
Mucosal wounds in the oral cavity exhibit rapid resolution of inflammation and healing with minimal scarring (Griffin et al., 2021), and these unique properties have established oral wound healing as an ideal model to study mechanisms of regeneration. Here, we demonstrate that Prx1enh+ cells are critical for robust oral wound healing as shown by two approaches. Firstly, a vigorous healing response in Prx1-enriched mucosa was much greater than that in a Prx1-poor mucosa, and the poor healing response was rescued by transplantation of Prx1-enriched mucosa onto Prx1-deficient area. Secondly, robust healing in the Prx1-enriched mucosa was reversed by induced ablation of Prx1enh+ cells in adult mice. Genetic tracing and scRNA-seq analyses demonstrated that Prx1enh+ cells give rise to heterogeneous fibroblast lineages during wound healing, among which SCA1+ fibroblasts were most dominant. Mechanistically, CCL2 production by the Prx1-lineage SCA1+ fibroblasts was responsible for rapid wound healing, as deletion of CCL2 or its upstream transcription factor NF-κB in Prx1enh+ cells had a negative impact on macrophage trafficking and resolution of inflammation. Importantly, murine and human Prx1+ oral fibroblasts shared similar spatial localization in the gingiva, and scRNA-seq analyses revealed that they had similar transcriptomic profiles, validating the key features of Prx1+ fibroblasts in mouse and humans. Thus, a distinct Prx1enh+ fibroblast progenitor population enhances mucosal healing process by giving rise to chemotactic fibroblasts that subsequently modulates innate immune response and resolution of inflammation.
We found that ∼10% of connective tissue cells exhibit postnatal Prx1enh+ identity in the anterior gingiva in adult mice. This contrasts with its low abundance in skin that heals by repair over regeneration (Currie et al., 2019; Ko et al., 2022). In support of this pro-healing role, Prx1enh+ cell deletion has been shown to impair bone regeneration due to the loss cells that can differentiate into chondrocytes and osteoblasts (Esposito et al., 2020; Bassir et al., 2019). Our findings suggest that in an oral mucosal niche, Prx1enh+ cells uniquely differentiate toward immunomodulatory fibroblasts that are crucial for wound healing. It has been reported that Prx1-lineage cells are responsible for scar formation in ventral skin wounds (Leavitt et al., 2020). The different results are likely due to a constitutive labeling of postembryonic lineage cells from Prx1-Cre mice by Leavitt et al. (2020), whereas our study focused on postnatal fibroblasts with active 2.4 kb Prx1enh+ enhancer activity using tamoxifen inducible Cre recombinase model. Indeed, we found that Prx1enh+ oral fibroblasts minimally contributed to myofibroblast differentiation, a major cell type responsible for scarring (Guerrero-Juarez et al., 2019). However, this does not rule out the possibility that Prx1-lineage fibroblasts produced during development may contribute to excessive scar formation in wound healing or in specific disease conditions such as a tumor microenvironment (Lee et al., 2022).
Prx1enh+ cells were immunopositive for Prx1 protein, though other fibroblast subsets also expressed Prx1 protein, which accounted for 24% per total Prx1 immunopositive cells (Fig. S1 B). Moreover, scRNA-seq data demonstrated that Prrx1 mRNA is expressed in a gradient pattern and that low-to-moderate levels may be present in other fibroblast subtypes (Fig. 2 G). These data suggest that there is a discrepancy between Prx1 immunopositive cells and Prx1enh+ cells that are discretely labeled by transcriptional activity of a 2.4 kb element from the Prx1 promoter. Moreover, Prx1 alone may not be an ideal marker to detect Prx1enh+ cells. To further characterize the Prx1enh+ cells, we isolated Prx1enh+ and Prx1enh- fibroblasts by FACS and performed qPCR. The results show that Prx1enh+ cells highly expressed Prrx1 and Wnt-associated genes such as Wif1, Sfrp2, and Slpi compared to Prx1enh- fibroblasts. As Wif1 and Sfrp2 were distinctly expressed in Prrx1high clusters in mouse and human scRNA-seq dataset, these may be more reliable markers to detect Prx1enh+ cells in other organs. Functionally, Prx1enh+ cells were distinct from other fibroblast populations, as determined by the upregulated genes associated with inhibition of the Wnt pathway. This is consistent with the pro-healing role of Prx1enh+ cells, supported by the studies that report upregulation of these genes in progenitors in the skin and corneum (Poggi et al., 2018; Lim et al., 2013) and by the studies that demonstrate a requirement for Wnt inhibitory genes for proper wound healing (de Castro et al., 2021; Alfaro et al., 2013; Mirotsou et al., 2007).
A unique characteristic of oral wound healing is the early transition to pro-wound healing and inflammation-revolving phenotype (Griffin et al., 2021; Szpaderska et al., 2003). We found that this is one of the primary mechanisms by which Prx1-lineage fibroblasts contribute to robust wound healing. Areas of the gingiva enriched in Prx1enh+ cells had higher baseline neutrophil and macrophage numbers compared to Prx1-deficient gingiva. Upon injury, Prx1enh+-enriched wounds initially showed pronounced macrophage infiltration that quickly reduced over time, paired with timely transition from an inflammatory to pro-resolving phenotype. Ablation of Prx1enh+ cells in DTAPrx1 mice reversed these parameters and delayed healing, demonstrating that Prx1-lineage cells were responsible for the transition in this innate immune response. Consistent with our findings, a study comparing human buccal mucosa and skin wounds demonstrated that oral wounds exhibited heightened inflammation during early time points that subsided rapidly during the later healing phase (Iglesias-Bartolome et al., 2018). Therefore, oral wounds appear to follow a specific temporal pattern characterized by primed induction of inflammation and rapid transition to resolution that is regulated by Prx1-derived SCA1+ oral fibroblasts. These findings resonate with growing evidence implicating unconventional immunomodulatory functions of fibroblasts in skin inflammatory conditions and rheumatoid disorders (Croft et al., 2019; Reynolds et al., 2021; Boothby et al., 2021; Ko et al., 2022; Xu et al., 2022).
Prx1-derived SCA1+ oral fibroblasts were the primary source of CCL2 in unwounded and wounded gingiva, even when compared to chemokine-producing leukocytes. Specific deletion of Ccl2 or Ikbkb in Prx1-lineage cells resulted in impaired macrophage recruitment and stromal healing. This established the importance of fibroblast-derived CCL2 for an accelerated healing response that is consistent with the role of CCL2 in dermal healing (Low et al., 2001; Boniakowski et al., 2018). In diabetic skin wounds where CCL2 expression is reduced and macrophage recruitment is delayed, healing is sufficiently rescued with CCL2 treatment (Ishida et al., 2019; Wood et al., 2014). In addition to chemoattractant property, CCL2 has been shown to modulate macrophage polarization toward pro-resolving phenotype (Sierra-Filardi et al., 2014), consistent with our findings from Prx1enh+-enriched wounds. However, tight control of CCL2 is an important consideration given that aberrant CCL2 expression is linked to fibrotic disorders (Wong et al., 2011). In gingiva, submucosal SCA1+ fibroblasts express baseline CCL2 without intrinsic fibrosis, suggesting that enhanced macrophage trafficking during steady state likely requires external stimuli (i.e., bacterial products upon injury) to become activated. In addition, fibroblast-produced CCL2 has been reported in human gingiva, consistent with its expression by these cells in murine gingival tissue (Yu and Graves, 1995).
In conclusion, our study provides a cellular and mechanistic basis for rapid oral mucosal healing by identifying a distinct Prx1enh+ fibroblast progenitor subset and demonstrating its intrinsic immunomodulatory role. The primed immune response by this specific fibroblast population provides a mechanistic explanation of how resolution of inflammation and the switch to a pro-wound healing environment is accelerated in mucosal wounds compared to dermal wounds, which may be important for minimizing fibrotic healing response as demonstrated by the adoptive cell transfer experiments (Fig. 7). This is particularly important because mucosal healing is not resistant to complications as seen from corneal wound repair and intestinal fibrosis in diseased conditions (Latella and Rieder, 2017; Mobaraki et al., 2019). Given the remarkable geographic and transcriptomic similarities of Prx1+ cells between mouse and human, we postulate that human Prx1+ fibroblasts may enhance oral wound healing by differentiating toward immunomodulatory fibroblasts to rapidly transition to a pro-healing innate immune response. This is supported by the trajectory analysis of human scRNA-seq data that demonstrated a lineage progression of Prx1+ fibroblasts into CCL2 and CXCL1-expressing fibroblast subtypes. Since transplantation of Prx1-enriched oral tissue improves healing outcome, utilization of similar stromal subset in humans may lead to a development of new therapeutic strategies for optimal wound regeneration in patients.
Materials and methods
Animals
Animal studies were performed conforming to Institutional Animal and Use Committee–approved protocols (#804855 and #807062). The study used mice purchased from the Jackson Laboratory as follows: C57BL/6J (#000664), B6.Cg-Tg(Prrx1-cre/ERT2,-EGFP)1Smkm/J (#029211), B6.Cg-Gt(ROSA)26Sortm9(CAG-tdTomato)Hze/J (#007909), B6.129P2-Gt(ROSA)26Sortm1(DTA)Lky/J (#009669), B6.Cg-Ccl2tm1.1Pame/J (#016849). Ikbkbflox/flox mice were obtained from Dr. Michael Karin (University of California, San Diego, La Jolla, CA, USA). Mice were housed in regular corn bedding under 12/12 light–dark cycles. All mice had C57BL/6 genetic background and were backcrossed at least five times in-house. Littermate control mice were used unless otherwise indicated in the figure legends.
Lineage tracing of Prx1+ progenitors
8–10-wk-old Prx1CreERT-eGFP:R26RtdTomato mice were injected with tamoxifen (1 mg/25 g, intraperitoneal) for five consecutive days. Mice were euthanized after 1, 4, and 16 wk from the last injection date. For wound healing models, mice were injected with tamoxifen as above and wounds were created after 1 d from last injection date. Animals were euthanized for downstream analyses after healing for indicated period. Paraffin or cryoembedded sections were prepared, and tdTomato and GFP signals were detected with anti-RFP and anti-GFP antibodies.
Oral wound induction
Animals were anesthetized with a mixture of ketamine, xylazine, and acepromazine through intraperitoneal injection. Oral cavity was propped open using 0.01” stainless steel ligation wires by engaging maxillary and mandibular incisors, and buccal mucosae were retracted with a custom-made wire apparatus. 1-mm biopsy punch was used to create wounds in R2 and R4 palatal gingivae, and excised tissues were completely removed with surgical forceps and scalpel blade. Full thickness wounding was confirmed by exposing the underlying bone. Mice were supplemented with Diet-Gel (ClearH2O) and housed in ALPHA-Dri bedding throughout the entire healing process to prevent food impaction and infection in the wound. In a set of experiments, mice received 50 ng (10 ng/μl) of recombinant CCL2 (BD Biosciences) at the time of wound induction through vestibular infiltration using a customized 34-gauge microliter syringe (Hamilton).
Gingival autograft surgery
Mouse palate was rinsed with chlorhexidine gluconate (0.12%) prior to autograft surgery. 1 mm R2 or R4 gingival tissue was excised from anesthetized mice as above. Submarginal incision was made from the marginal gingiva medial to the first molar tooth, and additional linear incision approximately 2 mm in length was created 0.5 mm apical to first molar tooth. Full thickness flap was raised without disrupting greater palatine artery, and the autograft was inserted underneath the flap, exposing epithelium side through the linear incision. A sterile 1 × 1 mm paper dressing was placed on top of the recipient site, and cyanoacrylate adhesive was applied. After 7 d of healing, 0.5 mm wound was created with biopsy punch, and full-thickness tissue was removed with surgical forceps and scalpel blade. Mice were euthanized after 2 d for further downstream analysis.
Histology and histomorphometric analysis
Harvested samples were fixed in 10% formalin at 4°C overnight, washed, and de-calcified in 14% EDTA (pH 7.4) for 2 wk. Tissues were processed with either paraffin embedding for histomorphometric analysis or cryoembedding after dehydration in 30% sucrose solution for immunofluorescence. For R2 wound, sections were sliced diagonally parallel to the mucogingival junction, and for R4 wound, sections were sliced in a sagittal direction. Paraffin sections were prepared at 5 μm thickness, re-hydrated and stained for H&E or Masson’s trichrome agent per the manufacturer’s protocols (Azer Scientific). Cryosections were prepared in 10 μm thickness and utilized for downstream immunofluorescence analyses.
EdU incorporation assay
Oral wounds were created in 8-wk-old Prx1CreERT-eGFP:R26RtdTomato mice that received tamoxifen as above. After 3 d of healing, EdU (1 mg/25 g) was administered through intraperitoneal injection. Mice were euthanized after 24 h, and cryosections were prepared. EdU was visualized using Click-iT Cell Proliferation Kit (Thermo Fisher Scientific).
Immunofluorescence
Paraffin or frozen sections were utilized for immunofluorescence experiments. For paraffin-embedded samples, sections were re-hydrated, and epitopes were retrieved by heat and pressure treatment at 121°C in citrate buffer (pH 6). Cryosections were hydrated for 10 min in 1× PBS plus 0.05% Tween-20. Nonspecific binding was blocked by incubating slides in 1% bovine serum albumin, 0.1% Triton-X, 0.05% Tween-20, and 1% of serum from host species matching that of secondary antibody. Immunofluorescence was performed using primary and secondary antibodies following standard protocols. Species-matching IgG was used in place of primary antibody as a negative control to determine background signals. Antibody list is provided in Table S5.
Flow cytometry
Palatal gingiva was dissected by gliding the scalpel blade against underlying bone to prevent inadvertent isolation of nasal cavity-associated lymphoid tissues. For wound harvest, two wounds from one mouse were dissected and combined for enzymatic digestion. In some experiments, four wounds from two mice were combined to obtain a single experimental data point. Tissues were minced and digested at 37°C with constant agitation in RPMI or DMEM media (for leukocyte or fibroblast analysis, respectively) containing DNAse I (0.15 mg/ml; Roche), collagenase type IV (3.2 mg/ml; Gibco), and dispase (2.6 mg/ml; Sigma-Aldrich) for 1 h. Clumps and debris were removed by filtering cells through 70-μm mesh, and red blood cells were lysed with Ammonium-Chloride-Potassium buffer. Fc receptors were blocked with CD16/32 antibody, and viability dye (Zombie Yellow) was used to distinguish live and dead cells. Standard staining protocols were followed for cell surface antigens. For intracellular staining, cells were incubated in fixation buffer for 15 min at room temperature followed by washing and incubation with antibodies in permeabilization buffer for 30 min. For lineage-negative selection, cells were stained with CD45, CD31, Ter119, and Epcam antibodies, and positive events were excluded. Data were acquired using BD LSR II Flow Cytometer and analyzed using FlowJo software. Isotype IgG or fluorescence-minus-one controls were used to determine gating strategy.
scRNA-seq and computational analysis
Palatal gingivae from two 8-wk-old C57BL/6 mice were isolated and enzymatically digested as described above. Cells were sorted on FACSAria II SORP instrument to collect CD45−DAPI− singlets. Single-cell preparations were pooled and run on 10× Genomics platform per the manufacturer’s protocols by the University of Pennsylvania Next Generation Sequencing Core. cDNA library was sequenced on a HiSeq 4000. CellRanger Software was used on default settings, and raw reads were aligned to mouse genome GRCm38. Downstream analyses were performed using R language. Cells with >3 absolute median deviations (nmads) for mitrochondrial gene percent (>8.85%), nFeature (>3238), and nCount (>9951) were excluded from downstream analyses. In total, CellRanger generated 4,861 cells, and further outlier filtering generated 4,402 cells. Fibroblast clusters expressing >3 Col3a1 unique molecular identifiers (UMI) were selected, and cells expressing >3 nmads of stress-associated genes (Fos, Atf3, Hspa1a) were excluded, resulting in 2,695 oral fibroblasts for downstream analyses including gene ontology analysis using ClusterProfiler (Yu et al., 2012). Data visualization was performed using Seurat (Stuart et al., 2019; ver.3) such as Heatmap, Featureplot, Vlnplot, Ridgeplot, and Dotplot. Differentially expressed genes were calculated using Findmarker function with default Wilcoxon rank-sum analysis. Gene module score for Wnt-associated genes was determined by creating a list of known Wnt agonists (Wnt1-16 and Slpi), Wnt antagonist (Sfrp1-5, Dkk1-4, and Wif1), and Wnt receptor genes (Fzd1-10, Lrp5/6). Pseudotime trajectory analysis was performed using Monocle 3 package (v.3). After preprocessing by principal component analysis and uniform manifold approximation and projection (UMAP) dimensional reduction, mesenchymal cells were identified by enriched expression of Col3a1, Dcn, and Eln. Post-pseudotime calculation, root node was set to Prrx1high cells, and major cell branches towards Ly6a+, Pi16+, and Acan+ were selected for further gene expression analysis.
For human oral fibroblast analysis, patients undergoing free gingival graft surgery consented to donating anterior rugae tissues (Institutional Review Board approval #844933). Rugae tissues were processed within 2 h of initial isolation as described above. Cells that expressed <200 or >25,000 UMI or more than 10% mitochondrial gene expression were excluded. Fibroblasts were selected based on the enriched expression of COL3A1, DCN, and PDGFRA. Visualization, differential gene expression, and trajectory analyses were carried out as described above. For CellChat analysis, fibroblasts and myeloid cells including monocytes, macrophages, and dendritic cells were subset, and analyzed using conventional input as described (Jin et al., 2021) for CCL- and CXCL-specific pathways.
FACS and real-time qPCR
The anterior palate tissues were processed to obtain single-cell suspension as described above. Cells were stained with viability dye and antibodies for PDGFRA and lineage negative selection. Prx1+ cells that inherently express enhanced GFP were sorted by live lineage−GFP+PDGFRA+, whereas Prx1− fibroblasts were sorted by lineage−GFP−PDGFRA+ selection on FACSAria II flow cytometer. Cells were pooled from two to three mice per experiment to obtain at least 2 × 104 GFP+ Prx1+ fibroblasts to be sorted directly into cold lysis buffer. RNeasy Micro Kit (Qiagen) was used to extract RNA per the manufacturer’s protocols. For cDNA library preparation, High-capacity cDNA synthesis kit (Applied Biosystems) was used, and qPCR was performed with Fast Power SYBR Green (Applied Biosystems) on Step-one Plus machine (Thermo Fisher Scientific). L32 was used as a housekeeping gene and normalization process.
Adoptive cell transfer and skin wounding
Oral mucosal tissues from Prx1CreERT-eGFP or Prx1CreERT:R26RtdTomato mice were enzymatically digested as above and sorted by viability dye− and GFP+ or tdTomato+ signal to purify Prx1enh+ cells for cell transfer. Cells from two to three donor mice were pooled to obtain ∼104 Prx1enh+ cells. Recipient mice had scalp hair removed with a shaver and depilatory cream, followed by injection of 104 Prx1enh+ cells in 50 µl of PBS intradermally. In another set of experiment, recipient mice carrying Prx1CreERT:R26RtdTomato transgenes were grafted with Prx1enh+ cells to validate successful cell transplantation. After 48 h post–cell injection, full-thickness scalp wounds that included removal of periosteal layer were created. Wounds were splinted with a silicone ring that was 10 mm in outer diameter and 5 mm in inner diameter. Cyanoacrylate adhesive and non-resorbable polypropylene sutures were placed to secure the stent, which was removed day 4 after wounding.
Human gingival biopsy
De-identified clinical samples were collected from patients undergoing periodontal resective surgery at the University of Pennsylvania School of Dental Medicine, in accordance with Institutional Review Board–approved protocol (#844933). Consenting adult patients in general good health without history of smoking or uncontrolled diabetes were included. Age and sex were not considered as biological variables. Gingival tissues were fixed in 10% formalin within 2 h of resection and processed in paraffin wax as described above.
Image analysis
Histostained and immunofluorescent images were acquired using Keyence BZ-X800 Microscope. Images were processed using BZ-X800 Analyzer Software. ImageJ Software was used to count percent positive cells and fluorescent areas. Two to three independent examiners analyzed images in a double-blinded fashion.
Statistical analysis
Statistical analysis was carried out with GraphPad Prism software. All data represent mean ± SEM unless otherwise indicated in the figure legend. Sample size was determined by power analysis with type I error at 0.05 and 40% predicted effect. Normal distribution of the data points was determined by Shapiro–Wilk normality test. Unpaired Student’s t-test was performed for comparisons between two groups, whereas for multi-group comparisons, one-way ANOVA and Tukey’s post-hoc test were used. Effects were considered statistically significant when P < 0.05. Individual animal or pooled set (i.e., pooled from two mice) was used as a unit of measurement (N). Experiments were independently replicated two to three times. Sex was not considered a factor for wound healing analysis.
Online supplemental material
Fig. S1 shows characterization of Prx1enh+ cells in oral mucosa and validation of Prx1enh+ deletion in mice with DTA-mediated ablation. Fig. S2 shows scRNA-seq data from murine palatal mucosa and characterization of single-cell transcriptome. Fig. S3 shows in vivo validation of oral fibroblast cluster markers identified from scRNA-seq using immunofluorescence and flow cytometry approaches. Fig. S4 shows scRNA-seq data from human palatal rugae, characterization of cluster specific genes, and cell to cell ligand-receptor patterns. Tables S1 and S3 provide pseudobulk differential gene expression of fibroblast subtypes in murine and human oral palatal mucosa, respectively. Tables S2 and S4 provide gene features identified by gene ontology analysis that is characteristic of each fibroblast cluster from murine and human scRNA-seq data, respectively. Table S5 lists antibodies and primers used in this study.
Data availability
scRNA-seq data are available through Gene Expression Omnibus (accession number GSE217720). New codes were not generated from this study.
Acknowledgments
We thank Michael Karin (University of California, San Diego, La Jolla, CA, USA) for providing Ikbkbf/f transgenic mice.
This study was supported by grants from the National Institute of Dental and Craniofacial Research K08-DE027129 (K.I. Ko), R01-DE030415 (K.I. Ko), and R01-DE019108 (D.T. Graves).
Author contributions: K.I. Ko: conceptualization, methodology, computational analysis, validation, formal analysis, investigation, resources, data curation, writing—original draft, review & editing, visualization, supervision, project administration and funding acquisition; B.P. DerGarabedian: validation, formal analysis, investigation, resources, data curation, writing—review & editing; Z. Chen: validation, formal analysis, investigation, resources, data curation, writing—review & editing; R. Debnath: investigation, data curation, writing—review & editing; A. Ko: computational analysis, investigation, data curation; B.N. Link: investigation, data curation; J.M. Korostoff: resources, writing—review & drafting; D.T. Graves: writing—original draft, review & editing, funding acquisition.
References
Author notes
Disclosures: The authors declare no competing interests exist.
Supplementary data
lists mouse oralpalate fibro.
lists mouse fibro GO analysis.
lists human oralpalate fibro.
lists human fibro GO analysis.