B cell activating factor (BAFF) stimulation of the BAFF receptor (BAFF-R) is essential for the homeostatic survival of mature B cells. Earlier in vitro experiments with inhibitors that block MEK 1 and 2 suggested that activation of ERK 1 and 2 MAP kinases is required for BAFF-R to promote B cell survival. However, these inhibitors are now known to also inhibit MEK5, which activates the related MAP kinase ERK5. In the present study, we demonstrated that BAFF-induced B cell survival was actually independent of ERK1/2 activation but required ERK5 activation. Consistent with this, we showed that conditional deletion of ERK5 in B cells led to a pronounced global reduction in mature B2 B cell numbers, which correlated with impaired survival of ERK5-deficient B cells after BAFF stimulation. ERK5 was required for optimal BAFF up-regulation of Mcl1 and Bcl2a1, which are prosurvival members of the Bcl-2 family. However, ERK5 deficiency did not alter BAFF activation of the PI3-kinase–Akt or NF-κB signaling pathways, which are also important for BAFF to promote mature B cell survival. Our study reveals a critical role for the MEK5-ERK5 MAP kinase signaling pathway in BAFF-induced mature B cell survival and homeostatic maintenance of B2 cell numbers.
Survival of peripheral mature B cells depends on their stimulation via the receptor for the cytokine BAFF (B cell activating factor; Mackay et al., 2010). BAFF-receptor (BAFF-R; TNFRSF13) expression is induced on newly formed immature B cells in the BM and increases in the spleen as transitional B cells mature to become follicular or marginal zone B cells (Mackay et al., 2010). In BAFF or BAFF-R–deficient mice, B cell development is blocked at the transitional T1–T2 developmental step in the spleen, and numbers of mature follicular and marginal zone B cells are substantially reduced (Batten et al., 2000; Yan et al., 2001). In contrast, B1 B cells do not require BAFF for their survival. Transgenic overproduction of BAFF in mice induces the expansion of the B cell compartment and the development of an autoimmune disease resembling systemic lupus erythematosus (SLE; Mackay et al., 1999). The levels of BAFF in vivo therefore control the size of the mature B cell pool.
Stimulation of BAFF-R activates several intracellular signaling pathways that promote cell survival. These include those leading to the activation of NF-κB transcription factors, which induce the expression of pro-survival proteins, such as the Bcl-2 family proteins Bcl-xl and A1 (Rickert et al., 2011). BAFF strongly activates the alternative NF-κB pathway (Razani et al., 2011), which stimulates the partial proteolysis of NF-κB2 p100 to p52 via IκB kinase 1 (IKK1), resulting in nuclear translocation of p52/RelB heterodimers (Beinke and Ley, 2004). However, conditional deletion of Ikk1 in the B cell lineage has recently demonstrated that IKK1 is dispensable for BAFF-induced mature B cell survival and is also not required for the development of a substantial fraction of mature B cells (Jellusova et al., 2013). BAFF also weakly activates the canonical IKK2-regulated NF-κB pathway that stimulates the proteolysis of IκBα, promoting the nuclear translocation of NF-κB1 p50/RelA heterodimers. Mature B cell numbers are substantially reduced by B cell–specific deletion of IKK2 (Pasparakis et al., 2002). Furthermore, expression of constitutively active IKK2 substitutes for BAFF-R deficiency for generation of peripheral mature B cells (Sasaki et al., 2006). BAFF activation of the canonical NF-κB pathway therefore appears to be required for the survival and/or development of mature B cells, while activation of the alternative NF-κB pathway does not appear to be essential.
Phosphatidylinositol (PtdIns) 3-kinase (PI3K) is also activated by BAFF stimulation of mature B cells (Patke et al., 2006) as a result of BAFF-induced phosphorylation of the CD19 co-receptor (Jellusova et al., 2013). Phosphatidylinositide-3,4,5-trisphosphate (PIP3) generated then activates downstream signaling pathways by recruiting effector molecules to the plasma membrane via their PH domains. These include Akt, which has critical roles in cell growth and survival (Baracho et al., 2011). Pharmacological experiments indicate that PI3K activation is required for BAFF-induced survival of B cells in vitro (Henley et al., 2008), and additionally regulates cellular metabolism and growth by activating the mammalian target of rapamycin (mTOR; Patke et al., 2006). Deficiency of PTEN, which encodes a phosphatase that converts PIP3 to phosphatidlyinositide-4,5-bisphosphate and counteracts the activity of PI3 kinases, partially rescues the B cell maturation defect of Baff−/− mice, strongly supporting a key role for PI3K in BAFF-induced B cell survival in vivo (Jellusova et al., 2013). PI3K signaling stimulates B cell survival by up-regulating expression of the pro-survival Bcl-2 family member Mcl-1, which is essential for BAFF-induced B cell survival (Woodland et al., 2008).
ERK1 and ERK2 MAP kinases are critical for pre-BCR induced early B cell development (Yasuda et al., 2008). It has been proposed that ERK1/2 activation is required for optimal BAFF-induced mature B cell survival (Craxton et al., 2005). However, this conclusion was based on experiments with inhibitors of the ERK1/2 kinases MEK 1 and 2 that are now known to be unspecific. In the present study, we show that BAFF-induced B cell survival was actually independent of ERK1/2 activation but required ERK5 activation. Consistent with this, B cell–specific deletion of ERK5 led to a pronounced reduction in mature B2 cell numbers, which correlated with impaired survival of ERK5-deficient B cells after BAFF stimulation. These data identify the ERK5 MAP kinase pathway as a key component in BAFF survival signaling and B cell homeostasis.
RESULTS AND DISCUSSION
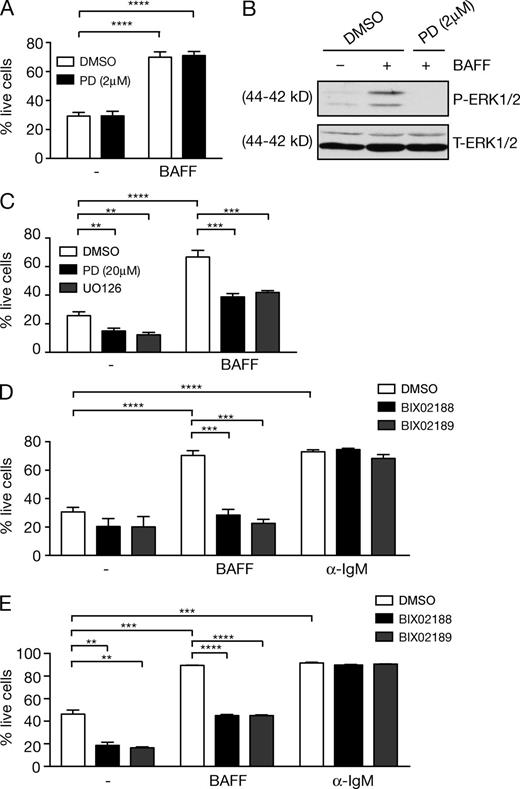
BAFF-induced FM B cell survival is dependent on MEK5 activity
Evidence linking the prosurvival effects of BAFF-R stimulation to the activation of ERK1/2 MAP kinases in B cells (Craxton et al., 2005) is based on the use of the pharmacological inhibitor U0126, which blocks the catalytic activity of the upstream MAP kinase kinases (MAP2K) MEK1 and MEK2 (Bain et al., 2007). However, U0126 also inhibits with similar potency MEK5, a MAP 2-kinase closely related to MEK1 that phosphorylates and activates ERK5 MAP kinase (Nithianandarajah-Jones et al., 2012). It was therefore possible that the inhibitory effect of U0126 on B cell survival was due to blockade of ERK5 activation, rather than ERK1/2.
We reinvestigated the role of ERK1/2 activation in BAFF-induced B cell survival using a second-generation MEK1/2 inhibitor, PD184352, which inhibits MEK1/2 and MEK5 with different potencies (Bain et al., 2007). 2 µM PD184352, a concentration that inhibits MEK1/2 but not MEK5, did not affect BAFF-induced survival of murine splenic follicular mature (FM) B cells (Fig. 1 A and Fig. S1), although BAFF-induced ERK1/2 phosphorylation was clearly blocked (Fig.1 B). However, increasing the concentration of PD184352 to 20 µM, which blocks MEK1/2 and MEK5 activation (Bain et al., 2007), reduced BAFF-induced B cell survival similar to U0126 (Fig. 1 C). These observations raised the possibility that activation of ERK5 might be critical for BAFF-induced mature B cell survival, rather than ERK1/2 activation. Consistent with this hypothesis, we found that two MEK5 inhibitors, BIX02188 and BIX02189 (Tatake et al., 2008), did not inhibit MEK1/2 activity (unpublished data) but reduced BAFF-induced FM B cell survival (Fig. 1 D). BIX02188 and BIX02189 MEK5 inhibitors also reduced BAFF-induced survival of highly purified FACS-sorted FM B cells (Fig. 1 E), ruling out any indirect effects of contaminating non-FM B cells. In contrast, neither MEK5 inhibitor affected BCR-induced B cell survival (Fig. 1, D and E). These results indicated a critical role for activation of the ERK5 MAP kinase pathway in BAFF-induced mature B cell survival, while BCR survival signaling was independent of ERK5 activation.
BAFF-induced FM B cell survival depends on MEK5 activity. (A and B) WT purified splenic FM B cells were cultured for 48 h with BAFF or control medium (–) plus 2 µM PD184352 (PD) or vehicle control (DMSO). (A) The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; pooled cells cultured in triplicates). Gating strategies used are shown in Fig. S1. (B) Lysates of purified splenic FM B cells were immunoblotted for the indicated proteins. (C and D) WT purified splenic FM B cells were cultured for 48 h with BAFF, α-IgM, or control medium (-), in the presence of 20 µM PD184352, 10 µM U0126, 10 µM BIX02188, 10 µM BIX02189, or control DMSO. The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; pooled cells cultured in triplicates). (E) Sorted splenic FM B cells (B220+AA4.1−IgM+CD23+) were cultured for 48 h with BAFF, α-IgM, or control medium (-) plus 5 µM BIX02188, 5 µM BIX02189, or control DMSO. The fraction of live cells was determined as in D. All results are representative of at least three independent experiments. **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
BAFF-induced FM B cell survival depends on MEK5 activity. (A and B) WT purified splenic FM B cells were cultured for 48 h with BAFF or control medium (–) plus 2 µM PD184352 (PD) or vehicle control (DMSO). (A) The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; pooled cells cultured in triplicates). Gating strategies used are shown in Fig. S1. (B) Lysates of purified splenic FM B cells were immunoblotted for the indicated proteins. (C and D) WT purified splenic FM B cells were cultured for 48 h with BAFF, α-IgM, or control medium (-), in the presence of 20 µM PD184352, 10 µM U0126, 10 µM BIX02188, 10 µM BIX02189, or control DMSO. The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; pooled cells cultured in triplicates). (E) Sorted splenic FM B cells (B220+AA4.1−IgM+CD23+) were cultured for 48 h with BAFF, α-IgM, or control medium (-) plus 5 µM BIX02188, 5 µM BIX02189, or control DMSO. The fraction of live cells was determined as in D. All results are representative of at least three independent experiments. **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
B cell–specific ERK5 deletion reduces B2 B cell numbers
It was important to confirm the role for ERK5 signaling in B cell survival genetically, as pharmacological inhibitors can have off-target effects. Since an Erk5-null mutation results in embryonic lethality (Nithianandarajah-Jones et al., 2012), we crossed mice harboring a conditional Erk5 allele (erk5fl/fl; Wang et al., 2006) to mb1-cre+ mice that express Cre at the pro–B cell stage in the BM (Hobeika et al., 2006) to generate mice with ERK5-deficient B cells. Efficient depletion of ERK5 protein in splenic mature B cells from mb1-cre+ erk5fl/fl mice was confirmed by immunoblotting (Fig. 2 A).
B cell–specific deletion of ERK5 reduces B2 cell numbers. (A) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were analyzed for ERK5 expression by immunoblotting. (B–F) Flow cytometric analysis of B cell populations in mb1-cre+ erk5wt/wt and mb1-cre+ erk5fl/fl mice from the indicated organs, as shown in Fig. S2. (B) Absolute numbers of total B cells (CD19+B220+), pro–B (B220+CD19+IgD−IgM−CD2−), pre-B (B220+CD19+IgD−IgM−CD2+), immature B (B220+CD19+IgD−IgM+CD2+), and mature B (B220+CD19+IgD+IgM+CD2+) cells in the BM (mean ± SEM; n = 7 mice/genotype) were quantified. (C) Absolute splenic numbers (mean ± SEM; n = 14 mice/genotype) of total B cells (IgM+ or IgD+), immature B cells (B220+AA4.1+), separated into transitional T1 B cells (IgMhiCD23−) and T2 B cells (IgMhiCD23+) were quantified. Splenic mature B cells (B220+AA4.1−), separated into FM B cells (IgM+CD23+) and MZ B cells (IgMhiCD23−). (D) Absolute numbers (mean ± SEM; n = 14 mice/genotype) of B cells (IgM+CD19+) in peripheral LN (pools of single cervical, axillary, and inguinal nodes; mean ± SEM; n = 14 mice/genotype) were quantified. (E) Proportion of B2 (B220+CD19+CD5−CD23+) cells in the peritoneal cavity (mean ± SEM; n = 5 mice/genotype) was quantified. (F) mb1-cre+ erk5wt/wt or mb1-cre+ erk5fl/fl Ly5.2+ BM cells were mixed with WT Ly5.1+ BM cells at the indicated ratios, and transferred into sublethally irradiated Rag1−/− mice. After 8-wk reconstitution, absolute numbers of Ly5.2+ B2 B cells from LN and spleen were assessed by flow cytometry. Graphs show B2 B cell absolute cell numbers (mean ± SEM; n = 8 independent mice/genotype). Numbers below the graphs represents the ratio between WT Ly5.2+ controls compared to ERK5-deficient B cells. In A–F, results are representative of at least two independent experiments. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
B cell–specific deletion of ERK5 reduces B2 cell numbers. (A) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were analyzed for ERK5 expression by immunoblotting. (B–F) Flow cytometric analysis of B cell populations in mb1-cre+ erk5wt/wt and mb1-cre+ erk5fl/fl mice from the indicated organs, as shown in Fig. S2. (B) Absolute numbers of total B cells (CD19+B220+), pro–B (B220+CD19+IgD−IgM−CD2−), pre-B (B220+CD19+IgD−IgM−CD2+), immature B (B220+CD19+IgD−IgM+CD2+), and mature B (B220+CD19+IgD+IgM+CD2+) cells in the BM (mean ± SEM; n = 7 mice/genotype) were quantified. (C) Absolute splenic numbers (mean ± SEM; n = 14 mice/genotype) of total B cells (IgM+ or IgD+), immature B cells (B220+AA4.1+), separated into transitional T1 B cells (IgMhiCD23−) and T2 B cells (IgMhiCD23+) were quantified. Splenic mature B cells (B220+AA4.1−), separated into FM B cells (IgM+CD23+) and MZ B cells (IgMhiCD23−). (D) Absolute numbers (mean ± SEM; n = 14 mice/genotype) of B cells (IgM+CD19+) in peripheral LN (pools of single cervical, axillary, and inguinal nodes; mean ± SEM; n = 14 mice/genotype) were quantified. (E) Proportion of B2 (B220+CD19+CD5−CD23+) cells in the peritoneal cavity (mean ± SEM; n = 5 mice/genotype) was quantified. (F) mb1-cre+ erk5wt/wt or mb1-cre+ erk5fl/fl Ly5.2+ BM cells were mixed with WT Ly5.1+ BM cells at the indicated ratios, and transferred into sublethally irradiated Rag1−/− mice. After 8-wk reconstitution, absolute numbers of Ly5.2+ B2 B cells from LN and spleen were assessed by flow cytometry. Graphs show B2 B cell absolute cell numbers (mean ± SEM; n = 8 independent mice/genotype). Numbers below the graphs represents the ratio between WT Ly5.2+ controls compared to ERK5-deficient B cells. In A–F, results are representative of at least two independent experiments. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
B cell development in the BM was similar between mb1-cre+ erk5fl/fl and mb1-cre+ erk5wt/wt mice, with similar absolute numbers of pro–B cells, pre–B cells, and immature B cells (Fig. 2 B and Fig. S2). Total numbers of B cells in spleen were also equivalent in ERK5-deficient and control mice (Fig. 2 C), as were the number of splenic transitional type 2 (T2) B cells. Numbers of splenic T1 and marginal zone (MZ) B cells were both fractionally, but significantly, increased by ERK5 absence. In contrast, there was approximately a 40% reduction in the number of FM B cells in the spleen in ERK5-deficient mice compared to controls. The numbers of mature B2 cells in the BM (Fig. 2 B) and in peripheral LN (Fig. 2 D), as well as the proportion of B2 cells in the peritoneal cavity (Fig. 2 E), were also decreased by ERK5 deficiency. In contrast, the fraction of peritoneal B1 cells, the survival of which is not regulated by BAFF (Mackay et al., 2010), was unaffected by ERK5 absence (unpublished data). These results indicated that ERK5 was required for optimal development and/or homeostasis of mature B2 B cells, consistent with a role for ERK5 in BAFF-induced B2 cell survival. However, ERK5 was not needed for the generation of T2 B cells, in contrast to BAFF and BAFF-R (Mackay et al., 2010), indicating that ERK5 signaling was dispensable for the BAFF-induced development of transitional B cells.
Mixed BM chimeras were generated to determine whether the effect of ERK5 deficiency on the generation of mature B2 cells was due to a cell-intrinsic defect or could be rescued by WT B cells. The hematopoietic system of irradiated Rag1−/− mice was reconstituted with mb1-cre+ erk5fl/fl or mb1-cre+ erk5wt/wt BM cells (Ly5.2+) alone, or mixed with BM from WT (Ly5.1+) mice at the ratio 80:20 (Ly5.1/Ly5.2). The numbers of B2 cells in LN was reduced by ERK5 deficiency in mice reconstituted with BM from only mb1-cre+ erk5fl/fl compared to BM from mb1-cre+ erk5wt/wt mice (Fig. 2 F), similar to intact donor mice. Competition with WT Ly5.1+ B cells resulted in a larger reduction in the numbers of ERK5-deficient Ly5.2+ B2 cells in LN compared to WT Ly5.2+ B2 cells. In the spleen, WT Ly5.1+ B cells similarly decreased the fraction of ERK5-deficient Ly5.2+ FM B cell numbers compared to WT Ly5.2+ FM B cells, while the fraction of ERK5-deficient Ly5.2+ MZ B cells was normalized. These data showed that the reduction in B2 B cell numbers observed in mb1-cre+ erk5fl/fl mice was the consequence of a cell-intrinsic defect, and suggested that ERK5-deficient Ly5.2+ B2 cells could not compete with WT Ly5.1+ B2 cells for a limiting environmental resource. In contrast, the increase in MZ B cell numbers in intact mb1-cre+ erk5fl/fl mice was not caused by a cell-intrinsic ERK5 signaling defect. It is possible that the reduction in FM B cell numbers in such mice promotes the differentiation of T2 B cells to MZ B cells due to an increase in the local availability of BAFF.
ERK5 signaling in B cells is dispensable for antibody responses
After establishing that ERK5 expression was required to maintain normal B2 B cell numbers, we tested whether ERK5 deficiency impaired B cell antibody responses. ERK5 deficiency did not affect the production of serum anti-NP IgM and IgG1 following immunization with the T-dependent (TD) antigen NP27-CGG precipitated in alum (Fig. 3 A). The generation of anti-NP IgG1+–specific germinal center B cells was also equivalent in the presence and absence of ERK5 expression in B cells. Antibody responses of mb1-cre+ erk5fl/fl and mb1-cre+ erk5wt/wt mice to a type-1 T-independent (TI) antigen (TNP-LPS) and a type-2 TI antigen (TNP-Ficoll) were also tested. The amounts of TNP-specific IgM, IgG2b, IgG2c, and IgG3 antibodies detected in sera after TNP-LPS injection were the same in both genotypes (Fig. 3 B and not depicted). Similarly, the amounts of anti-TNP IgM, IgG1, IgG2b, IgG2c, and IgG3 antibodies detected in sera after TNP-Ficoll immunization were unaffected by ERK5 deficiency (Fig. 3 C and not depicted). ERK5 therefore was not required for B cell antibody responses to TD or TI antigens. This suggested that BCR-induced clonal expansion in response to TD antigens compensated for the fractional reduction in steady-state B2 cell numbers caused by ERK5 absence. Consistent with this hypothesis, ERK5 was not required for BCR-induced FM B cell growth or proliferation in vitro (unpublished data).
ERK5 is dispensable for B cell antibody responses to TD and TI antigens. (A) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were immunized with NP27-CGG alum or PBS alum controls. α-NP IgM (left) and α-NP IgG1 (middle) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units), 14 days after NP27-CGG immunization. The right panel represents splenic α−NP IgG1+ GC B cells 14d after NP27-CGG immunization (mean absolute number ± SEM). (B and C) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were immunized with TNP-LPS (B) or TNP-Ficoll (C) and bled at the indicated time points. (B) α-TNP IgM (left), α-TNP IgG3 (middle) and α-TNP IgG2b (right) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units). (C) α-TNP IgM (left), α-TNP IgG1 (middle), and α-TNP IgG2b (right) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units). All results are representative of two independent experiments (at least four independent mice per genotype each).
ERK5 is dispensable for B cell antibody responses to TD and TI antigens. (A) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were immunized with NP27-CGG alum or PBS alum controls. α-NP IgM (left) and α-NP IgG1 (middle) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units), 14 days after NP27-CGG immunization. The right panel represents splenic α−NP IgG1+ GC B cells 14d after NP27-CGG immunization (mean absolute number ± SEM). (B and C) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were immunized with TNP-LPS (B) or TNP-Ficoll (C) and bled at the indicated time points. (B) α-TNP IgM (left), α-TNP IgG3 (middle) and α-TNP IgG2b (right) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units). (C) α-TNP IgM (left), α-TNP IgG1 (middle), and α-TNP IgG2b (right) serum antibody levels were quantified by ELISA (mean ± SEM; a.u., arbitrary units). All results are representative of two independent experiments (at least four independent mice per genotype each).
ERK5 is required for BAFF-induced B cell survival
We next investigated the effect of ERK5 deficiency on B cell survival in vitro, focusing on FM B2 B cells whose numbers were regulated by cell-intrinsic ERK5 signaling. BAFF-induced survival of ERK5-deficient FM B cells was substantially reduced compared to WT controls (Fig. 4 A), which did not result from decreased BAFF-R expression (Fig. 4 B). Together with MEK5 inhibitor experiments, these results suggested that BAFF stimulation of BAFF-R activated the MEK5–ERK5 signaling pathway and that this was required for optimal mature B cell survival in vitro. Consistent with this hypothesis, overnight BAFF stimulation of FM B cells induced a phosphorylation shift of ERK5 on SDS-PAGE (Fig. 4 C) that was blocked by inhibition of MEK5 but not MEK1/2 kinase activity (Fig. 4, D and E). In contrast, BCR stimulation did not promote a detectable ERK5 phosphorylation shift (Fig. 4 F) and anti-IgM–induced FM B cell survival was independent of ERK5 expression (Fig. 4 A). BAFF-induced phosphorylation of ERK5 was inhibited with an antibody to BAFF-R (Fig. 4 G). This indicated that BAFF stimulated ERK5 phosphorylation via BAFF-R and not TACI (transmembrane activator CAML interactor), a BAFF receptor that negatively regulates B cell survival and is also expressed on FM B cells (Mackay et al., 2010).
BAFF-induced FM B cell survival requires ERK5. (A) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were cultured for 48 h with BAFF, α-IgM, BAFF plus α-IgM, or control medium (-). The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; n = 3 mice/genotype; triplicate cultures). Results are representative of at least three separate experiments. (B) BAFF-R protein levels on gated AA4.1-B220+IgM+CD23+ FM B cells of the indicated genotypes were analyzed directly ex vivo by flow cytometry. T cells from mb1-cre+ erk5wt/wt mice were used as a negative control. (C) Purified splenic FM B cells were cultured for 16 h ± BAFF. Immunoprecipitated ERK5 from whole-cell lysates was incubated ± λ phosphatase and analyzed by immunoblotting. (D and E) Pooled WT purified splenic FM B cells were cultured for 16 h with BAFF or control medium (-) plus vehicle control (DMSO), 10 µM BIX02188, 10 µM BIX02189, or 2 µM PD184352 (PD). Cell lysates were immunoblotted. (F) Total lysates of purified splenic FM B cells stimulated with α-IgM were immunoblotted. (G) Pooled WT purified splenic FM B cells were cultured for 16 h with BAFF or control medium (-) plus BAFF-R blocking antibody or isotype control. Total cell lysates were immunoblotted. All results are representative of two or more independent experiments. ****, P < 0.0001.
BAFF-induced FM B cell survival requires ERK5. (A) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were cultured for 48 h with BAFF, α-IgM, BAFF plus α-IgM, or control medium (-). The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; n = 3 mice/genotype; triplicate cultures). Results are representative of at least three separate experiments. (B) BAFF-R protein levels on gated AA4.1-B220+IgM+CD23+ FM B cells of the indicated genotypes were analyzed directly ex vivo by flow cytometry. T cells from mb1-cre+ erk5wt/wt mice were used as a negative control. (C) Purified splenic FM B cells were cultured for 16 h ± BAFF. Immunoprecipitated ERK5 from whole-cell lysates was incubated ± λ phosphatase and analyzed by immunoblotting. (D and E) Pooled WT purified splenic FM B cells were cultured for 16 h with BAFF or control medium (-) plus vehicle control (DMSO), 10 µM BIX02188, 10 µM BIX02189, or 2 µM PD184352 (PD). Cell lysates were immunoblotted. (F) Total lysates of purified splenic FM B cells stimulated with α-IgM were immunoblotted. (G) Pooled WT purified splenic FM B cells were cultured for 16 h with BAFF or control medium (-) plus BAFF-R blocking antibody or isotype control. Total cell lysates were immunoblotted. All results are representative of two or more independent experiments. ****, P < 0.0001.
It has been proposed that BAFF induces B cell survival via ERK1/2 induced phosphorylation and subsequent degradation of the proapoptotic protein Bim (Craxton et al., 2005). However, BAFF stimulation did not alter the abundance of Bim in either mb1-cre+ erk5wt/wt or mb1-cre+ erk5fl/fl FM B cells (Fig. 5 A), suggesting that ERK5 did not promote B cell survival by regulating Bim levels. BAFF stimulation of B cells also induces expression of Mcl1, an antiapoptotic member of the Bcl2 protein family that is critical for BAFF-induced B cell survival (Woodland et al., 2008). BAFF up-regulation of Mcl1 protein levels in FM B cells was reduced by ERK5 deficiency (Fig. 5 A). BAFF controls Mcl1 protein levels in part by the Akt-dependent inactivation of Gsk-3β, which constitutively phosphorylates and induces the proteolysis of Mcl1 in unstimulated cells (Maurer et al., 2006). However, BAFF induced phosphorylation of Akt (Fig. 5 A) to a similar degree in mb1-cre+ erk5fl/fl and mb1-cre+ erk5wt/wt FM B cells. ERK5 therefore regulated Mcl1 expression levels separately from BAFF-induced activation of the PI3K-Akt signaling pathway. BAFF activation of the alternative NF-κB2 pathway (Fig. 5 A), assaying levels of NF-κB2 p52, and the canonical NF-κB1 pathway (Fig. 5 B), assaying IKK2 phosphorylation of NF-κB1 p105 (Lang et al., 2003), were also unaffected by ERK5 deficiency.
Optimal BAFF induction of Mcl1 and A1 expression requires ERK5. (A and B) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were cultured with BAFF or control medium (-) for 16 h (A) or 1 h (B). Whole-cell lysates were immunoblotted for the indicated proteins. Results are representative of three independent experiments (3 mice/genotype each). In A, graphs represent mean band intensities for the indicated proteins (±SEM; n ≥ 5/genotype each), normalized to tubulin loading control. In B, quantitation of band intensities (6 mice/genotype) demonstrated that ERK5 deficiency did not significantly alter P-p105 levels in BAFF-stimulated cells (data not shown). (C) mRNA levels of the indicated genes from rested purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were determined by quantitative RT-PCR. Data were normalized to Hprt1 mRNA and are represented as mean fold change (±SEM) relative to unstimulated WT cells. (D) Purified splenic FM B cells from the indicated mouse strains were cultured in duplicate with BAFF for 16 h and mRNA expression was assessed as indicated in C. (E) Purified splenic FM B cells from the indicated mouse strains were cultured for 48 h with BAFF or control medium (-) plus PI-103 (PI; 0.5 µM), GDC-0941 (GDC; 0.5 µM), or vehicle control (DMSO). The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; triplicates). In C–E, results are representative of three separate experiments (three mice/genotype each). (F) WT mice were injected intraperitoneally twice (day 1 and 3) with 100 µg anti-BAFF antibody or isotype control. At day 4, splenic FM B cells were purified and cell lysates immunoblotted. Results are representative of two independent experiments (4 mice/genotype each). Graphs (right) represent mean band intensity (±SEM; n = 7/genotype each), normalized to tubulin loading control. (G) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were injected intraperitoneally with BrdU daily for 5 days. Spleens were harvested at day 6 and BrdU+ cells assayed by flow cytometry. Results are representative of two independent experiments (five mice/genotype each). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
Optimal BAFF induction of Mcl1 and A1 expression requires ERK5. (A and B) Purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were cultured with BAFF or control medium (-) for 16 h (A) or 1 h (B). Whole-cell lysates were immunoblotted for the indicated proteins. Results are representative of three independent experiments (3 mice/genotype each). In A, graphs represent mean band intensities for the indicated proteins (±SEM; n ≥ 5/genotype each), normalized to tubulin loading control. In B, quantitation of band intensities (6 mice/genotype) demonstrated that ERK5 deficiency did not significantly alter P-p105 levels in BAFF-stimulated cells (data not shown). (C) mRNA levels of the indicated genes from rested purified splenic FM B cells from mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were determined by quantitative RT-PCR. Data were normalized to Hprt1 mRNA and are represented as mean fold change (±SEM) relative to unstimulated WT cells. (D) Purified splenic FM B cells from the indicated mouse strains were cultured in duplicate with BAFF for 16 h and mRNA expression was assessed as indicated in C. (E) Purified splenic FM B cells from the indicated mouse strains were cultured for 48 h with BAFF or control medium (-) plus PI-103 (PI; 0.5 µM), GDC-0941 (GDC; 0.5 µM), or vehicle control (DMSO). The fraction of live FM B cells was determined by flow cytometric analysis of 7AAD staining (mean ± SEM; triplicates). In C–E, results are representative of three separate experiments (three mice/genotype each). (F) WT mice were injected intraperitoneally twice (day 1 and 3) with 100 µg anti-BAFF antibody or isotype control. At day 4, splenic FM B cells were purified and cell lysates immunoblotted. Results are representative of two independent experiments (4 mice/genotype each). Graphs (right) represent mean band intensity (±SEM; n = 7/genotype each), normalized to tubulin loading control. (G) mb1-cre+ erk5fl/fl mice and mb1-cre+ erk5wt/wt control mice were injected intraperitoneally with BrdU daily for 5 days. Spleens were harvested at day 6 and BrdU+ cells assayed by flow cytometry. Results are representative of two independent experiments (five mice/genotype each). *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
Mcl1 mRNA expression in ex vivo FM B cells was decreased by approximately 50% by the absence of ERK5 (Fig. 5 C). Furthermore, BAFF induction of Mcl1 mRNA in cultured FM B cells was substantially reduced by ERK5 absence (Fig. 5 D). ERK5 deficiency also reduced ex vivo expression of Bcl2a1 mRNA (encoding the pro-survival Bcl2 family protein A1) and BAFF-induced Bcl2a1 mRNA (Fig. 5, C and D) and A1 protein expression (Fig. 5 A). mRNA concentrations of Bcl2 and Bcl-xl ex vivo and after BAFF stimulation were not unaffected by ERK5 deficiency (unpublished data). These data suggested that ERK5 was required for optimal transcriptional up-regulation of Mcl1 and Bcl2a1 after BAFF stimulation, providing a possible explanation for the reduced survival of ERK5-deficient B cells compared to WT.
Activation of the PI3K–Akt pathway is required for BAFF-R survival signaling (Rickert et al., 2011). Because BAFF-R induced Akt phosphorylation independently of ERK5 expression, we tested the effect of blocking PI3K-Akt and ERK5 signaling pathways simultaneously on BAFF-induced FM B cell survival in vitro. Pretreatment of cells with the small molecule PI3K inhibitors PI-103 (Bain et al., 2007) or GDC-0941 (Folkes et al., 2008) reduced the survival of FM B cells after BAFF stimulation to a similar degree to that caused by genetic deletion of ERK5 (Fig. 5 E; ∼40% live cells). However, treatment of ERK5-deficient mb1-cre+ erk5fl/fl FM B cells with either PI-103 or GDC-0941 reduced the fraction of live cells to <10% in BAFF-containing cultures. The PI3K-Akt and ERK5 signaling pathways therefore function cooperatively to promote the survival of FM B cells.
Antibody blockade of BAFF leads to the rapid loss of mature B cells in vivo, indicating that BAFF stimulation is continuously required to promote mature B cell survival (Mackay et al., 2010). To investigate whether tonic BAFF stimulation activates the ERK5 signaling pathway in mature B cells, WT C57BL/6 mice were injected intraperitoneally twice (day 1 and 3) with neutralizing BAFF antibody or isotype control antibody, and culled at d 4. Purified FM B cells were immediately lysed, and extracted proteins were immunoblotted for ERK5. In mice injected with control antibody, a slower mobility phosphorylated form of ERK5 was clearly evident, but this was lost after anti-BAFF injection (Fig. 5 F). These results suggested that tonic BAFF stimulation of FM B cells continuously induced ERK5 phosphorylation and might be required to maintain FM B cell survival in vivo.
To determine whether ERK5 regulated FM B cell turnover, mb1-cre+ erk5fl/fl and mb1-cre+ erk5wt/wt mice were injected daily with BrdU for 5 d. Spleens were harvested at day 6 and BrdU labeling in splenic B cell subsets was determined by flow cytometry (Fig. 5 G). ERK5 deficiency had no effect on BrdU labeling of T1 or MZ B cells, whereas there was a small increase in the fraction of BrdU+ T2 cells. In contrast, the fraction of BrdU+ FM B cells was increased 2.5-fold in the absence of ERK5. 7-AAD labeling demonstrated that ERK5 deficiency did not alter the proportion of S-phase cells at any stage of B cell development in the BM or spleen (unpublished data). These results suggested that FM B cells turned over more rapidly in the absence of ERK5 expression, consistent with a key role for ERK5 signaling in maintaining their survival.
Our study has established a critical role for the MEK5–ERK5 signaling in BAFF-induced mature B cell survival, providing an explanation for the reduction in B2 cell numbers in mice lacking ERK5 expression in their B cell lineage. Recent data have indicated that the alternative NF-κB2 signaling pathway may be dispensable for BAFF-induced mature B cell survival (Jellusova et al., 2013). In contrast, pharmacological and genetic experiments demonstrate an important role for BAFF activation of PI3K to promote the survival of peripheral B cells (Patke et al., 2006; Jellusova et al., 2013). BAFF stimulation of Akt phosphorylation was independent of ERK5 activation, suggesting that ERK5 and PI3K signaling independently regulate BAFF-induced mature B cell survival. However, the outputs of these signaling pathways converge on Mcl1, which is required for BAFF-induced B cell survival (Woodland et al., 2008; Jellusova et al., 2013). ERK5 signaling induces Mcl1 transcription, whereas PI3K–Akt signaling stabilizes Mcl1 protein via inhibition of GSK-3β. In line with this, experiments with PI3K inhibitors revealed that PI3K–Akt and ERK5 signaling cooperate to promote BAFF-induced B cell survival.
In addition to BAFF stimulation, the survival of peripheral B cells depends on a tonic signal from the BCR that is distinct from that induced by antigen binding (Mackay et al., 2010). Activation of PI3K or inactivation of its inhibitor PTEN rescues the survival of BCR-deficient B cells, suggesting that the BCR promotes mature B cell survival via activation of PI3K (Srinivasan et al., 2009). BCR expression is needed to maintain peripheral B cells due to the BAFF-R requiring Syk tyrosine kinase, a key signal transducer from the BCR, to activate PI3K and promote B cell survival (Schweighoffer et al., 2013). However, BCR-deficient PI3K-rescued B cells are still dependent on BAFF for survival in vitro (Srinivasan et al., 2009). Our data raise the possibility that BAFF stimulation of such cells is needed to activate the ERK5 MAP kinase pathway, which is not required for BCR-induced survival signaling.
MATERIAL AND METHODS
Mouse strains.
Mouse strains were bred in the specific pathogen–free animal facility of the National Institute for Medical Research (NIMR). 7-12-wk–old mice were used for all experiments, which were performed in accordance with the UK Home Office regulations and approved by the NIMR Ethical Review Panel. Rag1−/− (Mombaerts et al., 1992), erk5fl/fl (Wang et al., 2006), and mb1-cre+ (Hobeika et al., 2006) mouse strains were all fully backcrossed onto a C57BL/6 background.
BM chimeras.
BM cells were harvested from femora and tibiae of the indicated donor mice, and resuspended in ACK lysis buffer (155 mM NH4Cl, 10 mM KHCO3, and 100 µM EDTA) to lyse red blood cells. Cells (4-5 × 106 per mouse) were then transferred by intravenous injection into sublethally irradiated Rag1−/− mice (500 rad; Cs source). For competition experiments, BM cell suspensions from CD45.1+ C57BL/6 were mixed with either mb1-cre+erk5fl/fl or mb1-cre+erk5wt/wt CD45.2+ BM cells at a 80:20 ratio. Chimeric mice were analyzed 8 wk after reconstitution.
Antibody responses.
For TD antibody responses, experiments were performed as previously described (Jacque et al., 2014). For TI antibody responses, mice were immunized intraperitoneally with 50 µg of either TNP-LPS (T1) or TNP-Ficoll (T2) in PBS (Biosearch Technologies). Mice were bled before immunization and at day 7, 14, and 21 after immunization. The production of TNP-specific immunoglobulins was monitored by ELISA, using 96-well Maxisorp ELISA plates pretreated with 50 µl/well of 33 µg/ml TNP4-BSA (Biosearch Technologies). Antibody isotypes and subclasses were detected by ELISA (SouthernBiotech).
In vivo BAFF blockade.
Mice were injected intraperitoneally twice (day 1 and 3) with 100 µg anti-BAFF antibody (10F4; GlaxoSmithKline) or isotype control. At day 4, mice were culled and splenic FM B cells were purified.
BrdU labeling.
BrdU (Sigma-Aldrich, 1 mg in 200 µl PBS) was injected intraperitoneally daily for 5 d. At day 6, mice were culled and spleens were harvested. BrdU+ cells were assayed by flow cytometry using the FastImmune anti–BrdU-FITC antibody (BD).
Flow cytometry.
Single-cell suspensions were generated from peripheral LN (pools of single cervical, axillary, and inguinal nodes), spleen, BM, and peritoneal washes by gentle homogenization through nylon mesh filters (70 µM; BD). Erythrocytes in BM and spleens were lysed with ACK lysis buffer before staining, and cell concentrations determined using a Casy Counter (Scharfe Instrument Systems). CD1d-PE and CD1d-bio (clone 1B1), CD21-FITC (eBio8D9), CD23-PE (B3B4), B220-FITC, B220-PE, B220-APC and B220-eFluor450 (RA3-6B2), CD93-APC (AA4.1), IgM-PECy7 (II/41), IgD-FITC, IgD-eFluor450, IgD-PE and IgD-bio (11-26), Ly5.1-FITC and Ly5.1-APC (A20), Ly5.2-FITC and Ly5.2-APC-eFluor780 (104), CD43-bio (eBioR2/60), IgG1-APC (X56), Gr1-bio (RB6-8C5), CD11b-Bio (M1/70), CD11c-Bio (N418), CD5-PE (53-7.3), CD19-FITC (MB19-1), TCRβ-FITC (H57-597), and BAFF-R-FITC (eBio7H22-E16) were obtained from eBioscience. CD4-PerCP (RM4-5), B220-PcP (RA3-6B2), B220-BV605 (RA3-6B2), CD19-BV786 (6D5), and CD8-PerCP (53-6.7) were purchased from BioLegend; CD2-PE (RM2-5), CD9-bio (KMC8), CD21-PE (7G6), Streptavidin-PerCP (554064), and GL7-FITC (Ly77) from BD; PNA-Bio (FL-1075) from Vector Laboratories; CD19-APC (RM705) and CD23-APC (MCD2305) from Life Technologies; and CD93-PE (REA298) from Miltenyi Biotec. For thymus-dependent (TD) antibody response assays, NP27-BSA (Biosearch Technologies) was labeled with the LYNX rapid RPE antibody conjugation kits (AbD Serotec), according to the manufacturer’s protocol. Dead cells were labeled with the Live/Dead Fixable Near-IR Dead Cell Stain kit (Life Technologies) or Zombie Aqua Fixable Viability kit (BioLegend) before antibody staining.
Stained cells were analyzed by flow cytometry on a CyAn ADP Analyzer (Beckman Coulter), FACS LSRFORTESSA X20 Analyzer (BD), or FACSCANTO II Analyzer (BD). Data analyses were performed with FlowJo 887 software (Tree Star).
B cell isolation and in vitro culture.
Splenic FM B cells were isolated by sequential hypotonic RBC lysis, followed by magnetic bead separation using a mixture of biotinylated anti-CD43 and anti-CD1d antibodies and Streptavidin-coupled Dynabeads (Life Technologies). Purity of the resulting FM B cell population (B220+CD23hiCD21lo) was >96–98% as assessed by flow cytometry. Where indicated, FM B cells were fractionated by flow cytometry (B220+AA4.1−IgM+CD23+), using MoFlo XDP (Beckman Coulter) and FACSARIA II cell sorters, to achieve >99% purity. Cells were cultured at a density of 2–5 × 106/mL in complete medium (DMEM plus 10% heat inactivated FCS [Lonza], 100 U/ml Penicillin, 100 µg/ml Streptomycin, 100 µM nonessential amino acids, 100 mM Na-Pyruvate, and 50 µM 2-mercaptoethanol), and stimulated with anti-IgM (10 µg/ml; Jackson ImmunoResearch Laboratories), or BAFF (200 ng/ml; PeproTech).
For in vitro survival assays, 3 × 105 B cells were cultured in a 96-well tissue-culture plate in complete medium (200 µl) ± stimulation (see above). Cells were harvested 2 d later, stained for a B cell marker (B220 or CD19) and 7-amino-actinomycin D (7-AAD; Sigma-Aldrich). Cells were preincubated with pharmacological inhibitors or DMSO vehicle control for 30 min before stimulation. MEK inhibitors (PD184352, U0126, BIX02188, and BIX02189) were purchased from Selleckchem, PI-103 PI3K inhibitor from BioVision, and GDC-0941 PI3K inhibitor from SYNkinase. Mouse BAFF-R blocking antibody (R&D Systems; 5 µg/ml) or isotype control (R&D Systems) were added to cultures simultaneously with BAFF. Cells were harvested and lysed 16 h later.
Protein analyses.
For immunoblots of total lysates, FM B cells (1 × 107 cells per point) were lysed in RIPA buffer (50 mM Tris, pH 7.5, 150 mM NaCl, 2 mM EDTA, 50 mM NaF, 2 mM Na4P2O7, 5 mM dithiothreitol, 1 mM Na3VO4, protease inhibitors [Roche], 0.5% deoxycholate, 1% Triton X-100, and 0.1% SDS). After centrifugation, lysates (10–15 µg) were mixed with an equal volume of 2× SDS-PAGE sample buffer. For in vitro dephosphorylation of ERK5, cells were lysed in S-buffer (10 mM Tris, pH 7.5, 150 mM NaCl, 5 mM EDTA, 1% Triton X-100, and protease inhibitors [Roche] with or without phosphatase inhibitors [10 mM Na3VO4 and 50 mM NaF]). After overnight immunoprecipitation, beads were washed and incubated with 400 U Lambda phosphatase (New England Biolabs) following the manufacturer’s instruction (30°C for 30 min). Reactions were stopped by addition of Laemmli sample buffer and heating to 100°C. Isolated proteins were then resolved by SDS-PAGE and immunoblotted. Band intensities were quantified using a GS-800 Calibrated Densitometer (Bio-Rad Laboratories).
ERK5, ERK1/2, P-ERK1/2, P-Akt (Ser473), Akt, Foxo1, P-NF-κB1 p105, and Bim antibodies were purchased from Cell Signaling Technology, while Mcl1 antibody was from Rockland Immunochemistry. NF-κB1 antibody was purchased from Delta Biolabs and NF-κB2 antibody has been previously described (Coope et al., 2002). A1 antibody was provided by M. Herald (Walter and Eliza Hall Institute of Medical Research [WEHI], Melbourne, Australia) and tubulin mAb by K. Gull (University of Oxford, Oxford, England, UK).
Real-time quantitative PCR.
RNA from stimulated and unstimulated FM B cells (5 × 106/ml) was isolated using the RNeasy kit. Contaminating DNA was removed using RNase-free DNase set (QIAGEN) according to manufacturer’s instructions and by DNase treatment (Roche). cDNA was produced using the SuperScript III First-Strand Synthesis SuperMix for qRT-PCR (Life Technologies) and standard protocols. Expression of target genes were determined by real-time PCR using a Perkin Elmer ABI Prism 7000 Sequence Detection System, and commercial FAM labeled probes (Applied Biosystems) with the TaqMan Gene Expression Master Mix (Applied Biosystems). Target gene mRNA levels were normalized against Hprt mRNA levels.
Statistical analysis.
All data analyses were performed using GraphPad software (GraphPad Software Inc.). In vitro data were compared using Student’s t-test (two tailed and unpaired test). For in vivo experiments, all statistical comparisons were carried out using the nonparametric two-tailed Mann-Whitney test. Statistically significant differences are indicated on the figures. *, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001.
Online supplemental material.
Acknowledgments
We thank the NIMR Photographics department, NIMR Biological Services, NIMR flow cytometry service and other members of the Ley laboratory for help during the course of this work. We are also grateful to Simon Arthur and Philip Cohen (University of Dundee) and Fabienne Mackay (Monash University, Melbourne, Australia) for advice, Cathy Tournier (University of Manchester) for providing the erk5fl/fl mice, Michael Reth (Freiburg, Germany) for the mb1-cre+ mice, Marco Herald (WEHI, Melbourne, Australia) for A1 antibody, and GlaxoSmithKline for 10F4 BAFF antibody.
This work was funded by the UK Medical Research Council, a Leukaemia and Lymphoma Research Project Grant (LRF:06050), and an award from the Bettencourt-Schueller Foundation to E. Jacque.
The authors declare no competing financial interests.
References
- BAFF
B cell activating factor
- BAFF-R
BAFF receptor
- ERK
extracellular signal-regulated kinase
- FM
follicular mature
- GC
germinal center
- Ig
immunoglobulin
- IκB
inhibitor of NF-κB
- IKK
IκB kinase
- IP
immunoprecipitation
- MAPK
mitogen-activated protein kinase
- MEK
MAPK ERK kinase
- MZ
marginal zone
- NP-CGG
4-Hydroxy-3-nitrophenylacetyl-chicken gammaglobulin
- PEC
peritoneal cavity
- PI3K
Phosphatidylinositol 3-kinase
- PIP3
Phosphatidylinositide-3,4,5-trisphosphate
- TD
T dependent
- TI
T independent
- TLR
Toll-like receptor
- TACI
transmembrane activator CAML interactor