T cell acute lymphoblastic leukemia (T-ALL) is an aggressive cancer of immature T cells that often shows aberrant activation of Notch1 and PI3K–Akt pathways. Although mutations that activate PI3K–Akt signaling have previously been identified, the relative contribution of growth factor-dependent activation is unclear. We show here that pharmacologic inhibition or genetic deletion of insulin-like growth factor 1 receptor (IGF1R) blocks the growth and viability of T-ALL cells, whereas moderate diminution of IGF1R signaling compromises leukemia-initiating cell (LIC) activity as defined by transplantability in syngeneic/congenic secondary recipients. Furthermore, IGF1R is a Notch1 target, and Notch1 signaling is required to maintain IGF1R expression at high levels in T-ALL cells. These findings suggest effects of Notch on LIC activity may be mediated in part by enhancing the responsiveness of T-ALL cells to ambient growth factors, and provide strong rationale for use of IGF1R inhibitors to improve initial response to therapy and to achieve long-term cure of patients with T-ALL.
T cell acute lymphoblastic leukemia (T-ALL) is an aggressive cancer of immature T cell progenitors that often shows aberrant activation of NOTCH1 and PI3K–Akt pathways. Activating mutations of Notch1 occur in >50% of cases of T-ALL (Weng et al., 2004), whereas mutations in related Notch pathway elements such as Sel10/Fbw7 occur in 8–16% of cases (O’Neil et al., 2007; Thompson et al., 2007). PI3K–Akt pathway activation occurs in >85% of cases (Silva et al., 2008) via diverse mechanisms, including mutation or inactivation of PTEN (Kawamura et al., 1999; Perentesis et al., 2004; Maser et al., 2007; Palomero et al., 2007; Silva et al., 2008; Gutierrez et al., 2009) and mutation of PIK3 and Akt (Kawamura et al., 1999; Gutierrez et al., 2009). Activation of PI3K–Akt has been shown to collaborate with Notch in leukemogenesis (Medyouf et al., 2010), enhance growth of established leukemias (Chiarini et al., 2009; Cullion et al., 2009; Levy et al., 2009; Sanda et al., 2010), and in some contexts to relieve dependence on Notch signaling (Palomero et al., 2007). For cases that lack such mutations, however, the mechanisms that support activation of the pathway are unknown. More generally, it is also unknown to what extent growth factor–dependent stimulation of cognate receptor tyrosine kinases (RTKs) contributes to the net signaling output.
Although previous works have focused on the role of IL-7 signaling in T-ALL, including effects on downstream PI3K–Akt activation (Dibirdik et al., 1991; Barata et al., 2004a,b,c, 2005; González-Garcia et al., 2009; Shochat et al., 2011; Silva et al., 2011), we considered that insulin-like growth factor (IGF)-1 receptor (IGF1R) may also play an important role. IGFs and their receptors regulate normal cell growth and contribute to transformation and growth of malignant cells in many contexts (Pollak et al., 2004). IGF1 and IGF2 bind to IGF1R, a transmembrane receptor tyrosine kinase (RTK), thereby initiating a cascade of downstream phosphorylation events that bifurcates along both PI3K–Akt and Ras–Raf–MAPK pathways. PI3K–Akt activation leads to enhanced cellular metabolism and protein synthesis via mTOR and enhanced survival via BAD/Bcl2, p53, NF-kB, and FOXOs, whereas Ras–Raf–MAPK activation generally results in increased cellular proliferation (Pollak et al., 2004; Greer and Brunet, 2005). Signaling through IGF1R has also been implicated in self-renewal of stem cells, both in embryonic (Bendall et al., 2007) and hematopoietic (Ivanova et al., 2002) contexts.
RESULTS
IGF1R is broadly expressed in T-ALL
To begin to address a potential role for IGF1R in T-ALL, we assessed IGF1R expression in mouse and human T-ALL cells. Analysis of IGF1R by Western blot and flow cytometry revealed IGF1R was expressed in all cases examined, albeit at varying levels (Fig. 1). For human cells, we examined both established cell lines and xenograft-expanded primary human samples (Weng et al., 2004; Weng et al., 2006; Medyouf et al., 2010). For mouse cells, we examined primary leukemias derived by retroviral transduction/transplantation of bone marrow with an activated form of NOTCH1 termed ΔE (Pear et al., 1996). To confirm IGF1R-stimulated PI3K–Akt in these contexts, we pulsed serum-starved leukemia cells with recombinant IGF-1 and measured phospho-Akt activation by flow cytometry. We observed that both human and mouse leukemia cells respond robustly to IGF-1 stimulation under these conditions (Fig. S1).
IGF1R is expressed broadly in human and mouse T-ALL. (A and B) Western blot and (C and D) flow cytometric analysis of total and surface IGF1R protein expression, respectively, from human cell lines (A and C), primary mouse leukemias (B) derived by retroviral transduction/transplantation of bone marrow with an activated form of Notch1 termed ΔE, and xenograft-expanded primary human samples (D). Western blot controls in (B) are mouse embryonic fibroblasts derived from IGF1Rnull mouse embryos (R−) and the same cells stably transfected with an IGF1R cDNA expression construct (R+). At least 20,000 events were collected within each gate for all flow cytometry assays. Data depicted are representative of at least two independent experiments.
IGF1R is expressed broadly in human and mouse T-ALL. (A and B) Western blot and (C and D) flow cytometric analysis of total and surface IGF1R protein expression, respectively, from human cell lines (A and C), primary mouse leukemias (B) derived by retroviral transduction/transplantation of bone marrow with an activated form of Notch1 termed ΔE, and xenograft-expanded primary human samples (D). Western blot controls in (B) are mouse embryonic fibroblasts derived from IGF1Rnull mouse embryos (R−) and the same cells stably transfected with an IGF1R cDNA expression construct (R+). At least 20,000 events were collected within each gate for all flow cytometry assays. Data depicted are representative of at least two independent experiments.
Pharmacologic inhibition of IGF1R compromises T-ALL cell growth
To assess the extent to which T-ALL cells are dependent on IGF1R signaling, we used pharmacologic IGF1R inhibitors. Most small molecule IGF1R inhibitors also affect insulin receptor caused by their close homology and at higher doses may be expected to cross react with more distantly related receptor tyrosine kinases. One such inhibitor, BMS-536924 (Wittman et al., 2005), substantially inhibited growth/proliferation of both mouse and human leukemia cells in vitro (Fig. 2 and Fig. S2). Primary leukemia cells were generally more sensitive to BMS-536924 with IC50 values in the 0.1–1.0 µM range, whereas established cell lines required somewhat higher doses (2–4 µM) to achieve similar effects. As expected, BMS-536924 completely suppressed Akt activation by both IGF-1 and insulin in human and mouse T-ALL cells (Fig. S1).
Pharmacologic inhibition of IGF1R blocks growth of T-ALL cells. Flow cytometric analysis of cell proliferation by BrdU incorporation after treatment with a small molecule IGF1R inhibitor (BMS-536924) versus DMSO vehicle (mock), or IGF1R blocking antibody (αIR3), for 48–72 h in vitro. (A) Three representative independent primary mouse Notch1(ΔE) leukemias (#324, #327, #329). (B) Four independent xenograft-expanded primary human T-ALL samples (D115, K419, K424, and M69). (C) Two human T-ALL cell lines (ALLSIL and HPBALL). Error bars indicate standard deviation for assays performed in triplicate. Data depicted are representative of at least three independent experiments.
Pharmacologic inhibition of IGF1R blocks growth of T-ALL cells. Flow cytometric analysis of cell proliferation by BrdU incorporation after treatment with a small molecule IGF1R inhibitor (BMS-536924) versus DMSO vehicle (mock), or IGF1R blocking antibody (αIR3), for 48–72 h in vitro. (A) Three representative independent primary mouse Notch1(ΔE) leukemias (#324, #327, #329). (B) Four independent xenograft-expanded primary human T-ALL samples (D115, K419, K424, and M69). (C) Two human T-ALL cell lines (ALLSIL and HPBALL). Error bars indicate standard deviation for assays performed in triplicate. Data depicted are representative of at least three independent experiments.
We next examined whether blocking antibodies against IGF1R, which might be expected to exhibit a higher degree of specificity than small molecule kinase inhibitors, would recapitulate effects seen with BMS-536924. The IGF1R-blocking antibody αIR3 substantially inhibited proliferation in three of four primary human leukemias tested (Fig. 2 B, rightmost column). Notably, the one resistant case (K419) expressed lower levels of IGF1R compared with the others (Fig. S3) and showed minimal response to IGF-1 stimulation (unpublished data), suggesting the effect of BMS-536924 on this particular tumor may be mediated by inhibition of insulin receptor. In contrast, case M69 is highly sensitive to αIR3, but largely resistant to BMS-536924, raising the possibility of either off-target antibody effects or inherent drug resistance. Notwithstanding these exceptions, the overall results support that pharmacologic inhibition of IGF1R signaling can significantly inhibit the growth of most T-ALL cells.
Generation of T cell leukemias with reduced IGF1R signaling
We next used a genetic approach to further explore the effects of reduced and/or complete loss of IGF1R on T-ALL cells. Specifically, we generated primary mouse T cell leukemias by transduction of bone marrow from conditional IGF1R mice with Notch1(ΔE) retrovirus, followed by transplantation into congenic WT recipients. The conditional allele used, IGF1Rneo, carries loxP sites flanking the third exon, but also retains a loxP-flanked neo cassette within the second intron (Fig. 3 A). This neo cassette interferes with normal transcript splicing and results in reduced expression of full-length IGF1R protein in homozygous IGF1Rneo/neo cells (Holzenberger et al., 2000). Despite the decreased level of IGF1R expression, we were able to generate primary IGF1Rneo/neo leukemias with Notch1(ΔE) retrovirus, albeit with slightly increased latency as compared with WT background (median survival 64.5 vs. 51.5 d; Fig. 3 B). All other disease parameters (penetrance, immunophenotype, histology, disease distribution, and extent) were highly comparable to control IGF1R+/+ ΔE leukemias (Fig. 3, C and D; and not depicted). Importantly, we confirmed that IGF1Rneo/neo ΔE leukemias expressed reduced levels of full-length IGF1R protein (32 ± 20% of levels observed in IGF1R+/+ ΔE leukemias, n = 7; Fig. 3 E and not depicted).
Primary mouse T cell leukemias are generated efficiently by activated Notch1 despite reduced IGF1R expression. (A) Schematic of the IGF1Rneo allele. The retained neo cassette within the second intron results in reduced expression of full-length IGF1R protein in IGF1Rneo/neo mice (Holzenberger et al., 2000). (B) Survival of mice transplanted with retroviral Notch1(ΔE)-transduced bone marrow from either WT (IGF1R+/+; n = 6) or IGF1R hypomorph (IGF1Rneo/neo; n = 14) donor animals. ***, P < 0.0001 (Log-rank test). (C) Spleen and liver organ weights at necropsy of individual morbid mice transplanted with Notch1(ΔE)-transduced bone marrow from IGF1R+/+ (n = 6) and IGF1Rneo/neo (n = 11) backgrounds. Error bars indicate standard deviation. (D) Immunophenotypic analysis of representative primary mouse ΔE leukemias derived on the IGF1Rneo/neo background. (E) Western blot analysis of total IGF1R protein expression in representative primary mouse ΔE leukemias on IGF1R+/+, IGF1Rneo/+, and IGF1Rneo/neo backgrounds. R+ and R− mouse embryonic fibroblast staining controls and Erk2 loading control are indicated. Data depicted in D and E are representative of at least seven independent samples.
Primary mouse T cell leukemias are generated efficiently by activated Notch1 despite reduced IGF1R expression. (A) Schematic of the IGF1Rneo allele. The retained neo cassette within the second intron results in reduced expression of full-length IGF1R protein in IGF1Rneo/neo mice (Holzenberger et al., 2000). (B) Survival of mice transplanted with retroviral Notch1(ΔE)-transduced bone marrow from either WT (IGF1R+/+; n = 6) or IGF1R hypomorph (IGF1Rneo/neo; n = 14) donor animals. ***, P < 0.0001 (Log-rank test). (C) Spleen and liver organ weights at necropsy of individual morbid mice transplanted with Notch1(ΔE)-transduced bone marrow from IGF1R+/+ (n = 6) and IGF1Rneo/neo (n = 11) backgrounds. Error bars indicate standard deviation. (D) Immunophenotypic analysis of representative primary mouse ΔE leukemias derived on the IGF1Rneo/neo background. (E) Western blot analysis of total IGF1R protein expression in representative primary mouse ΔE leukemias on IGF1R+/+, IGF1Rneo/+, and IGF1Rneo/neo backgrounds. R+ and R− mouse embryonic fibroblast staining controls and Erk2 loading control are indicated. Data depicted in D and E are representative of at least seven independent samples.
Reduced IGF1R signaling compromises leukemia-initiating cell (LIC) activity
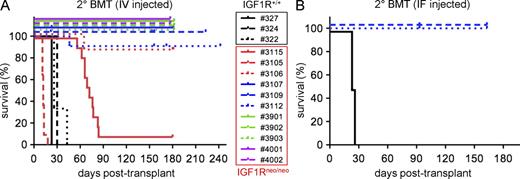
To explore whether the increased latency observed for IGF1Rneo/neo ΔE leukemias might be caused by delayed transformation or a reduced net proliferative rate, we performed serial transplantation of IGF1R+/+ and IGF1Rneo/neo ΔE leukemias into syngeneic/congenic recipients by tail vein injection. Unexpectedly, we observed that only a subset of IGF1Rneo/neo primary leukemias was capable of transferring disease to secondary recipients. Among 11 independent primary leukemias assessed, 7 (64%) were nontransplantable, 2 (18%) were transplantable in a minority of recipients, and 2 (18%) were fully transplantable, including one with prolonged latency (Fig. 4 A). In contrast, all IGF1R+/+ primary leukemias were fully transplantable and exhibited short latencies. We also performed tertiary and quarternary transplants for a subset of cases. The IGF1Rneo/neo leukemia with prolonged latency in secondary recipients (#3115) failed to produce disease in tertiary recipients, whereas the other transplantable IGF1Rneo/neo leukemia (#3105) and all IGF1R+/+ leukemias produced short latency disease in all tertiary and quarternary recipients (Table S1). Notably, injection of IGF1Rneo/neo leukemia cells directly into the femoral bone marrow space of recipient mice failed to produce leukemia, suggesting that their defect in transplantability is not caused by impaired homing to the bone marrow (Fig. 4 B). Thus, these results indicate that although moderate levels of IGF1R signaling are sufficient for expansion of bulk leukemia cells, higher levels are required to support LIC activity as assayed by serial transplantation in syngeneic/congenic recipients.
Mouse T cell leukemias with reduced IGF1R expression are defective in serial transplantation. Survival of mice transplanted with primary mouse ΔE leukemia cells from either IGF1R+/+ or IGF1Rneo/neo backgrounds by i.v. (A) intrafemoral (IF; B) injection route. Each numbered sample represents a different primary leukemia from mice in Fig. 3 B injected into secondary recipients. Raw survival data, including numbers of animals in each cohort, are provided in Table S1. Data depicted are collated from four independent transplantation experiments.
Mouse T cell leukemias with reduced IGF1R expression are defective in serial transplantation. Survival of mice transplanted with primary mouse ΔE leukemia cells from either IGF1R+/+ or IGF1Rneo/neo backgrounds by i.v. (A) intrafemoral (IF; B) injection route. Each numbered sample represents a different primary leukemia from mice in Fig. 3 B injected into secondary recipients. Raw survival data, including numbers of animals in each cohort, are provided in Table S1. Data depicted are collated from four independent transplantation experiments.
Deletion or pharmacologic inhibition of IGF1R prevents disease establishment/progression
To assess the effect of complete loss of IGF1R in T-ALL cells, we transduced bone marrow from IGF1Rneo/neo Rosa26CreERT mice with Notch1(ΔE) retrovirus, followed by transplantation into WT recipient mice. At 4 wk after transplant, peripheral blood was assessed by flow cytometry to confirm engraftment by GFP+ CD4+ CD8+ leukemia cells. Mice were then divided into equivalent control versus treatment groups, and the latter was fed tamoxifen-containing chow (1 g/kg) for a 7-d period to induce deletion of IGF1R. Mice from both groups were then sacrificed, and tissues were harvested. Preliminary results show that tamoxifen-treated mice demonstrate significantly lower levels of disease involvement in all tissues examined (bone marrow, spleen, and thymus), and leukemia cells from tamoxifen-treated animals exhibited lower proliferative and higher apoptotic indices as compared with controls (Fig. S4, A–E). Furthermore, leukemia cells explanted from moribund, untreated animals (∼8 wk post-transplant) and cultured with 4-hydroxytamoxifen (4-OHT) in vitro to induce deletion of IGF1R also demonstrated growth arrest and loss of viability (Fig. S4 F). 4-OHT treatment showed no discernible toxic effects on control IGF1Rneo/neo Rosa26+/+ leukemia cells. Additionally, preliminary results from mice engrafted with a T-ALL cell line (144CLP) derived from a mouse KrasG12D tumor (Chiang et al., 2008) show that treatment with BMS-754807, a potent IGF1R inhibitor currently in clinical development (Carboni et al., 2009), significantly prolonged survival (Fig. S5), thus corroborating the genetic deletion results. Collectively, these data support that abrogation of IGF1R signaling impedes T-ALL cell growth/survival and interferes with disease establishment/progression.
Transplantable IGF1Rneo leukemias show evidence for downstream compensation
Our observation that a minority of IGF1Rneo/neo leukemias is transplantable suggests secondary alterations may be selected for in vivo that compensate for reduced IGF1R signaling. Despite the known frequency of PTEN loss and PIK3CA/PIK3R1 mutations in T-ALL (Maser et al., 2007; Palomero et al., 2007; Gutierrez et al., 2009), we were unable to detect these alterations in any of the transplantable ΔE-IGF1Rneo/neo leukemias (Fig. 5, A and B; and not depicted). We thus considered that enhanced responsiveness to growth factor stimulation could compensate for reduced IGF1R expression. In fact, transplantable leukemias #3112 and #3115 demonstrated enhanced pAkt response to serum stimulation as compared with nontransplantable leukemias, though not to the degree exhibited by IGF1R+/+ cells (Fig. 5 C). We excluded the possibility that IGF1R itself was up-regulated as transplantable IGF1Rneo/neo leukemias remained only minimally responsive to IGF-1, and are comparable to nontransplantable IGF1Rneo/neo leukemias in this regard (Fig. 5 D). We also noted the one IGF1Rneo/neo leukemia exhibiting very short latency in secondary recipients (#3105) had unusually high levels of pAkt at steady state (twofold greater than IGF1R+/+ cells; Fig. 5 E). The elevated pAkt in these cells was not reduced even after prolonged serum starvation (unpublished data), suggesting the presence of some constitutively activating mutation that confers growth factor independence. In contrast, we have not found similarly compelling evidence for hyperactivation of the Ras–Raf–MAPK pathway to explain the unique transplantability of these IGF1Rneo/neo clones (unpublished data). These few examples suggest PI3K–Akt activation may potentially be more important than Ras–Raf–MAPK signaling in conferring LIC activity; however, further study is needed to clarify this issue.
Transplantable clones with reduced IGF1R expression show compensatory PI3K–Akt activation. (A) Western blot and (B) flow cytometric analysis of PTEN protein expression in primary and transplantable secondary ΔE-IGF1Rneo leukemias. (C and D) Akt activation as measured by intracellular phospho-Akt(Ser473) flow cytometry in response to stimulation with FBS (C) or (D) recombinant IGF-1 × (D) 10 min after 5-h serum starvation. (E) Steady-state level of Akt activation as measured by pAkt(Ser473) in transplantable ΔE-IGF1R+/+ and ΔE-IGF1Rneo (#3105) leukemias (n = 3 independent mice for each cohort). For C and D, the five ΔE-IGF1Rneo transplantable clones shown are (4 x #3115, 1 x #3112). Error bars indicate standard deviation. *, P < 0.05; **, P < 0.01 (Student’s t test). Data depicted in A and B are representative of at least three independent experiments. Data depicted in C–E are representative of two independent experiments and include at least three independent mice per cohort per experiment.
Transplantable clones with reduced IGF1R expression show compensatory PI3K–Akt activation. (A) Western blot and (B) flow cytometric analysis of PTEN protein expression in primary and transplantable secondary ΔE-IGF1Rneo leukemias. (C and D) Akt activation as measured by intracellular phospho-Akt(Ser473) flow cytometry in response to stimulation with FBS (C) or (D) recombinant IGF-1 × (D) 10 min after 5-h serum starvation. (E) Steady-state level of Akt activation as measured by pAkt(Ser473) in transplantable ΔE-IGF1R+/+ and ΔE-IGF1Rneo (#3105) leukemias (n = 3 independent mice for each cohort). For C and D, the five ΔE-IGF1Rneo transplantable clones shown are (4 x #3115, 1 x #3112). Error bars indicate standard deviation. *, P < 0.05; **, P < 0.01 (Student’s t test). Data depicted in A and B are representative of at least three independent experiments. Data depicted in C–E are representative of two independent experiments and include at least three independent mice per cohort per experiment.
IGF1R mRNA and protein expression is up-regulated by Notch
To explore mechanisms that may support the high-level IGF1R expression required for LIC activity, we considered a potential role for Notch signaling. Notch has previously been shown to promote activation of PI3K–Akt in developing thymocytes (Ciofani and Zuñiga-Pflucker, 2005), and we noted in our expression profile datasets evidence for up-regulation of IGF1R by Notch signaling. More specifically, we performed microarray-based expression profiling of five human T-ALL cell lines (ALLSIL, DND41, HPBALL, KOPTK1, and TALL-1) treated with γ-secretase inhibitor (GSI) to inhibit Notch signaling or vehicle control (DMSO). These five cell lines were selected because they undergo growth arrest upon GSI treatment (Weng et al., 2004; Palomero et al., 2007). To control for off-target effects of GSI, we also profiled cell lines that had been retrovirally transduced with ICN1, and then treated with GSI or DMSO. ICN1, the intracellular domain of Notch1, is unaffected by GSI treatment and thus maintains Notch signaling in the presence of GSI (Weng et al., 2003, 2004). Further, because Notch1 induces transcription of c-Myc (Palomero et al., 2006b; Sharma et al., 2006; Weng et al., 2006), which in turn induces transcription of many other genes (Fernandez et al., 2003), we also profiled cells that had been transduced with c-Myc and then treated with GSI or DMSO to segregate Notch targets from c-Myc targets. We identified genes whose expression level most strongly correlated with Notch activity, but not with c-Myc, and noted the conspicuous presence of IGF1R on this list (Table S2) and in other published microarray datasets generated from GSI-treated T-ALL cells (Fig. S6; Palomero et al., 2006b).
We also performed a similar expression profiling experiment using the human T-ALL cell line, CUTLL1, which harbors a TcrB/Notch1 fusion gene that drives expression of a form of Notch1 resembling ΔE (Palomero et al., 2006a). CUTLL1 cells transduced with empty retrovirus (MigR1) or dominant-negative (DN) MAML1-GFP, a specific Notch inhibitor (Weng et al., 2003; Maillard et al., 2004; Weng et al., 2004), were treated with GSI for 3 d, after which GSI was washed out to create a timed pulse of Notch signaling activity. Expression profiling was done on cells before washout and at 2 and 4 h after washout to identify likely direct Notch1 target genes. We observed GSI washout up-regulated expression of IGF1R along with other known Notch1 target genes, such as Hes1, Hes4, Hes5, NRARP, and DTX1 in MigRI control cells, and that DN-MAML1-GFP partially or completely abrogated up-regulation of the same set of genes (Fig. S7).
To validate these expression profile data, we performed qRT-PCR for IGF1R mRNA. Notch-dependent T-ALL cell lines (ALLSIL, DND41, HPBALL, KOPTK1, and TALL-1) down-regulated IGF1R mRNA levels up to twofold when treated with GSI, whereas Notch-independent lines (Jurkat, PF382, and RPMI 8402) showed 6–14-fold down-regulation of IGF1R mRNA (Fig. 6 A). GSI blockade also down-regulated IGF1R protein levels 2–3 fold on average, as judged by flow cytometry and Western blotting of human T-ALL cell lines and xenograft-expanded primary samples (Fig. 6, B–D; and Fig. S8). This effect of GSI is likely specific to Notch, as DN-MAML1 also decreased IGF1R protein levels (Fig. 7 A), and the effect of GSI on IGF1R was rescued by retroviral transduction with ICN1 (Fig. 7 B). Conversely, culture of xenograft-expanded primary human T-ALL cells on MS5 stromal cells expressing the Notch ligand DL1, but not MS5 control cells, increased IGF1R levels, whereas no such change was observed for the control marker CD45 (Fig. 7 C). In addition, this effect of DL1 ligand on IGF1R levels was abrogated by treatment with GSI (Fig. 6 D). Thus, these data are consistent with a role for Notch signaling in up-regulation of IGF1R expression in human T-ALL cells.
Inhibition of Notch signaling with GSI down-regulates IGF1R expression in human T-ALL cells. (A) qRT-PCR analysis of IGF1R mRNA in human T-ALL cell lines treated in vitro with γ-secretase inhibitor (GSI) to block Notch signaling versus DMSO vehicle (mock) for 2–10 d. Error bars indicate standard deviation for assays performed in triplicate. (B and C) Flow cytometric analysis of surface IGF1R expression by human T-ALL cell lines treated with GSI versus DMSO vehicle for 6–8 d. Data are representative of multiple replicates. (D) Flow cytometric analysis of surface IGF1R expression by xenograft-expanded primary human T-ALL cells. Cells were cultured on MS5-DL1 feeders to stimulate Notch signaling versus MS5 control feeders and then treated with 1.0 µM compound E (GSI) for 2–4 d to block Notch signaling. Flow histograms for a representative case are depicted on the left, and results from six different patient samples are summarized on the right. Error bars indicate standard deviation. **, P < 0.01 (Student’s t test). At least 20,000 gated live events were collected for all flow cytometry assays.
Inhibition of Notch signaling with GSI down-regulates IGF1R expression in human T-ALL cells. (A) qRT-PCR analysis of IGF1R mRNA in human T-ALL cell lines treated in vitro with γ-secretase inhibitor (GSI) to block Notch signaling versus DMSO vehicle (mock) for 2–10 d. Error bars indicate standard deviation for assays performed in triplicate. (B and C) Flow cytometric analysis of surface IGF1R expression by human T-ALL cell lines treated with GSI versus DMSO vehicle for 6–8 d. Data are representative of multiple replicates. (D) Flow cytometric analysis of surface IGF1R expression by xenograft-expanded primary human T-ALL cells. Cells were cultured on MS5-DL1 feeders to stimulate Notch signaling versus MS5 control feeders and then treated with 1.0 µM compound E (GSI) for 2–4 d to block Notch signaling. Flow histograms for a representative case are depicted on the left, and results from six different patient samples are summarized on the right. Error bars indicate standard deviation. **, P < 0.01 (Student’s t test). At least 20,000 gated live events were collected for all flow cytometry assays.
Inhibition of Notch signaling with dominant-negative MAML1 and activation by ICN1 or DL1 ligand confirm IGF1R regulation by Notch. Flow cytometric analysis of surface IGF1R expression. (A) Human T-ALL cell lines were transduced with dominant negative Mastermind-like-1 retrovirus (DN-MAML1-GFP) or empty virus control (MigRI). (B) Human T-ALL cell lines were transduced with ICN retrovirus (Mig ICN) and then treated with 1.0 µM compound E (GSI) for 4 d (HPBALL and TALL-1) or 8 d (PF382) to block endogenous Notch signaling or DMSO vehicle (mock). Retrovirally transduced cells in A and B were discriminated from nontransduced cells by gating for GFP. Data depicted in A and B are representative of multiple replicates. (C) Xenograft-expanded primary human T-ALL cells were cultured in vitro on MS5-DL1 feeders to stimulate Notch signaling versus control MS5 feeders. CD45 expression levels were also assessed simultaneously with IGF1R by flow cytometry. Flow histograms for a representative case are depicted on the left, and results from six different patient samples are summarized on the right. Error bars indicate standard deviation. *, P < 0.05 (Student’s t test). At least 20,000 live events were collected within each gate for all flow cytometry assays.
Inhibition of Notch signaling with dominant-negative MAML1 and activation by ICN1 or DL1 ligand confirm IGF1R regulation by Notch. Flow cytometric analysis of surface IGF1R expression. (A) Human T-ALL cell lines were transduced with dominant negative Mastermind-like-1 retrovirus (DN-MAML1-GFP) or empty virus control (MigRI). (B) Human T-ALL cell lines were transduced with ICN retrovirus (Mig ICN) and then treated with 1.0 µM compound E (GSI) for 4 d (HPBALL and TALL-1) or 8 d (PF382) to block endogenous Notch signaling or DMSO vehicle (mock). Retrovirally transduced cells in A and B were discriminated from nontransduced cells by gating for GFP. Data depicted in A and B are representative of multiple replicates. (C) Xenograft-expanded primary human T-ALL cells were cultured in vitro on MS5-DL1 feeders to stimulate Notch signaling versus control MS5 feeders. CD45 expression levels were also assessed simultaneously with IGF1R by flow cytometry. Flow histograms for a representative case are depicted on the left, and results from six different patient samples are summarized on the right. Error bars indicate standard deviation. *, P < 0.05 (Student’s t test). At least 20,000 live events were collected within each gate for all flow cytometry assays.
ICN1/CSL binds to an intronic IGF1R enhancer
Once cleaved from the plasma membrane by γ-secretase, ICN1 translocates to the nucleus where it forms a ternary complex on DNA with the DNA-binding factor CSL and the coactivator MAML1 to stimulate transcription of target genes (Aster et al., 2008). To define the regulatory elements through which Notch1 up-regulates target genes in an unbiased fashion, we performed chromatin immunoprecipitation (ChIP)-Seq analysis on the human T-ALL cell line, CUTLL1, using antibodies directed against Notch1 and CSL (Wang et al., 2011). Alignment of sequencing reads from duplicate CSL and Notch1 libraries identified one high-confidence ICN1/CSL-binding site within intron 20 of IGF1R at a position >250 kb 3′ of the proximal promoter (Fig. 8 A). The next nearest ICN1/CSL-binding site lies ∼10.5 Mbp 5′ of the proximal promoter, making this 3′ binding site the only likely candidate response element for Notch1 regulation. Motif analysis using recently developed algorithms derived from protein-binding microarrays (Del Bianco et al., 2010) showed that the center of the region under the ICN1/CSL-binding peak contains high (CATGGGAA) and moderate (GCTGAGAA) affinity CSL sites oriented head-to-head and separated by a 17-bp spacer (Fig. 8 B). This architecture is typical of a sequence-paired site, a special type of Notch response element first identified in Drosophila enhancer of split (E[spl]) locus that is present in mammalian E(spl) homologues such as Hes1 (Jarriault et al., 1995) and other genes such as preTα (Liu et al., 2010). Loading of ICN1 onto this site was confirmed by ChIP/quantitative PCR (qPCR) analyses, performed on CUTLL1 cells and two additional human T-ALL cell lines, HPBALL and KOPTK1 (Fig. 8 C). In addition, GSI treatment of CUTLL1 cells depleted both CSL and ICN1 from this site (in line with work from Bray’s group indicating that ICN stabilizes CSL interactions with DNA; Krejcí and Bray, 2007), and GSI washout resulted in rapid reloading, indicating that ICN1/CSL occupancy is dynamic (Fig. 8 D).
ICN1/CSL binds dynamically to a site within intron 20 of human IGF1R. (A) Alignment of sequencing reads over intron 20 of the IGF1R locus from ChIP libraries prepared from the human T-ALL cell line, CUTLL1, with antibodies specific for Notch1 and CSL as compared with input cells. (B) Genomic DNA sequence from human IGF1R intron 20 (NT_010274.17: 14461410–14461610) with sequence-paired ICN1/CSL binding sites highlighted in bold and 17-bp spacer underlined. (C) ChIP/qPCR analysis of IGF1R intron 20 from three different human T-ALL cell lines using a Notch1-specific antibody as compared with preimmune antiserum. (D) ChIP/qPCR analysis of IGF1R intron 20 from CUTLL1 cells using antibodies specific for Notch1 and CSL compared with control rabbit IgG. Cells were treated with GSI for 3 d to block Notch signaling (GSI x 3d) versus DMSO vehicle (DMSO). GSI was washed out, and cells were harvested 4 h later (GSI x 3d, then wash 4 h). Quantitation of immunoprecipitated DNA is expressed relative to input DNA (% input). Error bars indicate standard deviation for qPCR assays performed in triplicate. Numbers above bars in D indicate relative enrichment over control. ChIP libraries were prepared in duplicate, and local ChIP/qPCR analyses were performed twice. Representative results are shown.
ICN1/CSL binds dynamically to a site within intron 20 of human IGF1R. (A) Alignment of sequencing reads over intron 20 of the IGF1R locus from ChIP libraries prepared from the human T-ALL cell line, CUTLL1, with antibodies specific for Notch1 and CSL as compared with input cells. (B) Genomic DNA sequence from human IGF1R intron 20 (NT_010274.17: 14461410–14461610) with sequence-paired ICN1/CSL binding sites highlighted in bold and 17-bp spacer underlined. (C) ChIP/qPCR analysis of IGF1R intron 20 from three different human T-ALL cell lines using a Notch1-specific antibody as compared with preimmune antiserum. (D) ChIP/qPCR analysis of IGF1R intron 20 from CUTLL1 cells using antibodies specific for Notch1 and CSL compared with control rabbit IgG. Cells were treated with GSI for 3 d to block Notch signaling (GSI x 3d) versus DMSO vehicle (DMSO). GSI was washed out, and cells were harvested 4 h later (GSI x 3d, then wash 4 h). Quantitation of immunoprecipitated DNA is expressed relative to input DNA (% input). Error bars indicate standard deviation for qPCR assays performed in triplicate. Numbers above bars in D indicate relative enrichment over control. ChIP libraries were prepared in duplicate, and local ChIP/qPCR analyses were performed twice. Representative results are shown.
The distant location of the putative ICN1/CSL response element in IGF1R suggested that it represents an enhancer. In support of this possibility, additional ChIP analyses documented recruitment of the histone acetyltransferases CREB-binding protein (CBP) and p300, as well as RNA polymerase II, to this site (Fig. S9 A; Hatzis and Talianidis, 2002; Wang et al., 2005). Moreover, we observed enrichment of histone H3K4 mono- and dimethylation chromatin marks (characteristic of enhancers) relative to H3K4 trimethylation marks (a feature of promoters; Heintzman et al., 2007), and low levels of the repressive trimethyl histone H3K27 chromatin mark (Fig. S9 B; Kirmizis et al., 2004). Finally, additional ChIP-Seq data showed a local decrease in H3K4 methylation at the precise site of the intronic IGF1R sequence-paired site, indicating that this site lies in accessible chromatin that has been depleted of nucleosomes (Fig. S9 C). Collectively, these data support the presence of an intronic Notch1-responsive enhancer in human IGF1R that is active in human T-ALL cells.
Physiological relevance of Notch-induced up-regulation of IGF1R
Notch signaling is likely one of several factors that influence IGF1R expression in T-ALL, as Notch inhibition results in only a two- to threefold decrease in surface IGF1R expression, on average. To assess whether this two- to threefold change in IGF1R protein levels has a significant effect on downstream signaling, we measured levels of intracellular phosphorylated Akt (pAkt) after pulsing serum-starved leukemia cells with recombinant IGF-1. We observed GSI-treated cells to be ∼20-fold less responsive to IGF-1 than vehicle-treated control cells (Fig. 9 A). Although some of this effect is presumably caused by GSI effects on IGF1R expression, it has also been reported that Notch represses PTEN via HES-1 (Palomero et al., 2007), which could also impact the pAkt response. Indeed, GSI treatment induced PTEN expression in HPBALL cells; however, there was no detectable change in ALLSIL or DND41 cells (Fig. 9 B). Accordingly, restoration of IGF1R expression in GSI-treated cells by retroviral transduction only partially rescued the pAkt response to IGF1 in HPBALL cells (Fig. 9 C), whereas rescue was complete in PTENnull PF382 cells (Fig. 9 D; Palomero et al., 2007). Importantly, we confirmed retroviral IGF1R restored expression in GSI-treated cells to levels comparable to parental cells. Thus, although Notch inhibition can affect both IGF1R and PTEN expression, Notch contributes to supporting IGF1R expression at high levels, thereby allowing T-ALL cells to respond more robustly to ambient levels of IGF-1/2.
Notch-induced IGF1R expression enhances PI3K–Akt signaling in response to IGF1. (A) Flow cytometric analysis of intracellular phospho-Akt levels. Cells were treated with GSI versus DMSO vehicle for 6–8 d, serum starved overnight, and then pulsed with recombinant IGF1 for 10 min before assay. (B) Western blot analysis of PTEN protein expression in cells treated with GSI versus mock for 4 d. (C and D) Flow cytometric analysis for intracellular phospho-Akt levels (left) and surface IGF1R levels (right). Cells were retrovirally transduced with Mig IGF1R, treated with GSI versus DMSO vehicle for 4 d, serum starved overnight, and then pulsed with recombinant IGF1 for 10 min before phospho-Akt assay. Transduced versus nontransduced cells within the same culture were distinguished by gating for GFP. Surface IGF1R expression level was also assessed by flow cytometry immediately before stimulation with IGF1. Filled grey histograms represent second antibody staining controls. At least 20,000 events were collected within each gate for all flow cytometry assays. Data depicted are representative of at least two independent experiments.
Notch-induced IGF1R expression enhances PI3K–Akt signaling in response to IGF1. (A) Flow cytometric analysis of intracellular phospho-Akt levels. Cells were treated with GSI versus DMSO vehicle for 6–8 d, serum starved overnight, and then pulsed with recombinant IGF1 for 10 min before assay. (B) Western blot analysis of PTEN protein expression in cells treated with GSI versus mock for 4 d. (C and D) Flow cytometric analysis for intracellular phospho-Akt levels (left) and surface IGF1R levels (right). Cells were retrovirally transduced with Mig IGF1R, treated with GSI versus DMSO vehicle for 4 d, serum starved overnight, and then pulsed with recombinant IGF1 for 10 min before phospho-Akt assay. Transduced versus nontransduced cells within the same culture were distinguished by gating for GFP. Surface IGF1R expression level was also assessed by flow cytometry immediately before stimulation with IGF1. Filled grey histograms represent second antibody staining controls. At least 20,000 events were collected within each gate for all flow cytometry assays. Data depicted are representative of at least two independent experiments.
DISCUSSION
We have presented data showing that IGF1R mediates important growth/survival signals in T-ALL cells, and that although moderate levels of signaling are adequate for maintenance of the bulk cell population, high levels are required for maintenance of LIC activity as indicated by serial transplantation assay. The effect of reduced IGF1R signaling on disease transplantability could be caused by (a) a quantitative decrease in the number of leukemia stem cells; (b) a qualitative defect in self-renewal, engraftment ability, and/or immune resistance of leukemia stem cells; or (c) a reduced probabilistic likelihood of engraftment/propagation of bulk leukemia cells. Notably, the last possibility does not presume the existence of leukemia stem cells in this model; however, we have generated data that supports that Notch T-ALLs indeed contain bona fide leukemia stem cells (unpublished data). Nonetheless, further studies will be required to distinguish between these possibilities.
We have also shown that Notch directly up-regulates IGF1R expression in human T-ALL cells to an extent that substantially enhances their sensitivity to ambient ligand. It is worthwhile to note that we have found no evidence to support that Notch regulates IGF1R in mouse T-ALL cells. In fact, the sequence-paired ICN1/CSL-binding site within intron 20 of human IGF1R is not conserved in the mouse, suggesting that sequence divergence has decoupled this point of interaction between the Notch1 and IGF1R pathways. This is perhaps not surprising given that 40–90% of transcription factor binding events are not conserved between mouse and human (Odom et al., 2007), underscoring the importance of studying human cells and animal models in parallel.
The existence of leukemia stem cells in human T-ALL is supported by xenograft transplantation assays (Cox et al., 2007; Chiu et al., 2010; Gerby et al., 2011) and also in mouse models of T-ALL by transplantation into SCID or Rag1−/− immunocompromised recipients (Guo et al., 2008; Tremblay et al., 2010). The precise cellular compartment most enriched for LIC activity appears to vary from patient to patient and between different animal models, and likely depends on the particular complement of genetic alterations present in the leukemic clone and the particular transplant recipient used (Cox et al., 2007; Quintana et al., 2008; Chiu et al., 2010; Gerby et al., 2011). Nonetheless, it is interesting to note that Notch signaling has been shown to contribute to human T-ALL LIC activity in nonobese diabetic (NOD)/Scid transplantation assays (Armstrong et al., 2009). As Notch signaling is mediated through transcriptional activation, identification of downstream target genes has received much attention. In several such studies, c-Myc has been identified as a direct transcriptional target of Notch1 (Palomero et al., 2006b; Sharma et al., 2006; Weng et al., 2006), and may constitute part of a self-renewal genetic program similar to that demonstrated in induced pluripotent stem cells (Takahashi and Yamanaka, 2006; Takahashi et al., 2007). Notch may also repress PTEN (Palomero et al., 2007), and thus potentiate PI3K–Akt/mTOR signaling which may also promote LIC activity (Yilmaz et al., 2006).
Most prior studies have focused on signaling via IL-7 (Dibirdik et al., 1991; Barata et al., 2004a,b,c, 2005; González-Garcia et al., 2009; Shochat et al., 2011; Silva et al., 2011) or mutational activation of intermediates in the PI3K–Akt pathway (Palomero et al., 2007; Gutierrez et al., 2009). The importance of PI3K–Akt activation in T-ALL is underscored by multiple studies showing that PI3K–Akt/mTOR inhibitors block growth/survival of T-ALL cells (Avellino et al., 2005; Wei et al., 2006; Palomero et al., 2007; Chiarini et al., 2009; Cullion et al., 2009). Notably, our observation that complete inhibition of IGF1R signaling blocks growth/survival of bulk leukemia cells confirms previous results with PI3K–Akt/mTOR inhibitors, and further illustrates that IGF1-dependent activation contributes in a substantive way to net PI3K–Akt signaling output. Perhaps most intriguing, however, is our observation that moderately decreased IGF1R signaling (as modeled by IGF1Rneo/neo leukemias) results in selective loss of LIC activity, yet leaves the growth and survival of bulk cells relatively unaffected. These findings suggest that LICs in T-ALL may be uniquely sensitive to inhibition of IGF1R signaling, and raise the possibility that pharmacologic IGF1R inhibitors now in clinical development could, in combination with standard regimens, enhance initial response to therapy and reduce rates of disease relapse. Importantly, normal hematopoietic stem cell function appears not to be affected in IGF1Rneo/neo mice (unpublished data), suggesting a therapeutic window exists in which IGF1R inhibitors block LIC activity yet have minimal effects on normal hematopoiesis.
MATERIALS AND METHODS
Mice.
The IGF1Rneo line contains a PGK-neo-poly(A) expression cassette integrated within the second intron, resulting in decreased expression of full-length IGF1R (Holzenberger et al., 2000). The IGF1Rneo allele has been backcrossed onto the C57BL/6 background for over 20 generations. Rosa26CreERT mice were obtained from The Jackson Laboratory. All transplants involving mouse cells were performed using C57BL/6J (B6) or B6(Cg)-Tyrc-2J/J (“B6-albino”) recipient mice (The Jackson Laboratory). Transplants involving human cells were performed using NOD-Scid/IL2Rγc−/− recipient mice. Animals were housed under specific pathogen–free conditions at the BC Cancer Agency or Dana-Farber Cancer Institute animal facilities, and experimental studies performed under protocols approved by the University of British Columbia or Dana-Farber Institutional Review Boards, respectively. IGF1R genotyping was performed by multiplex assay with primers YNex (5′-CCATGGGTGTTA AATGTTAATGGC-3′), YNvl (5′-ATGAATGCTGGTGAGGGTTGTCTT-3′) and YNmt2 (5′-ATCTTGGAGTGGTGGGTCTGTTTC-3′), as previously described (Leneuve et al., 2001). Rosa26 genotyping was performed with primers RcreFW (5′-AAAGTCGCTCTGAGTTGTTAT-3′) and RcreRV (5′-GCGAAGAGTTTGTCCTCAACC-3′) for the CreERT allele, and with RwtFW (5′-GCACTTGCTCTCCCAAAGTC-3′) and RwtRV (5′-GGCGGATCACAAGCAATAAT-3′) for the WT allele.
Plasmids.
Retroviral vectors all used the MSCV-IRES-GFP (Mig) backbone. Mig ICN1, Mig ΔE, and MSCV-DN-MAML1-GFP have been described previously (Aster et al., 1997; Weng et al., 2003). Mig IGF1R was generated by subcloning a 4.2-kb NotI-BamHI fragment containing the human IGF1R cDNA from pBABE-bleo IGF1R (Addgene) into the Mig vector.
Retroviral transduction/bone marrow transplantation.
High titer, replication-defective retroviral supernatants were produced by transient transfection of PlatE cells as described previously (Medyouf et al., 2010). MSCV-based retroviral expression vectors included IRES-GFP cassettes for fluorescent tagging of transduced cells. Retroviral transduction of 5-fluorouracil-treated or lineage-depleted (CD4, CD8, CD11b, B220, Ter119, and Gr1) bone marrow cells was performed as described previously (Medyouf et al., 2010). For primary transplants, WT C57BL/6 recipient mice were lethally irradiated (810 rads), and then injected intravenously with 30,000–45,000 transduced GFP+ cells along with a minimum of 105 syngeneic whole bone marrow cells to ensure hematopoietic reconstitution. For serial transplants, 0.5–1.0 × 106 GFP+ leukemia cells (from spleen or bone marrow of a moribund leukemic mouse) were injected into the tail vein (i.v.) or intramedullary space of the femur (intrafemoral [IF]) of nonirradiated C57BL/6 recipients. We define LIC activity here as the ability to produce aggressive disease in syngeneic/congenic secondary recipients under these transplant conditions within 20 wk.
Human leukemia samples.
Cryopreserved lymphoblast samples were provided by collaborating institutions. Primary samples were obtained at initial diagnosis with informed consent from patients or their legal guardians or as discarded pathological material under approved Institutional Review Board protocols at the Karmanos Cancer Institute, Hôpital Armand-Trousseau, Hopital Saint-Louis, BC Cancer Agency, and BC Children’s and Women’s Hospital following guidelines established by the Declaration of Helsinki. Expansion of primary human T-ALL cells in irradiated NOD-Scid/IL2Rγc−/− mice has been described previously (Medyouf et al., 2010).
In vitro culture of primary T-ALL cells.
Primary human T-ALL cells were cultured on MS5-DL1/MS5 stromal feeder cells as previously described (Medyouf et al., 2010). Primary mouse T-ALL cells were cultured without feeders in complete media with supplemental cytokines.
Cell lines.
IGF1R knockout mouse embryo fibroblasts (R-cells) that were engineered to re-express human IGF1R (R+) were a gift from P. Sorensen (BC Cancer Agency, Vancouver, British Columbia, Canada). All established T-ALL cell lines were grown in RPMI 1640 medium supplemented with 10% FCS, 1 mM sodium pyruvate, 2 mM l-glutamine, and antibiotics.
Ligand stimulation assay.
Leukemia cells were serum starved overnight, and then stimulated for 10 min with either recombinant IGF-1 (PeproTech), recombinant insulin (Sigma-Aldrich), or fetal calf serum (Invitrogen), at indicated doses. Cells were then fixed immediately by addition of formaldehyde (Electron Microscopy Services) to 1.5%, and permeabilized with ice-cold methanol for at least 1 h before analysis by flow cytometry.
In vitro proliferation/apoptosis assays.
Cells were pulsed with 10 mM BrdU for 1 h at 37°C, and then fixed/stained with anti-BrdU antibody according to manufacturer instructions (FITC or APC BrdU Flow kit; BD) and analyzed by flow cytometry with gating for mouse and human leukemia cells by GFP- and hCD45-positive events, respectively. Cell growth was also measured by CellTiter-Blue assay (Promega) or Ki67-Alexa Fluor 647/Hoechst staining (BD Biosciences). Apoptotic cells were identified by staining with Annexin V-FITC/7-aminoactinomycin or active caspase-3-PE (BD). Cell viability was assessed by propidium iodide exclusion and analysis by flow cytometry.
Antibody reagents.
Antibodies directed against the following proteins were used: IGF1Rα (sc-712, Santa Cruz Biotechnology), PTEN (Y184, Abcam), and ERK2 (sc-154, Santa Cruz Biotechnology) for Western blots, and hCD45 (eBioscience), CD4 (L3T4; eBioscience), CD8a (53–6.7, BioLegend), IGF1Ra (αIR3; EMD), and P-AKT (S473; Cell Signaling Technology) for flow cytometry.
Flow cytometry.
Acquisition was performed on FACSCalibur or LSRII cytometers (BD) and data analyzed using FlowJo software (Tree Star, Inc.).
Drugs.
IGF1R inhibitors BMS-536924 and BMS-754807 were obtained under Material Transfer Agreement from the manufacturer. BMS-536924 was resuspended in DMSO at 10 mM, then serially diluted in Hank’s Balanced Salt Solution (Invitrogen) before addition to culture media. BMS-754807 was resuspended in PEG400:H2O (80:20 by volume) at 5 mg/ml final concentration before administration to mice. An azide-free preparation of αIR3 antibody was used for IGF1R blocking studies (EMD). γ-Secretase inhibitor XXI (compound E) was used at 1.0 µM final concentration for all studies (EMD). GSI washout was performed by washing cells twice with prewarmed, complete culture medium. For induction of CreERT activity in vivo, 1 g tamoxifen (Sigma-Aldrich) was admixed per 1 kg mouse chow (Altromin) and mice were allowed to feed ad libitum. For induction of CreERT activity in vitro, 4-OHT (Sigma-Aldrich) was dissolved in ethanol and added to culture media at 50 nM final concentration.
Expression profiling.
Total RNA was isolated by TRIzol (Invitrogen) extraction or RNeasy Mini kit (QIAGEN) and submitted to the McGill University/Génome Québec Innovation Centre or Harvard Medical School Biopolymers Core for expression profiling using Affymetrix HG-U133 Plus 2.0 GeneChips. Data were analyzed using dChip software (Schadt et al., 2001). Microarray data are available from the Gene Expression Omnibus accession no. GSE29959.
Quantitative real-time PCR.
Total RNA was extracted after cell lysis in TRIzol reagent (Invitrogen). First-strand cDNA was generated by reverse transcription with SuperScript III (Invitrogen) using a mix of random 15-mer and anchored oligo(dT)20+1 primers, and then amplified using Platinum SYBR Green qPCR SuperMix-UDG (Invitrogen) and the following specific primer sets: hIGF1R forward 5′-ACTTACTCGGACGTCTGGTCCTTC-3′, hIGF1R reverse 5′-ATCTTGGGGTTATACTGCCAGCAC-3′ for IGF1R, and hActB RT5 5′-CGCGAGAAGATGACCCAGAT-3′plus hActB RT3 5′-GAT AGC ACA GCC TGG ATA GCA AC-3′ for β-actin. Each sample was assayed in triplicate using a Dyad Disciple thermal cycler equipped with Chromo4 optical head (Bio-Rad Laboratories). Expression levels were calculated by the ΔΔCt method with normalization to β-actin.
ChIP.
ChIP was performed with the ChIP Assay kit (Millipore). In brief, cells were cross-linked with 1% formaldehyde for 10 min at 37°C, lysed in 1% SDS, and 10 mM EDTA, 50 mM Tris, pH 8.1, and then sonicated to obtain DNA fragments from 200–600 bp. Chromatin was then immunoprecipitated with the following antibodies: Notch1(Tc; Aster et al., 1997), CSL (gift from E. Kieff, Brigham and Women’s Hospital, Boston, MA), H3K4me1 (ab8895; Abcam), H3K4me2 (07–030; Millipore), H3K4me3 (07–745; Millipore), CBP (ab10489; Abcam), p300 (sc-585; Santa Cruz Biotechnology, Inc.), RNA pol2 (05623B; Millipore), and mouse IgG (12–371; Millipore) as control. After overnight incubation with antisera at 4°C, immunoprecipitated chromatin was captured with Protein A-agarose beads, washed, and eluted. After reversal of cross-links, DNA was purified using the QIAquick PCR purification kit (QIAGEN). Input control DNA was also prepared in parallel, omitting the immunoprecipitation steps. Primers used for qPCR after ChIP were as follows: IGF1Rchip forward 5′-GGTGGGTGAGGGAGAGCGGT-3′ and IGF1Rchip reverse 5′-GGCTGCGTCCCAGGCAGTTT-3′.
ChIP-Seq and data analysis.
ChIP-Seq libraries were prepared according to the Illumina ChIP DNA library preparation kit. After addition of adaptors, libraries were amplified by 18 cycles of PCR, size selected (150–250 bp) by electrophoresis, and purified using a QIAGEN gel extraction kit. After quality control testing on an Agilent 2100 Bioanalyzer, the library was subjected to deep sequencing using an Illumina Genome Analyzer II in the Harvard Medical School Biopolymers Core facility. Sequencing reads were aligned to human genome build hg18 and analyzed using CisGenome (Ji et al., 2008). One-sample and two-sample analyses were performed using 100-bp windows and reads of >10 bp.
Online supplemental material.
Fig. S1 shows Akt response to stimulation with IGF1 and insulin and effects of BMS-536924 inhibitor in T-ALL cells. Fig. S2 shows effects of the BMS-536924 inhibitor on T-ALL cell growth and viability in vitro. Fig. S3 shows resistance to growth inhibition by IGF1R blocking antibody correlates with reduced surface IGF1R expression. Fig. S4 shows effects of IGF1R deletion on T-ALL development in vivo and cell growth/viability in vitro. Fig. S5 demonstrates in vivo efficacy of the IGF1R inhibitor BMS-754807 in a mouse T-ALL model. Fig. S6 shows that IGF1R is a Notch target gene in an independent expression profiling dataset. Fig. S7 shows IGF1R is a Notch target gene in the human T-ALL cell line, CUTLL1. Fig. S8 shows Western blot analysis of total IGF1R expression after GSI treatment of human T-ALL cell lines. Fig. S9 shows chromatin modification marks over the human IGF1R locus in the vicinity of the Notch/CSL binding site. Table S1 shows survival data for all secondary, tertiary, and quarternary transplant experiments. Table S2 lists genes whose mRNA expression level decreases most after inhibition of Notch signaling.
Acknowledgments
We thank Amina Kariminia (BC Children’s Hospital, Vancouver) for sample preparation, Drs. Kirk Schultz (BC Children’s Hospital, Vancouver), Larry H. Matherly (Karmanos Cancer Institute, Detroit), Paola Ballerini (Hôpital Armand-Trousseau, Paris), and Thierry Leblanc (Hopital Saint-Louis, Paris) generously contributed human T-ALL samples.
This work was funded by grants from the Canadian Cancer Society Research Institute/Terry Fox Foundation, US National Cancer Institute (K22CA112538, P01CA119070), Leukemia and Lymphoma Society of Canada, Lymphoma Foundation Canada, Cancer Research Society, and Agence Nationale de la Recherche (NT05-3 42491). H. Medyouf is supported by a Human Frontier Science Program Fellowship. J.C. Aster is supported by grants from the Leukemia and Lymphoma Society and the William Lawrence Foundation. A.P. Weng is a Michael Smith Foundation for Health Research Scholar.
J. Carboni and M. Gottardis are employees of Bristol-Myers Squibb Company. The remaining authors have no competing interests to declare.
Author contributions: H. Medyouf designed and performed experiments, analyzed data, and wrote the manuscript. S. Gusscott, H. Wang, C. Wai, and O. Nemirovsky performed experiments. J. Carboni and M. Gottardis developed and provided the BMS inhibitor compounds. A. Trumpp, F. Pflumio, M. Pollak, and M. Holzenberger provided reagents and resources and discussed results. J. Tseng and A. Kung designed and performed in vivo experiments with BMS-754807. J. Aster designed ChIP experiments, analyzed data, and discussed results. A. Weng supervised the project, designed and performed experiments, analyzed data, and wrote the manuscript.
References
Author notes
H. Medyouf’s present address is Heidelberg Institute for Stem Cell Technology and Experimental Medicine (HI-STEM), and Division of Stem Cells and Cancer, German Cancer Research Center (DKFZ), D-69120 Heidelberg, Germany