During asymmetric cell division, the mitotic spindle and polarized myosin can both determine the position of the cytokinetic furrow. However, how cells coordinate signals from the spindle and myosin to ensure that cleavage occurs through the spindle midzone is unknown. Here, we identify a novel pathway that is essential to inhibit myosin and coordinate furrow and spindle positions during asymmetric division. In Caenorhabditis elegans one-cell embryos, myosin localizes at the anterior cortex whereas the mitotic spindle localizes toward the posterior. We find that PAR-4/LKB1 impinges on myosin via two pathways, an anillin-dependent pathway that also responds to the cullin CUL-5 and an anillin-independent pathway involving the kinase PIG-1/MELK. In the absence of both PIG-1/MELK and the anillin ANI-1, myosin accumulates at the anterior cortex and induces a strong displacement of the furrow toward the anterior, which can lead to DNA segregation defects. Regulation of asymmetrically localized myosin is thus critical to ensure that furrow and spindle midzone positions coincide throughout cytokinesis.
Introduction
During asymmetric cell division, establishment of a polarity axis and proper orientation of the mitotic spindle before mother cell division are key steps to allow the unequal inheritance of cell fate determinants between the two daughter cells (Knoblich, 2010). Asymmetric cell division often leads to size asymmetry of the daughter cells, implying that the spindle midzone is not symmetrically positioned within the mother cell. In some instances, such as in the one-cell Caenorhabditis elegans embryo, the spindle is built in a symmetric manner and pulled toward one side of the mother cell (Knoblich, 2010). Other asymmetrically dividing cells, such as Drosophila melanogaster neuroblasts, have a spindle that elongates asymmetrically (Knoblich, 2010). In both cases, it is critical that the cytokinetic furrow aligns with the spindle midzone to ensure both asymmetric cell division and proper DNA segregation. Two pathways have been shown to coordinate spindle and furrow positions by inducing cytokinesis furrow formation in the vicinity of the spindle midzone: the centralspindlin and the astral microtubule pathway (Dechant and Glotzer, 2003). The centralspindlin complex, formed by the kinesin MKLP-1/ZEN-4 and MgcRacGAP/CYK-4, is localized at both the spindle midzone and the equatorial cortex where it triggers the accumulation of active RhoA and contractile ring components (Yüce et al., 2005; Nishimura and Yonemura, 2006; Basant et al., 2015). Astral microtubules have been proposed to inhibit the accumulation of contractile ring proteins at the poles of the dividing cell (Werner et al., 2007; Lewellyn et al., 2010).
A spindle-independent furrowing mechanism has also been described in Drosophila and C. elegans neuroblasts (Cabernard et al., 2010; Ou et al., 2010). In these cells, myosin accumulates asymmetrically at the cell cortex during early anaphase. It then drives furrow contraction and asymmetric elongation of the spindle. As a consequence, neuroblasts divide asymmetrically, giving rise to a smaller daughter cell on the side on which myosin has accumulated. The ability of myosin to induce cytokinesis suggests the existence of regulatory mechanisms to prevent it from inducing cytokinesis in an inappropriate manner. Such regulation may be particularly critical in cells in which myosin localization does not correlate with spindle position and where furrow localization may thus result from a tug-of-war between the signals emanating from myosin and the spindle.
The one-cell C. elegans embryo is a well-established model to study the different aspects of asymmetric cell division. After fertilization, myosin II (NMY-2) flows toward the anterior pole of the embryo and leads to the asymmetric distribution of polarity proteins: PAR-3, PAR-6, and PKC-3 accumulate at the anterior cortex, whereas PAR-2 and PAR-1 localize at the posterior cortex (Kemphues, 2000; Munro et al., 2004). Independently of myosin, posterior microtubules also contribute to the posterior accumulation of PAR-2 (Motegi et al., 2011). In turn, polarity proteins control the forces that are exerted on the mitotic spindle to pull it toward the posterior pole of the embryo (Grill et al., 2001). The centralspindlin and astral microtubule pathways then induce cytokinesis (Dechant and Glotzer, 2003), and the furrow ingresses through the spindle midzone and gives rise to a large anterior cell (AB) and a small posterior cell (P1). In contrast to what happens in neuroblasts, furrow ingression occurs in the posterior half of the embryo, despite myosin being present at the anterior cortex.
Another essential PAR protein, the kinase PAR-4/LKB1, uniformly localizes at the one-cell embryo cortex and mildly modulates the establishment of polarity through the regulation of actomyosin contractions and anillins (Morton et al., 1992; Watts et al., 2000; Chartier et al., 2011). Drosophila and mammalian anillin interact with nonmuscle myosin and several essential players of cytokinesis, including septins, the centralspindlin subunit MgcRacGAP/CYK-4, and RhoA (Piekny and Maddox, 2010). In C. elegans, the anillin ANI-1 carries all the characteristic binding domains of anillin and is required for cortical actomyosin contractility during polarity establishment; it is also present at the cytokinesis furrow (Maddox et al., 2005). ANI-2 is a shorter form of anillin that lacks the N-terminal actin and myosin binding domains. During polarity establishment, PAR-4 positively regulates actomyosin contractions by inhibiting ANI-2, which in turn has been proposed to inhibit ANI-1, possibly by acting as a competitor (Chartier et al., 2011; Amini et al., 2014).
Here we identify a novel regulatory pathway that is essential to maintain a low level of myosin at the anterior cortex during cytokinesis and we find that excessive myosin activity shifts the furrow toward the anterior of the embryo. Our work thus demonstrates that the regulation of asymmetrically localized myosin is critical to ensure that the positions of the furrow and of the spindle midzone coincide throughout cytokinesis.
Results
PAR-4 and CUL-5 are required to coordinate spindle and furrow positions
cullin-5 (cul-5) deletion mutants are viable and do not display obvious defects during embryonic development; however, they enhance embryonic lethality caused by weak cul-2 depletion (Sasagawa et al., 2007), indicating that CUL-5 plays a role, albeit nonessential on its own, during embryonic development. To decipher the role of CUL-5, we tested possible genetic interactions with different par mutants and found that the loss of CUL-5 strongly enhances par-4 mutant defects. cul-5(ok1706) embryos have a very low embryonic lethality and par-4(RNAi) or par-4(it47) thermosensitive embryos grown at permissive temperature (15°C) show 27% and 56% embryonic lethality, respectively. cul-5(ok1706) par-4(RNAi) and cul-5(ok1706) par-4(it47) double mutant embryos show stronger embryonic lethality, at 67% and 95%, respectively (Fig. S1 A). We next recorded the first division of cul-5(ok1706) par-4(it47) embryos by differential interference contrast (DIC) microscopy at semi-permissive temperature (20°C) and measured the position of the furrow once it spanned the entire embryo. Like wild-type embryos, single cul-5(ok1706) mutants divide asymmetrically (Fig.1, A and B; and Video 1). In par-4(it47) single mutants the cytokinetic furrow is slightly shifted toward the anterior (Fig.1, A and B; and Video 1). The furrow is localized further toward the anterior in cul-5(ok1706) par-4(it47) double mutants (Fig.1, A and B; and Video 1). Similar results were obtained at restrictive temperature (25°C; unpublished data) and upon par-4 RNAi (Fig. S1 C). Thus, both CUL-5 and PAR-4 are involved in regulating the position of the cytokinetic furrow during asymmetric cell division.
CUL-5/RBX-2 and PAR-4 regulate cytokinetic furrow position (related to Fig. 1 A). DIC recordings of wild-type, cul-5(ok1706), rbx-2(ok1617), par-4(it47), cul-5(ok1706) par-4(it47) , and rbx-2(ok1617); par-4(it47) dividing one-cell embryos. Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
CUL-5/RBX-2 and PAR-4 regulate cytokinetic furrow position (related to Fig. 1 A). DIC recordings of wild-type, cul-5(ok1706), rbx-2(ok1617), par-4(it47), cul-5(ok1706) par-4(it47) , and rbx-2(ok1617); par-4(it47) dividing one-cell embryos. Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
CUL-5/RBX-2 and PAR-4 regulate cytokinetic furrow position. (A and B) The furrow is shifted toward the anterior in cul-5(ok1706) par-4(it47) and rbx-2(ok1617); par-4(it47) embryos. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow spanned the entire embryo. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. As depicted in the inset (B), furrow position is the distance between the anterior pole and the furrow (AB size) expressed as a percentage of total embryo length (0% corresponds to the anterior pole, 100% to the posterior pole). See also Fig. S1 and Video 1. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D) Localization of the spindle midzone protein SPD-1 and of DNA during anaphase is normal in cul-5(ok1706) par-4(it47) embryos. Confocal section of wild-type and cul-5(ok1706) par-4(it47) one-cell anaphase embryos stained for α-tubulin (red), SPD-1 (green), and DNA (blue). More than 10 anaphase embryos per genotype were observed and representative images are shown. (E and F) Furrow and spindle positions are uncoupled in cul-5(ok1706) par-4(it47) embryos. Superposed confocal sections and DIC images (E) and quantification of furrow/spindle coupling (F) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Green dashed lines correspond to the spindle center and arrowheads to furrow position. As depicted in the inset (F), the distance between the anterior aster and the furrow (A-F) was measured during ingression and expressed as a percentage of spindle length (50% corresponds to the furrow position being coupled with spindle center, below 50% corresponds to a shift of the furrow toward the anterior, and above 50% corresponds to a shift toward the posterior). See also Video 2.In all images, embryos are oriented with the anterior to the left. Bars, 10 µm. P-values from Student’s t test.
CUL-5/RBX-2 and PAR-4 regulate cytokinetic furrow position. (A and B) The furrow is shifted toward the anterior in cul-5(ok1706) par-4(it47) and rbx-2(ok1617); par-4(it47) embryos. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow spanned the entire embryo. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. As depicted in the inset (B), furrow position is the distance between the anterior pole and the furrow (AB size) expressed as a percentage of total embryo length (0% corresponds to the anterior pole, 100% to the posterior pole). See also Fig. S1 and Video 1. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D) Localization of the spindle midzone protein SPD-1 and of DNA during anaphase is normal in cul-5(ok1706) par-4(it47) embryos. Confocal section of wild-type and cul-5(ok1706) par-4(it47) one-cell anaphase embryos stained for α-tubulin (red), SPD-1 (green), and DNA (blue). More than 10 anaphase embryos per genotype were observed and representative images are shown. (E and F) Furrow and spindle positions are uncoupled in cul-5(ok1706) par-4(it47) embryos. Superposed confocal sections and DIC images (E) and quantification of furrow/spindle coupling (F) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Green dashed lines correspond to the spindle center and arrowheads to furrow position. As depicted in the inset (F), the distance between the anterior aster and the furrow (A-F) was measured during ingression and expressed as a percentage of spindle length (50% corresponds to the furrow position being coupled with spindle center, below 50% corresponds to a shift of the furrow toward the anterior, and above 50% corresponds to a shift toward the posterior). See also Video 2.In all images, embryos are oriented with the anterior to the left. Bars, 10 µm. P-values from Student’s t test.
We also found that the loss of the RING-containing protein RBX-2, which associates with CUL-5 in ubiquitine ligase complexes (Kamura et al., 2004), increases the embryonic lethality of par-4(RNAi) and par-4(it47) embryos (Fig. S1 B). Moreover, whereas rbx-2(ok1617) mutants divide asymmetrically, the cytokinetic furrow is shifted toward the anterior in rbx-2(ok1617); par-4(it47) and rbx-2(ok1617); par-4(RNAi) embryos, similar to cul-5(ok1706) par-4(it47) and cul-5(ok1706) par-4(RNAi) embryos (Fig. 1, A and B; Video 1; and Fig. S1 C). These results suggest that CUL-5 and RBX-2 may function in the same ubiquitin ligase complex to regulate cytokinetic furrow position.
To test whether the furrow position defects of cul-5(ok1706) par-4(it47) embryos were a result of the mispositioning of the mitotic spindle along the anteroposterior axis of the embryo, we measured the position of the spindle at the onset of cytokinesis in strains expressing an α-tubulin::YFP (α-tub::YFP) transgene. Surprisingly, we found the spindle to be correctly located toward the posterior pole in α-tub::YFP; cul-5(ok1706) par-4(it47) embryos, similar to α-tub::YFP, α-tub::YFP; cul-5(ok1706), and α-tub::YFP; par-4(it47) embryos (Fig. 1 C). Furthermore, both in wild-type and cul-5(ok1706) par-4(it47) embryos, the spindle appears symmetrical, with the midzone localized centrally and the two sets of chromosomes distributed at equal distance from the midzone during anaphase (Fig. 1 D). Altogether, our results show that furrow mispositioning in cul-5(ok1706) par-4(it47) embryos is not caused by defects in spindle position or general organization.
To compare spindle and furrow positions, we measured the distance between the anterior spindle aster and the furrow during ingression and compared it to spindle length (Fig. 1 F, inset). In α-tub::YFP and α-tub::YFP; cul-5(ok1706) embryos, the anterior aster-furrow distance is close to 50% of spindle length, indicating that furrow position correlates with the spindle center (Fig. 1, E and F; and Video 2). In α-tub::YFP; par-4(it47) embryos, the anterior aster-furrow distance is slightly shorter. It is further decreased in α-tub::YFP; cul-5(ok1706) par-4(it47) embryos (Fig. 1, E and F; and Video 2), showing that the furrow is shifted toward the anterior compared with the spindle center. Thus the position of the cytokinetic furrow does not coincide with the spindle center in cul-5(ok1706) par-4(it47) mutants, a phenotype that to our knowledge has never been described thus far.
Video 2 - CUL-5 and PAR-4 regulate cytokinetic furrow position (related to Fig. 1 E). Superposed DIC and confocal recordings of control, cul-5(ok1706), par-4(it47), and cul-5(ok1706) par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Video 2 - CUL-5 and PAR-4 regulate cytokinetic furrow position (related to Fig. 1 E). Superposed DIC and confocal recordings of control, cul-5(ok1706), par-4(it47), and cul-5(ok1706) par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Furrow mispositioning is not caused by defects in the centralspindlin or astral microtubule pathway
Two redundant pathways, depending on centralspindlin and astral microtubules, induce cytokinesis in the early C. elegans embryo (Dechant and Glotzer, 2003). We tested whether either of these pathways was affected in cul-5(ok1706) par-4(it47) embryos. Inactivation of the centralspindlin protein ZEN-4/MKLP1 does not prevent furrow ingression but leads to furrow regression during the late steps of cytokinesis (Raich et al., 1998; Fig. 2 A). cul-5(ok1706) par-4(it47) mutants undergo complete cytokinesis (Fig. 2 A), suggesting that the centralspindlin pathway is functional in these embryos. To uncover possible subtle defects in this pathway, we tested whether inactivating the astral microtubule pathway in cul-5(ok1706) par-4(it47) embryos leads to cytokinesis defects. Indeed, inactivating astral microtubule signaling by depleting the serine-rich protein NOP-1 or the DEP domain protein LET-99, for instance, does not prevent cytokinesis on its own but blocks furrow ingression when centralspindlin signaling is also impaired (Bringmann et al., 2007; Tse et al., 2012; Fig. 2 A). However, neither nop-1 nor let-99(RNAi) affects furrow ingression in cul-5(ok1706) par-4(it47) embryos (Fig. 2 A), showing that centralspindlin signaling is fully functional in these embryos. To test whether astral microtubule signaling is defective in cul-5(ok1706) par-4(it47) mutants, we performed the inverse experiment. Inactivating centralspindlin using zen-4 RNAi leads to complete furrow ingression followed by regression in a wild-type background and completely inhibits furrow ingression in embryos with inactive astral microtubule signaling (nop-1(it142) mutants; Tse et al., 2012; Fig. 2 B). When zen-4 RNAi was performed in cul-5(ok1706) par-4(it47), the furrow either ingressed completely (5/23 embryos) or partially (18/23 embryos) before regressing (Fig. 2 B). Thus the astral microtubule pathway is active in cul-5(ok1706) par-4(it47) embryos, albeit to a lower extent than in wild type.
Inhibition of centralspindlin or astral microtubule pathway does not account for furrow mispositioning. (A) Inactivation of the astral microtubule pathway by nop-1 and let-99 RNAi does not inhibit furrow ingression in cul-5(ok1706) par-4(it47) one-cell embryos. nop-1 and let-99 RNAi effectively inhibited astral microtubule signaling as judged by their ability to inhibit furrow ingression in zen-4(or153) mutant. (B) Inactivation of the centralspindlin pathway by zen-4 RNAi partially inhibits furrow ingression in cul-5(ok1706) par-4(it47) one-cell embryos. zen-4 RNAi effectively inhibited centralspindlin signaling as the furrow regresses in zen-4(RNAi) embryos and does not ingress in nop-1(it142); zen-4(RNAi) embryos. (C–F) Inactivation of centralspindlin or the astral microtubule pathway does not shift the furrow toward the anterior. Furrow position was measured in zen-4(or153) and nop-1(it142) mutants (C) and in wild-type, cul-5(ok1706), or par-4(it47) embryos treated with zen-4 (D), nop-1 (E), or let-99 (F) RNAi. Furrow position was measured when the furrow spanned the entire embryo. P-values from Student’s t test.
Inhibition of centralspindlin or astral microtubule pathway does not account for furrow mispositioning. (A) Inactivation of the astral microtubule pathway by nop-1 and let-99 RNAi does not inhibit furrow ingression in cul-5(ok1706) par-4(it47) one-cell embryos. nop-1 and let-99 RNAi effectively inhibited astral microtubule signaling as judged by their ability to inhibit furrow ingression in zen-4(or153) mutant. (B) Inactivation of the centralspindlin pathway by zen-4 RNAi partially inhibits furrow ingression in cul-5(ok1706) par-4(it47) one-cell embryos. zen-4 RNAi effectively inhibited centralspindlin signaling as the furrow regresses in zen-4(RNAi) embryos and does not ingress in nop-1(it142); zen-4(RNAi) embryos. (C–F) Inactivation of centralspindlin or the astral microtubule pathway does not shift the furrow toward the anterior. Furrow position was measured in zen-4(or153) and nop-1(it142) mutants (C) and in wild-type, cul-5(ok1706), or par-4(it47) embryos treated with zen-4 (D), nop-1 (E), or let-99 (F) RNAi. Furrow position was measured when the furrow spanned the entire embryo. P-values from Student’s t test.
We next tested whether the inactivation of centralspindlin or of the astral microtubule pathway could lead to furrow mispositioning. Centralspindlin (zen-4(or153)) and astral microtubule (nop-1(it142)) defective mutants divide asymmetrically (Severson et al., 2000; Tse et al., 2012; Fig. 2 C). Moreover, inactivation of centralspindlin (zen-4 RNAi) or astral microtubule signaling (nop-1 and let-99 RNAi) in either par-4(it47) or cul-5(ok1706) single mutants does not shift the furrow toward the anterior (Fig. 2, D–F). In conclusion, although astral microtubule signaling is only partially active in cul-5(ok1706) par-4(it47) embryos, our data indicate that defects in either centralspindlin or astral microtubule signaling cannot account for the furrow positioning defects observed in these embryos.
PAR-4 and CUL-5 prevent cortical accumulation of ANI-2
We next sought to identify the mechanism by which PAR-4 and CUL-5 regulate furrow position. PAR-4 has been previously shown to be a negative regulator of ANI-2 (Chartier et al., 2011). ANI-2 is barely detected in wild-type one- and two-cell embryos but a weak accumulation of ANI-2 is observed at the cortex between the anterior and posterior cells in par-4(it47) two-cell embryos (Fig. 3, A and B; Chartier et al., 2011). We found that ANI-2 also accumulates at this cortex in cul-5(ok1706) embryos (Fig. 3, A and B). Furthermore, we observed a strong cortical accumulation of ANI-2 in cul-5(ok1706) par-4(it47) double mutants (Fig. 3, A and B). Thus PAR-4 and CUL-5 are both negative regulators of ANI-2 (see Fig. 8 E). No increase in total ANI-2 protein levels could be detected in cul-5(ok1706) par-4(it47) embryonic extracts (Fig. S1 D), suggesting that CUL-5 and PAR-4 may not regulate the global stability of ANI-2 but rather its accumulation at the cortex.
ANI-2 accumulates and is required to misposition the furrow in cul-5 par-4 embryos. (A and B) ANI-2 strongly accumulates at the cortex between AB and P1 in cul-5(ok1706) par-4(it47) embryos. Confocal sections of two-cell embryos (A) of the indicated genotypes stained with anti-ANI-2 antibody and quantification (B) of ANI-2 cortical accumulation (see Materials and methods for details). Most wild-type embryos show no ANI-2 staining. ANI-2 weakly accumulates in cul-5(ok1706) and par-4(it47) embryos (arrowheads) and strongly in cul-5(ok1706) par-4(it47) embryos. Bars, 10 µm. See also Fig. S1 D. (C) ani-2 RNAi suppresses furrow position defects of cul-5(ok1706) par-4(it47) embryos. Furrow position was measured when the furrow spanned the entire embryo. P-values from Student’s t test.
ANI-2 accumulates and is required to misposition the furrow in cul-5 par-4 embryos. (A and B) ANI-2 strongly accumulates at the cortex between AB and P1 in cul-5(ok1706) par-4(it47) embryos. Confocal sections of two-cell embryos (A) of the indicated genotypes stained with anti-ANI-2 antibody and quantification (B) of ANI-2 cortical accumulation (see Materials and methods for details). Most wild-type embryos show no ANI-2 staining. ANI-2 weakly accumulates in cul-5(ok1706) and par-4(it47) embryos (arrowheads) and strongly in cul-5(ok1706) par-4(it47) embryos. Bars, 10 µm. See also Fig. S1 D. (C) ani-2 RNAi suppresses furrow position defects of cul-5(ok1706) par-4(it47) embryos. Furrow position was measured when the furrow spanned the entire embryo. P-values from Student’s t test.
To determine whether ANI-2 is responsible for furrow mispositioning in cul-5(ok1706) par-4(it47) embryos, we tested whether ANI-2 depletion can restore normal furrow position in those embryos. ani-2(RNAi) does not lead to changes in furrow position on its own but furrows are localized more posteriorly in ani-2(RNAi); cul-5(ok1706) par-4(it47) embryos than in cul-5(ok1706) par-4(it47) embryos (Fig. 3 C). ani-2(RNAi) thus rescues the defects of cul-5(ok1706) par-4(it47) embryos, indicating that ANI-2 contributes to furrow mispositioning in cul-5(ok1706) par-4(it47) embryos.
PAR-4 and ANI-1 both regulate furrow position
Previous work suggested that ANI-2 is a negative regulator of ANI-1 (Amini et al., 2014). Cortical accumulation of ANI-2 in cul-5(ok1706) par-4(it47) embryos raises the possibility that ANI-1 may be inhibited in those embryos. However, consistent with a previous study (Maddox et al., 2005), we find that ani-1 depletion alone does not shift the furrow toward the anterior (Fig. 4, A and B; and Video 3). Thus inhibition of ANI-1 is not sufficient to explain furrow mispositioning in cul-5(ok1706) par-4(it47) embryos. We therefore hypothesized that par-4 and/or cul-5 might regulate furrow position not only via the regulation of anillins but also via an anillin-independent pathway. To test this hypothesis, we depleted ANI-1 in cul-5(ok1706) and in par-4(it47) single mutant embryos grown at 20°C. Although ani-1(RNAi) does not change furrow position in cul-5(ok1706) mutants, we observed strong furrow mispositioning in ani-1(RNAi); par-4(it47) embryos (Fig. 4, A and B; and Video 3). In most embryos, the furrow was even observed in the anterior half of the embryo. Similar results were obtained when ANI-1 was depleted in another par-4 mutant, par-4(it57) (Fig. S2 A).
The loss of PAR-4 and ANI-1 leads to strong furrow mispositioning (related to Fig. 4 A). DIC recordings of ani-1(RNAi) and ani-1(RNAi); par-4(it47) dividing one-cell embryos. Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
The loss of PAR-4 and ANI-1 leads to strong furrow mispositioning (related to Fig. 4 A). DIC recordings of ani-1(RNAi) and ani-1(RNAi); par-4(it47) dividing one-cell embryos. Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Loss of PAR-4 and ANI-1 leads to strong furrow mispositioning. (A and B) ani-1(RNAi); par-4(it47) embryos show strong furrow mispositioning. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow reached its most anterior position. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. See also Fig. S2 (A and D) and Video 3. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D and E) Furrow and spindle positions are strongly uncoupled in ani-1(RNAi); par-4(it47) embryos. Superposed confocal sections and DIC images (D) and quantification of furrow/spindle coupling (E) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Experiment was performed at 20°C. See also Fig. S2 (B and C) and Video 4. Bars, 10 µm. P-values from Student’s t test.
Loss of PAR-4 and ANI-1 leads to strong furrow mispositioning. (A and B) ani-1(RNAi); par-4(it47) embryos show strong furrow mispositioning. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow reached its most anterior position. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. See also Fig. S2 (A and D) and Video 3. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D and E) Furrow and spindle positions are strongly uncoupled in ani-1(RNAi); par-4(it47) embryos. Superposed confocal sections and DIC images (D) and quantification of furrow/spindle coupling (E) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Experiment was performed at 20°C. See also Fig. S2 (B and C) and Video 4. Bars, 10 µm. P-values from Student’s t test.
In α-tub::YFP; ani-1(RNAi); par-4(it47) embryos the spindle center is located toward the posterior pole, similar to α-tub::YFP, α-tub::YFP; ani-1(RNAi), and α-tub::YFP; par-4(it47) embryos (Fig. 4 C). Furthermore, the anterior aster-furrow distance is dramatically decreased in α-tub::YFP; ani-1(RNAi); par-4(it47) compared with control and single mutant embryos (Fig. 4, D and E; and Video 4). An even stronger effect is observed when worms are grown at fully restrictive temperature for par-4(it47) (25°C; Fig. S2 B). The cytokinetic furrow is therefore strongly shifted toward the anterior and away from the spindle center in ani-1(RNAi); par-4(it47) embryos. In the most severe cases, the furrow was displaced to such an extent that it could not properly segregate DNA (Fig. S2 C). Our results led us to propose that PAR-4 controls furrow position by regulating ANI-2 and ANI-1 together with CUL-5 but also by acting in an anillin-independent pathway (see Fig. 8 E). Strong furrow mispositioning is observed when these two pathways are simultaneously inhibited. Consistent with our hypothesis that ANI-2 acts upstream of ANI-1, we find that ANI-2 depletion does not suppress defects of ani-1(RNAi); par-4(it47) embryos (Fig. S2 D).
The loss of PAR-4 and ANI-1 leads to strong furrow mispositioning (related to Fig. 4 D). Superposed DIC and confocal recordings of control, ani-1(RNAi), par-4(it47), and ani-1(RNAi); par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
The loss of PAR-4 and ANI-1 leads to strong furrow mispositioning (related to Fig. 4 D). Superposed DIC and confocal recordings of control, ani-1(RNAi), par-4(it47), and ani-1(RNAi); par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
PIG-1 and ANI-1 both regulate furrow position
We next searched for PAR-4 targets that could be involved in the anillin-independent pathway. PAR-4 phosphorylates and activates AMPK-related kinases (Lizcano et al., 2004). Two of these kinases have been implicated in asymmetric cell division: PAR-1, which regulates several aspects of asymmetric division in early embryos (Kemphues et al., 1988; Tenlen et al., 2008; Benkemoun et al., 2014), and PIG-1, the homologue of vertebrate MELK, a kinase regulating cytokinesis in Xenopus laevis embryos (Le Page et al., 2011). In C. elegans, both PIG-1 and PAR-4 regulate neuroblast asymmetric division (Cordes et al., 2006; Ou et al., 2010; Chien et al., 2013) and pig-1 mutants enhance the polarity defects of weak par-2 mutants in two-cell embryos (Morton et al., 2012). PIG-1 is expressed in embryos (Fig. 5 A) where the endogenous protein is localized in the cytoplasm and at the cortex between adjacent cells (Fig. 5, B–G). Time-lapse imaging of embryos expressing a GFP::PIG-1 transgene shows that cortical recruitment occurs during mitosis (Video 5).
GFP::PIG-1 is recruited to the cortex during mitosis (related to Fig. 5). Confocal and DIC recording of a dividing four-cell embryo expressing GFP::PIG-1 (green). Video starts after nuclear envelop has broken down in the two cells marked with asterisks. GFP::PIG-1 is localized in the cytoplasm and recruited to the cortex (arrow) and the centrosomes (arrowheads) during mitosis. Images were recorded at 20-s intervals and movie is played at 3 frames per second. Embryo is oriented with anterior to the left.
GFP::PIG-1 is recruited to the cortex during mitosis (related to Fig. 5). Confocal and DIC recording of a dividing four-cell embryo expressing GFP::PIG-1 (green). Video starts after nuclear envelop has broken down in the two cells marked with asterisks. GFP::PIG-1 is localized in the cytoplasm and recruited to the cortex (arrow) and the centrosomes (arrowheads) during mitosis. Images were recorded at 20-s intervals and movie is played at 3 frames per second. Embryo is oriented with anterior to the left.
Loss of PIG-1 and ANI-1 leads to strong furrow mispositioning. (A–G) PIG-1 is expressed in embryos. Western blot (A) shows that PIG-1 is expressed in wild-type but not pig-1(gm344) embryonic extracts. Tubulin is used as a loading control. Immunostainings (B–G) show that PIG-1 is expressed in early wild-type (B–E) but not pig-1(gm344) embryos (F and G). PIG-1 is localized in the cytoplasm in one-cell (B) and early two-cell (C) embryos, it is also localized at the cortex between adjacent cells in late two-cell (D) and four-cell embryos (E). See also Video 5. (H–J) ani-1(RNAi); pig-1(gm344) embryos show strong furrow mispositioning. DIC images (H) and quantification of furrow position (I) and furrow/spindle coupling (J) in dividing one-cell embryos of the indicated genotypes. In J, all strains also express an α-tub::YFP transgene. Furrow position was measured when the furrow reached its most anterior position. (H) Orange dashed lines correspond to the embryo center, arrowheads to furrow position, and white asterisks to spindle asters. See also Fig. S3. (K) Strong furrow mispositioning can lead to DNA segregation defects in ani-1(RNAi); pig-1(gm344) embryos. DIC pictures of an ani-1(RNAi); pig-1(gm344) embryo in which all DNA is inherited by the posterior cell. This embryo also expressed an mCherry::HIS-58 transgene to monitor DNA position (red asterisks). Arrowheads indicate furrow position. 12 out of 28 embryos showed this DNA segregation phenotype. Bars, 10 µm. P-values from Student’s t test.
Loss of PIG-1 and ANI-1 leads to strong furrow mispositioning. (A–G) PIG-1 is expressed in embryos. Western blot (A) shows that PIG-1 is expressed in wild-type but not pig-1(gm344) embryonic extracts. Tubulin is used as a loading control. Immunostainings (B–G) show that PIG-1 is expressed in early wild-type (B–E) but not pig-1(gm344) embryos (F and G). PIG-1 is localized in the cytoplasm in one-cell (B) and early two-cell (C) embryos, it is also localized at the cortex between adjacent cells in late two-cell (D) and four-cell embryos (E). See also Video 5. (H–J) ani-1(RNAi); pig-1(gm344) embryos show strong furrow mispositioning. DIC images (H) and quantification of furrow position (I) and furrow/spindle coupling (J) in dividing one-cell embryos of the indicated genotypes. In J, all strains also express an α-tub::YFP transgene. Furrow position was measured when the furrow reached its most anterior position. (H) Orange dashed lines correspond to the embryo center, arrowheads to furrow position, and white asterisks to spindle asters. See also Fig. S3. (K) Strong furrow mispositioning can lead to DNA segregation defects in ani-1(RNAi); pig-1(gm344) embryos. DIC pictures of an ani-1(RNAi); pig-1(gm344) embryo in which all DNA is inherited by the posterior cell. This embryo also expressed an mCherry::HIS-58 transgene to monitor DNA position (red asterisks). Arrowheads indicate furrow position. 12 out of 28 embryos showed this DNA segregation phenotype. Bars, 10 µm. P-values from Student’s t test.
To determine whether PAR-1 or PIG-1 were involved in the ANI-1–independent pathway, we analyzed furrow position in embryos lacking ANI-1 and either PAR-1 or PIG-1. In ani-1(RNAi); par-1(zu310) embryos, the furrow is not shifted toward the anterior (Fig. S3 A). In contrast, although pig-1(gm344) embryos divide as wild type, ani-1(RNAi); pig-1(gm344) embryos behave similarly to ani-1(RNAi); par-4(it47) embryos: their furrows are located in the anterior half of the embryo (Fig. 5, H and I) and strongly shifted away from the spindle center (Fig. 5 J). This furrow mispositioning can lead to defects in DNA segregation (Fig. 5 K). Thus PIG-1 and ANI-1 both regulate furrow position and PIG-1 is likely to be a PAR-4 target in the anillin-independent pathway (see Fig. 8 E). Consistent with this model, pig-1(gm344); par-4(it47) embryos show only mild furrow position defects, similar to par-4(it47) embryos (Fig. S 3B). Finally, pig-1(gm344); cul-5(ok1706) embryos do not have furrow position defects (Fig. S3 C) and ANI-2 localization is not affected in pig-1(gm344) embryos (Fig. S3 D), suggesting that PIG-1 is not involved in regulating the anillin-dependent pathway.
PAR-4 and ANI-1 are required to maintain furrow position throughout cytokinesis
To determine the dynamics of the strong furrow mispositioning observed in ani-1(RNAi); par-4(it47) embryos, we followed the position of the furrow during ingression by visualizing it with an NMY-2::GFP transgene. In both control and ani-1(RNAi) embryos, furrow ingression initiates between 50% and 60% embryo length, in the vicinity of the spindle midzone (Fig. 6, A and C). The furrow then ingresses without major changes in its position (Fig. 6, A′, A″, and C; and Video 6). In par-4(it47) embryos, the furrow initiates between 50% and 60% embryo length and is then weakly displaced toward the anterior (Fig. 6 C). Most ani-1(RNAi); par-4(it47) embryos (18/21) initiate furrowing in the vicinity of the spindle midzone (Fig. 6, B and C). This is followed by a strong shift toward the anterior (Fig. 6, B′ and C; and Video 6). In 3/21 embryos, we observed that furrow ingression does not occur perpendicularly to the anteroposterior axis, one furrow tip starting ingression very close to the posterior pole and another very close to the anterior pole. Both furrow tips then move simultaneously toward the center of the embryo (Fig. S4, A–A″ and B; and Video 6). Thus, the initial position of furrow ingression is essentially correct in ani-1(RNAi); par-4(it47) embryos but PAR-4 and ANI-1 are then essential to maintain furrow position during ingression.
Dynamics of furrow ingression in ani-1; par-4 embryos (related to Figs. 6 and S4). Confocal recordings of control and ani-1(RNAi); par-4(it47) dividing one-cell embryos. The movie of the control embryo corresponds to the pictures shown in Fig. 6 A. The first (left) ani-1; par-4 embryo corresponds to the pictures shown in Fig. 6 B and the second ani-1; par-4 embryo corresponds to the pictures in Fig. S4 A. Embryos also express NMY-2::GFP (green) and mCherry::His-58 (red). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Dynamics of furrow ingression in ani-1; par-4 embryos (related to Figs. 6 and S4). Confocal recordings of control and ani-1(RNAi); par-4(it47) dividing one-cell embryos. The movie of the control embryo corresponds to the pictures shown in Fig. 6 A. The first (left) ani-1; par-4 embryo corresponds to the pictures shown in Fig. 6 B and the second ani-1; par-4 embryo corresponds to the pictures in Fig. S4 A. Embryos also express NMY-2::GFP (green) and mCherry::His-58 (red). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Dynamics of furrow ingression in ani-1; par-4 embryos. (A and B) During ingression, furrow position does not change in control embryos whereas it is strongly displaced in ani-1(RNAi); par-4(it47) embryos. Furrow position during ingression is monitored with an NMY-2::GFP transgene (green). DNA is monitored with an mCherry::HIS-58 transgene (red and asterisks). Images were acquired at the onset of cytokinesis (A and B), during ingression (A′ and B′), and just after furrow closure (A″ and B″). Arrowheads and white dashed lines indicate the position of initial ingression. Bars, 10 µm. See also Fig. S4 (A and B) and Video 6. (C) Graphs representing the position of the furrow tip during ingression of embryos of the indicated genotypes (all strains also express an NMY-2::GFP and mCherry::HIS-58 transgene). The furrow tip on the embryo side where ingression first initiated was tracked from ingression initiation until furrow closure. Its position is expressed as a percentage of embryo length (0% corresponds to the anterior pole and 100% to the posterior pole). Each colored track corresponds to the furrow tip position of a single embryo. 18/21 ani-1(RNAi); par-4(it47) embryos displayed such a furrow displacement; others are represented in Fig.S4 (A and B).
Dynamics of furrow ingression in ani-1; par-4 embryos. (A and B) During ingression, furrow position does not change in control embryos whereas it is strongly displaced in ani-1(RNAi); par-4(it47) embryos. Furrow position during ingression is monitored with an NMY-2::GFP transgene (green). DNA is monitored with an mCherry::HIS-58 transgene (red and asterisks). Images were acquired at the onset of cytokinesis (A and B), during ingression (A′ and B′), and just after furrow closure (A″ and B″). Arrowheads and white dashed lines indicate the position of initial ingression. Bars, 10 µm. See also Fig. S4 (A and B) and Video 6. (C) Graphs representing the position of the furrow tip during ingression of embryos of the indicated genotypes (all strains also express an NMY-2::GFP and mCherry::HIS-58 transgene). The furrow tip on the embryo side where ingression first initiated was tracked from ingression initiation until furrow closure. Its position is expressed as a percentage of embryo length (0% corresponds to the anterior pole and 100% to the posterior pole). Each colored track corresponds to the furrow tip position of a single embryo. 18/21 ani-1(RNAi); par-4(it47) embryos displayed such a furrow displacement; others are represented in Fig.S4 (A and B).
Notably, in 13/21 ani-1(RNAi); par-4(it47) embryos, the furrow moves so far toward the anterior that both sets of chromosomes are initially located on its posterior side (Fig. 6 B′ and Video 6). However, at the end of cytokinesis the furrow is slightly displaced toward the posterior whereas the DNA moves toward the anterior. As a result, the anterior set of chromosomes goes through the furrow before its final closure, allowing proper DNA segregation (Fig. 6, B″ and C; and Video 6).
PAR-4 and ANI-1 regulate furrow position in other asymmetrically dividing cells
Strikingly, furrow mispositioning in ani-1(RNAi); par-4(it47) embryos always occurs toward the anterior side of the embryo, suggesting that this may be linked to the inherent asymmetry of the one-cell embryo. We tested whether defects in furrow positioning are specific to asymmetrically dividing cells by observing two-cell stage embryos in which the anterior AB cell normally divides symmetrically and the posterior P1 cell divides asymmetrically (Fig. S5 A). In ani-1(RNAi); par-4(it47) two-cell embryos, the furrow is shifted toward the anterior in the posterior cell (Fig. S5, A and C) whereas the symmetrically dividing anterior cell is insensitive to the absence of PAR-4 and ANI-1 (Fig. S5, A and B). This suggests that PAR-4 and ANI-1 may regulate furrow position specifically in asymmetrically dividing cells.
Myosin abnormally accumulates at the anterior cortex of ani-1; par-4 and ani-1; pig-1 embryos
Our previous results suggest that furrow displacement is dictated by some intrinsic polarity of those cells. Considering its ability to induce cytokinesis in neuroblasts (Cabernard et al., 2010; Ou et al., 2010), myosin is a good candidate for causing the furrow to be displaced toward the anterior. We first analyzed myosin II dynamics during division using an NMY-2::GFP transgene. As previously described (Werner et al., 2007), in wild-type one-cell embryos, NMY-2::GFP localizes to the anterior cortex at metaphase, and then it briefly dissociates from the cortex and reaccumulates at the anterior cortex and in the equatorial region shortly after anaphase onset (Video 7). Myosin intensity at the anterior cortex increases after anaphase and then rapidly decreases before cytokinesis onset (Fig. 7 A and Video 7). In two-cell embryos, NMY-2::GFP also accumulates at the anterior cortex of the P1 cell before division whereas it is initially symmetrically localized in the AB cell and then removed from both poles of the cell before division (Video 8). In ani-1(RNAi); par-4(it47) embryos, myosin follows essentially the same pattern as in control embryos but myosin accumulation at the anterior cortex after anaphase is considerably stronger and lasts longer (Fig. 7 A and Video 7). No strong defects were observed in ani-1(RNAi) embryos. Consistent with previous observations (Chartier et al., 2011), myosin accumulation at the anterior cortex also lasts longer in par-4(it47) embryos, albeit to a lower levels than in ani-1(RNAi); par-4(it47) embryos (Fig. 7 A). Altogether, our results show that both PAR-4 and ANI-1 regulate myosin to prevent its excessive accumulation at the anterior cortex during cytokinesis (see Fig. 8, E and F). In pig-1(gm344) and ani-1(RNAi); pig-1(gm344) embryos, changes in myosin accumulation are very similar to those observed in par-4(it47) and ani-1(RNAi); par-4(it47) embryos, respectively (Fig. 7 B). This accumulation is not caused by a global change in myosin heavy or light chain levels (Fig. 7 C), suggesting that PIG-1 and ANI-1 prevent the cortical accumulation of myosin.
Dynamics of cortical myosin accumulation in control and ani-1; par-4 one-cell embryos (related to Fig. 7 A). Confocal recordings of control and ani-1(RNAi); par-4(it47) dividing one-cell embryos. Embryos also express NMY-2::GFP (green) and mCherry::His-58 (red). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Dynamics of cortical myosin accumulation in control and ani-1; par-4 one-cell embryos (related to Fig. 7 A). Confocal recordings of control and ani-1(RNAi); par-4(it47) dividing one-cell embryos. Embryos also express NMY-2::GFP (green) and mCherry::His-58 (red). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Dynamics of cortical myosin accumulation in control two-cell embryos. Confocal recording of a dividing two-cell embryo expressing NMY-2::GFP (green). Images were recorded at 5-s intervals and movie is played at 5 frames per second. Embryo is oriented with anterior to the left.
Dynamics of cortical myosin accumulation in control two-cell embryos. Confocal recording of a dividing two-cell embryo expressing NMY-2::GFP (green). Images were recorded at 5-s intervals and movie is played at 5 frames per second. Embryo is oriented with anterior to the left.
Myosin is up-regulated in ani-1; par-4 and ani-1; pig-1 embryos. (A and B) Myosin accumulation at the anterior cortex after anaphase onset is stronger and lasts longer in ani-1(RNAi); par-4(it47) (A) and ani-1(RNAi); pig-1(gm344) (B) embryos. Intensity of NMY-2::GFP fluorescence was measured during anaphase and cytokinesis at the anterior cortex of embryos of the indicated genotypes (all strains also express an NMY-2::GFP and mCherry::His-58 transgene) and is expressed relative to the intensity measured at anaphase onset. Arrows indicate the mean time of ingression initiation in the different strains. For examples of embryos used for quantifications, see Video 7. Error bars are SEM. (C) Western blots show that total myosin heavy and light chain levels are not affected in ani-1(RNAi); pig-1(gm344) embryos. Tubulin is used as a loading control. (D) At anaphase, P-MLC is not detected at the cortex of most (31/35) control embryos. It accumulates at the equatorial cortex in ani-1(RNAi) (24/32) and pig-1(gm344) (11/19) embryos and spreads along a broader cortical region in ani-1(RNAi); pig-1(gm344) (32/37) embryos. Asterisks indicate DNA. Bars, 10 µm.
Myosin is up-regulated in ani-1; par-4 and ani-1; pig-1 embryos. (A and B) Myosin accumulation at the anterior cortex after anaphase onset is stronger and lasts longer in ani-1(RNAi); par-4(it47) (A) and ani-1(RNAi); pig-1(gm344) (B) embryos. Intensity of NMY-2::GFP fluorescence was measured during anaphase and cytokinesis at the anterior cortex of embryos of the indicated genotypes (all strains also express an NMY-2::GFP and mCherry::His-58 transgene) and is expressed relative to the intensity measured at anaphase onset. Arrows indicate the mean time of ingression initiation in the different strains. For examples of embryos used for quantifications, see Video 7. Error bars are SEM. (C) Western blots show that total myosin heavy and light chain levels are not affected in ani-1(RNAi); pig-1(gm344) embryos. Tubulin is used as a loading control. (D) At anaphase, P-MLC is not detected at the cortex of most (31/35) control embryos. It accumulates at the equatorial cortex in ani-1(RNAi) (24/32) and pig-1(gm344) (11/19) embryos and spreads along a broader cortical region in ani-1(RNAi); pig-1(gm344) (32/37) embryos. Asterisks indicate DNA. Bars, 10 µm.
We assessed whether the accumulation of cortical myosin during anaphase in ani-1(RNAi); pig-1(gm344) embryos corresponds to activated myosin by analyzing the localization of phosphorylated myosin light chain (P-MLC). During anaphase, P-MLC is not detected at the cortex of control embryos whereas it localizes at the cortex surrounding the spindle midzone in ani-1(RNAi) and pig-1(gm344) embryos (Fig. 7 D). In ani-1(RNAi); pig-1(gm344) embryos, P-MLC strikingly accumulates in a broad region of the cortex: it forms a cortical band that spreads toward the anterior cortex (Fig. 7 D). Thus, together, ANI-1 and PIG-1 inhibit both the accumulation and the spreading of activated myosin along the cortex.
Myosin up-regulation leads to furrow mispositioning
We noticed that both ani-1(RNAi); par-4(it47) and ani-1(RNAi); pig-1(gm344) embryos, but not par-4(it47), pig-1(gm344), or ani-1(RNAi) embryos, show unusual cortical deformations during mitosis, in particular at 25°C (Video 9, Fig. S4 C, and not depicted). To test whether myosin activity contributes to these deformations and to the anterior displacement of the furrow observed in ani-1(RNAi); par-4(it47) embryos, we inactivated myosin during mitosis by using a fast-inactivated myosin thermosensitive mutant (see Material and methods for details). We found that, contrary to α-tub::YFP; ani-1(RNAi); par-4(it47) embryos, α-tub::YFP nmy-2(ne1490); ani-1(RNAi); par-4(it47) embryos do not show cortical deformations (Video 9). Moreover, the furrow position defects of α-tub::YFP; ani-1(RNAi); par-4(it47) embryos are rescued in α-tub::YFP nmy-2(ne1490); ani-1(RNAi); par-4(it47) embryos (Fig. 8, A and C). Myosin thus appears to be responsible for cortical deformations and furrow displacement in ani-1(RNAi); par-4(it47) embryos. Notably, when shifted to 25°C, α-tub::YFP control embryos also show a transient shift of the furrow toward the anterior. This shift is rescued in α-tub::YFP nmy-2(ne1490) embryos (Fig. 8 C) and is therefore likely caused by myosin activity. Our results show that PAR-4, PIG-1, and ANI-1 are negative regulators of myosin and that excessive myosin activity leads to cortical deformations and furrow mispositioning.
Cortical deformations and furrow position in ani-1; par-4 and nmy-2; ani-1; par-4 one-cell embryos (related to Figs. 8 A and S4 C). DIC recordings of ani-1(RNAi); par-4(it47) and nmy-2(ne1490); ani-1(RNAi); par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP not depicted). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Cortical deformations and furrow position in ani-1; par-4 and nmy-2; ani-1; par-4 one-cell embryos (related to Figs. 8 A and S4 C). DIC recordings of ani-1(RNAi); par-4(it47) and nmy-2(ne1490); ani-1(RNAi); par-4(it47) dividing one-cell embryos (embryos also express α-tubulin::YFP not depicted). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Myosin up-regulation leads to furrow mispositioning. (A–D) Furrow and spindle positions are coupled when myosin is inhibited in ani-1(RNAi); par-4(it47) embryos (A and C) and uncoupled when myosin is hyperactivated upon rga-3/4(RNAi) (B and D). Superposed confocal sections and DIC images (A and B) and quantification of furrow/spindle coupling (C and D) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Bars, 10 µm. P-values from Student’s t test. See also Videos 9 and 10. (E) A novel pathway regulating furrow positioning: PAR-4 and CUL-5 redundantly prevent ANI-2 accumulation at the cortex; ANI-2 in turns inhibits ANI-1. PAR-4 also regulates an anillin-independent pathway that involves the PIG-1/MELK kinase. Together, these two pathways prevent the cortical accumulation of activated myosin, thereby ensuring that myosin does not shift the furrow toward the anterior. (F) A model to explain furrow mispositioning in ani-1; par-4 and ani-1; pig-1 embryos. In wild-type as well as ani-1; par-4 or ani-1; pig-1 embryos, centralspindlin and astral microtubules induce furrowing in the vicinity of the spindle midzone. In wild-type embryos, PAR-4, PIG-1, and ANI-1 keep myosin accumulation and activity at the anterior cortex at a low level during cytokinesis, thereby preventing furrow displacement. In the absence of ANI-1 and PAR-4 or PIG-1, myosin accumulation at the anterior cortex increases during cytokinesis and excess of myosin activity drives the furrow toward the anterior pole of the embryo.
Myosin up-regulation leads to furrow mispositioning. (A–D) Furrow and spindle positions are coupled when myosin is inhibited in ani-1(RNAi); par-4(it47) embryos (A and C) and uncoupled when myosin is hyperactivated upon rga-3/4(RNAi) (B and D). Superposed confocal sections and DIC images (A and B) and quantification of furrow/spindle coupling (C and D) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Bars, 10 µm. P-values from Student’s t test. See also Videos 9 and 10. (E) A novel pathway regulating furrow positioning: PAR-4 and CUL-5 redundantly prevent ANI-2 accumulation at the cortex; ANI-2 in turns inhibits ANI-1. PAR-4 also regulates an anillin-independent pathway that involves the PIG-1/MELK kinase. Together, these two pathways prevent the cortical accumulation of activated myosin, thereby ensuring that myosin does not shift the furrow toward the anterior. (F) A model to explain furrow mispositioning in ani-1; par-4 and ani-1; pig-1 embryos. In wild-type as well as ani-1; par-4 or ani-1; pig-1 embryos, centralspindlin and astral microtubules induce furrowing in the vicinity of the spindle midzone. In wild-type embryos, PAR-4, PIG-1, and ANI-1 keep myosin accumulation and activity at the anterior cortex at a low level during cytokinesis, thereby preventing furrow displacement. In the absence of ANI-1 and PAR-4 or PIG-1, myosin accumulation at the anterior cortex increases during cytokinesis and excess of myosin activity drives the furrow toward the anterior pole of the embryo.
To independently confirm that an excess of myosin activity can lead to furrow mispositioning, we depleted RGA-3/4, two Rho GAPs that inhibit myosin (Schmutz et al., 2007). rga-3/4(RNAi) leads to anterior cortex hypercontractility during mitosis as well as strong furrow mispositioning (Fig. 8, B and D; and Video 10). Thus, excessive myosin activity, regardless of its origin, can result in furrow displacement. Altogether our results show that the regulation of myosin is essential to prevent it from shifting the cytokinesis furrow away from the spindle midzone.
Hyperactivation of myosin in rga-3/4(RNAi) embryos leads to strong furrow mispositioning (related to Fig. 8 B). Superposed DIC and confocal recordings of control and rga-3/4(RNAi) dividing one-cell embryos of the indicated genotypes (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Hyperactivation of myosin in rga-3/4(RNAi) embryos leads to strong furrow mispositioning (related to Fig. 8 B). Superposed DIC and confocal recordings of control and rga-3/4(RNAi) dividing one-cell embryos of the indicated genotypes (embryos also express α-tubulin::YFP green). Images were recorded at 5-s intervals and movies are played at 5 frames per second. Embryos are oriented with anterior to the left.
Discussion
Here we identify a novel pathway required to coordinate furrow and spindle positions during asymmetric division. This involves the regulation of anillins as well as an anillin-independent pathway (Fig. 8 E). This regulatory network prevents excessive myosin accumulation and activity at the anterior cortex. This regulation is crucial to prevent myosin from shifting the cytokinetic furrow away from the spindle midzone and is therefore essential to ensure proper DNA segregation. We propose that anterior myosin and the mitotic spindle have opposite effects on furrow position and that the final position of the furrow is the result of an equilibrium between these two opposing signals.
The major finding of our work is that the cytokinetic furrow is shifted toward the anterior in cul-5 par-4 and ani-1; par-4 embryos, yet their mitotic spindles are correctly localized toward the posterior pole. PAR-4, CUL-5, and ANI-1 are thus required to coordinate the position of the cytokinetic furrow with the spindle midzone. Our data show that furrowing is first induced in the vicinity of the spindle midzone, independently of the presence of PAR-4 and ANI-1. Both PAR-4 and ANI-1 are then required to maintain furrow position during ingression (Fig. 8 F). In the absence of PAR-4 and ANI-1, myosin levels are increased at the anterior cortex during cytokinesis. This leads to the displacement of the cytokinetic furrow toward the anterior (Fig. 8 F). Importantly, furrow mispositioning is also observed when anterior myosin activity is up-regulated by completely independent means, for instance by depleting the Rho GAPs RGA-3/4. Thus increased myosin activity at the anterior cortex is sufficient to pull the furrow toward the anterior. Our results show that, like in Drosophila or C. elegans neuroblasts (Cabernard et al., 2010; Ou et al., 2010), asymmetrically localized myosin can control cytokinesis furrow position in the one-cell C. elegans embryo. In Drosophila neuroblasts myosin and the spindle are both localized on the same side of the cell and both contribute to the precise localization of the cytokinetic furrow (Roth et al., 2015). However, in the one-cell C. elegans embryo, anterior myosin and the posterior spindle have opposing influences on furrow position. Tight regulation of myosin is therefore crucial to maintain the equilibrium between these two effects, ensuring that the cytokinetic furrow ingresses through the spindle midzone.
Two pathways appeared to be crucial in regulating myosin: an anillin-independent pathway that regulates the cortical accumulation of myosin through the PIG-1/MELK kinase as well as an anillin-dependent pathway involving ANI-1 and ANI-2 (Fig. 8 E). PAR-4 and CUL-5 prevent the accumulation of ANI-2 at the cortex between the AB and P1 cells and regulate furrow position in an ANI-2–dependent manner. Our data suggest that CUL-5 regulates furrow position through its function in a ubiquitin ligase complex that contains the RING protein RBX-2. CUL-5 could inhibit the cortical accumulation of ANI-2 by directly promoting its degradation. However, changes in protein levels cannot be detected by Western blot analysis. This could be explained were CUL-5 only to degrade a small pool of ANI-2 locally. Alternatively, CUL-5 may degrade another factor that in turn regulates the cortical recruitment of ANI-2. Moreover, the stronger cortical accumulation of ANI-2 observed in the absence of both PAR-4 and CUL-5 indicates that ANI-2 accumulation is also regulated by PAR-4 in a CUL-5–independent manner.
Previous work (Amini et al., 2014) and our data suggest that ANI-2 inhibits ANI-1. However, furrow mispositioning defects are much more pronounced in ani-1; par-4 embryos than in cul-5 par-4 embryos. Inhibition of ANI-1 through the accumulation of ANI-2 in cul-5 par-4 embryos is thus likely to be only partial. Our data show that ANI-1, together with PIG-1, inhibits both the accumulation of activated myosin and its spreading toward the anterior cortex during anaphase. One possibility is that ANI-1 and PIG-1 prevent the cortical accumulation of activated myosin by regulating its recruitment to the cortex. Alternatively they could indirectly inhibit the phosphorylation of myosin light chain. In tissue culture cells, anillin depletion leads to oscillations of the cytokinetic furrow and it has been proposed that anillin maintains the furrow in the vicinity of the spindle midzone by acting as a scaffold protein linking RhoA and MgcRacGap with actin and myosin (Piekny and Glotzer, 2008). Such a mechanism cannot account for the effect of ANI-1 on the accumulation of activated myosin. However, it could explain how ANI-1 restricts myosin localization close to the spindle midzone. ANI-1 localizes at the cortex during anaphase (Maddox et al., 2005; unpublished data) but we did not detect PIG-1 at the cortex in one-cell embryos. PIG-1 may thus regulate myosin in the cytoplasm before it is recruited to the cortex; alternatively, it may transiently interact with cortical proteins. Consistent with the latter hypothesis, PIG-1 localizes at the cortex between adjacent cells, suggesting that cell–cell contacts may stabilize its interaction with cortical proteins. Furthermore, GFP::PIG-1 appears to be recruited at the cortex at the end of mitosis, reminiscent of what happens in Xenopus embryos and human cells where MELK is recruited to the cortex at anaphase (Chartrain et al., 2006; Le Page et al., 2011).
PIG-1 is known to be required for the asymmetric division of both QR.p and QR.a neuroblasts (Ou et al., 2010). QR.p neuroblasts divide similarly to the one-cell embryo with their spindle being displaced toward the posterior and inducing furrowing, suggesting that PIG-1 may also coordinate spindle and furrow positions in those cells through myosin regulation. In QR.a neuroblasts, furrowing is induced by myosin accumulation at the anterior cortex and PIG-1 is required to exclude myosin from the posterior cortex (Ou et al., 2010). PIG-1 may thus regulate myosin localization in both types of asymmetric division, albeit with a different spatial pattern.
Finally, it should be noted that during the polarization phase of the embryo, both PAR-4 and ANI-1 positively regulate actomyosin contractility (Morton et al., 1992; Maddox et al., 2005; Chartier et al., 2011): contrary to what happens during anaphase, par-4 and ani-1 embryos show reduced pseudocleavage ingression and cortical contractility during polarity establishment. Similarly, we also observed that pseudocleavage formation is reduced in pig-1 embryos (unpublished data). Thus, the effect of PAR-4, PIG-1, and ANI-1 on myosin depends on the stage of asymmetric cell division.
In conclusion we have identified a novel regulatory pathway that is essential to restrict the accumulation and activity of asymmetrically localized myosin and to ensure that spindle and furrow positions are coordinated. Absence of this regulation leads to a dramatic mispositioning of the cytokinetic furrow, which can impair DNA segregation. However, it is noteworthy that in some embryos late changes in furrow and DNA positions correct the initial DNA segregation defects. This suggests the existence of a supplementary control mechanism that helps to ensure proper DNA segregation. Deciphering the mechanisms that allow the correction of DNA segregation defects in the later stages of cell division will be essential to further understand how successive control mechanisms ensure robust DNA segregation during cell division.
Materials and methods
Worm strains and RNAi
Because many of our strains carry thermosensitive mutations, they were maintained at 15°C using standard procedures. A list of strains used is provided in Table S1. A strain expressing GFP::PIG-1 under the control of the pie-1 promoter was obtained by biolistic bombardment of DP38 worms with pID3.01B-pig-1 (pig-1 cDNA cloned into pID3.01B; D’Agostino et al., 2006) under the control of the pie-1 promoter and fused at the N terminus with GFP).
RNAi was performed by feeding worms with RNAi clones from the Ahringer-Source BioScience library (Kamath et al., 2003) on plates containing 1 or 3 mM IPTG. L4440 was used as a control in all RNAi experiments. For rga-3/4 RNAi, the RNAi clone used is primarily directed against K09H11.3 (rga-3) but also targets Y75B7AL.4 (rga-4) (Schmutz et al., 2007). Double ani-2/ani-1 RNAi was performed by growing ani-2 and ani-1 feeding clones separately in liquid cultures. Liquid cultures were then mixed before seeding 3 mM IPTG plates. In this experiment, ani-2 and ani-1 “single” RNAi was performed by diluting ani-2 or ani-1 clones together with the L4440 control RNAi. Conditions used for each experiment are detailed in Table S2.
Lethality tests (related to Fig. S1)
par-4(RNAi) in cul-5(ok1706) was performed by feeding worms on plates containing 3 mM IPTG for 24 h at 25°C before isolating a few adults on counting plates. Adults were left to lay eggs 16–18 h before being removed from plates. par-4(RNAi) in rbx-2(ok1617) mutants was performed by feeding worms on plates containing 1 mM IPTG for 24 h at 25°C before isolating adults on counting plates. Adults were left to lay eggs 6–8 h before being removed from plates. In both cases, embryonic lethality was assessed 24 h after removing adult worms from plates, dividing the number of unhatched embryos by the total number of progeny. cul-5(ok1706) par-4(it47) and rbx-2(ok1617); par-4(it47) adults were left to lay eggs for 24 h at 15°C on OP50 plates before being removed from plates. Embryonic lethality was assessed 32–48 h after removing adult worms from plates. In each independent experiment, two to four plates were counted for each genotype. On all graphs, lethality is the mean of at least five independent experiments (more than 200 progeny were counted for each experiment).
Immunostainings
Embryos were fixed in methanol and stained according to standard procedures using rabbit anti–SPD-1 (1/50; Verbrugghe and White, 2004; provided by R. Chan, University of Michigan, Ann Arbor, MI), mouse anti-tubulin (1/1,000; DM1A; Sigma-Aldrich), rabbit anti–ANI-2 (1/1,000; residues 890–1015; Maddox et al., 2005; provided by A. Maddox, University of North Carolina, Chapel Hill, NC), rabbit anti-phospho (ser19) myosin light chain (1/30; Cell Signaling Technology), and rabbit anti–PIG-1 (1/100; this study) primary antibodies. A polyclonal anti–PIG-1 antibody was obtained after rabbit immunization with the synthetic peptide VKDNDKENASTGKNY. Immune sera were then affinity purified using the same peptide (Eurogentec). Secondary antibodies were conjugated with Alexa Fluor 488 or 568 (1/500; Invitrogen) and DNA was stained using Topro 3 (1/500; Life technologies). Observations were performed using a SPE or SP5 confocal microscope (Leica) equipped with a 63× objective (HCX Plan Apochromat, NA 1.4; Leica). Images were acquired with the LAS AF software and assembled for illustration using ImageJ and Photoshop (Adobe). Conditions used for each experiment are detailed in Table S2.
Time-lapse recordings
Embryos were mounted on 2% agarose pads in a drop of M9 medium and recordings were performed in a room maintained at 20°C or 25°C (see Table S2 for details). For experiments with α-tubulin::YFP nmy-2(ne1490); ani-1(RNAi); par-4(it47) embryos, worms were grown on RNAi plates at 20°C, dissected, and quickly shifted to 25°C for microscopy. Embryos that were undergoing mitosis during the temperature shift were recorded. Many embryos carrying the nmy-2(ne1490) mutation did not have cytokinetic furrow ingression but some still displayed at least partial furrow ingression. In a few cases, embryos had a centrally positioned spindle; those embryos were not included in the quantifications of aster/furrow distance.
DIC recordings were performed on an SPE confocal microscope equipped with a 63× objective (HCX Plan Apochromat, NA 1.4; Leica) and a charged couple device camera (DFC 360FX; Leica). Confocal and superposed confocal/DIC images were acquired on SPE or SP5 confocal microscopes with a 63× objective (HCX Plan Apochromat, NA 1.4; Leica). Images were acquired with the LAS AF software at 5-s intervals, except for Video 5 (20-s intervals), and assembled for illustration using ImageJ and Photoshop.
Image analysis and quantifications
AB size and anterior aster-furrow distance were measured using ImageJ software. The presence of the polar body was used to orient the embryos along the anteroposterior axis. Position of the furrow tip during ingression was tracked manually in ImageJ. When the two furrow tips on each side of the embryo had very close positions along the anteroposterior axis, only the first ingressing tip was tracked (Fig. 6, A–C).
Quantifications of ANI-2 (Figs. 3 B and S3 D) and myosin (Fig. 7, A and B) intensities were performed using ImageJ. For ANI-2, z-stacks were acquired (500-nm z-step) and the signal intensities of the brightest nine z-sections were summed; a 5-pixel-wide line was then drawn along the cortex separating AB and P1 and integrated density of the signal along this line was measured (cortical signal). This line was then moved to the adjacent cytoplasm and signal integrated density was measured (cytoplasmic signal). The accumulation of ANI-2 at the cortex was then expressed as the ratio between cortical and cytoplasmic signals. For myosin, a region corresponding to the most anterior 10–15% of the embryo cortex was defined and NMY-2::GFP cortical intensity was measured in a central confocal plane from anaphase onset to cytokinesis ring closure.
In all box plots, the box corresponds to the 25th to 75th percentile, the lower whisker to the first decile and the upper whisker to the ninth decile of the embryo population. The bar in the middle of the box corresponds to the median.
Worm extracts and Western blots
To analyze ANI-2 levels and PIG-1 expression, N2, pig-1(gm344), cul-5(ok1706), par-4(it47), and cul-5(ok1706) par-4(it47) worms were grown 24 h at 20°C before bleaching gravid adults. For the analysis of myosin levels, N2 and pig-1(gm3444) L1 were grown at 20°C until adulthood on L4440 or ani-1(RNAi) plates (1 mM IPTG) before being bleached. Embryos were then directly resuspended in SDS sample buffer. Western blots were performed according to standard procedures using rabbit anti–PIG-1 (1/5,000; this study), rabbit anti–ANI-2 (residues 890–1015 [Maddox et al., 2005]; 1/2,500), rabbit anti–NMY-2 (residues 945–1368 [Maddox et al., 2005]; 1/5,000; provided by A. Maddox), mouse anti-MLC (T14c, DSHB, 1/2,000), and mouse anti-tubulin (DM1A, 1/2,500; Sigma-Aldrich) antibodies.
Online supplemental material
Fig. S1 shows synthetic lethality between cul-5/rbx-2 and par-4 mutants, furrow positioning defects upon par-4 depletion in cul-5 and rbx-2 mutants, and ANI-2 levels in cul-5 par-4 embryonic extracts. Fig. S2 shows quantifications of furrow position in ani-1(RNAi); par-4 and ani-2(RNAi); ani-1(RNAi); par-4 embryos and DNA segregation defects in ani-1(RNAi); par-4 embryos. Fig. S3 shows quantifications of furrow position in ani-1(RNAi); par-1, pig-1; par-4 and pig-1; cul-5 embryos as well as ANI-2 localization in pig-1 embryos. Fig. S4 shows the dynamics of furrow ingression and cortical deformations in ani-1(RNAi); par-4 embryos. Fig. S5 shows furrow localization in ani-1(RNAi); par-4 two-cell embryos. Videos 1 and 2 show DIC and confocal recordings of dividing rbx-2; par-4 and cul-5 par-4 embryos (depicted in Fig. 1, A and E). Videos 3 and 4 show DIC and confocal recordings of dividing ani-1(RNAi); par-4 embryos (depicted in Fig. 4, A and D). Video 5 shows the dynamic localization of GFP::PIG-1. Video 6 shows the dynamics of furrow ingression in ani-1(RNAi); par-4 embryos (depicted in Figs. 6 and S4 A). Video 7 shows the dynamics of cortical myosin accumulation in control and ani-1(RNAi); par-4 one-cell embryos. Video 8 shows the dynamics of cortical myosin accumulation in control two-cell embryos. Video 9 shows cortical deformations in ani-1(RNAi); par-4 (depicted in Fig. S4 C) and nmy-2; ani-1(RNAi); par-4 embryos. Video 10 shows furrow ingression in rga-3/4(RNAi) embryos (depicted in Fig. 8 B). Additional data are available in the JCB DataViewer at http://dx.doi.org/10.1083/jcb.201503006.dv.
Acknowledgments
We would like to thank A. Maddox for providing ANI-2 and NMY-2 antibodies and R. Chan for anti-SPD-1 antibody. Some worm strains were provided by the Caenorhabidtis Genetics Center, which is funded by the National Institutes of Health Office of Research Infrastructure Programs (P40 OD010440). N. Loyer helped with furrow tip tracking and E. Daniel and R. Jugele contributed to the earlier steps of this project. We thank M. Gotta, in whose laboratory this project was initiated, and the Microscopy Rennes Imaging Center for assistance with microscopy. We would also like to thank R. Le Borgne, S. Le Bras, L. Chesneau, S. Dixon, A. Echard, G. Gillard, and J. Pécreaux for discussions and critical reading of the manuscript.
A. Pacquelet was supported by the Institut National de la Santé et de la Recherche Médicale and a Stratégie d’attractivité durable grant from the Région Bretagne and our group is supported by the Ligue contre le cancer (22/29/35/72), the Centre National de la Recherche Scientifique, and the Université Rennes 1.
The authors declare no competing financial interests.


![Figure 1. CUL-5/RBX-2 and PAR-4 regulate cytokinetic furrow position. (A and B) The furrow is shifted toward the anterior in cul-5(ok1706) par-4(it47) and rbx-2(ok1617); par-4(it47) embryos. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow spanned the entire embryo. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. As depicted in the inset (B), furrow position is the distance between the anterior pole and the furrow (AB size) expressed as a percentage of total embryo length (0% corresponds to the anterior pole, 100% to the posterior pole). See also Fig. S1 and Video 1. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D) Localization of the spindle midzone protein SPD-1 and of DNA during anaphase is normal in cul-5(ok1706) par-4(it47) embryos. Confocal section of wild-type and cul-5(ok1706) par-4(it47) one-cell anaphase embryos stained for α-tubulin (red), SPD-1 (green), and DNA (blue). More than 10 anaphase embryos per genotype were observed and representative images are shown. (E and F) Furrow and spindle positions are uncoupled in cul-5(ok1706) par-4(it47) embryos. Superposed confocal sections and DIC images (E) and quantification of furrow/spindle coupling (F) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Green dashed lines correspond to the spindle center and arrowheads to furrow position. As depicted in the inset (F), the distance between the anterior aster and the furrow (A-F) was measured during ingression and expressed as a percentage of spindle length (50% corresponds to the furrow position being coupled with spindle center, below 50% corresponds to a shift of the furrow toward the anterior, and above 50% corresponds to a shift toward the posterior). See also Video 2.In all images, embryos are oriented with the anterior to the left. Bars, 10 µm. P-values from Student’s t test.](https://cdn.rupress.org/rup/content_public/journal/jcb/210/7/10.1083_jcb.201503006/7/m_jcb_201503006_fig1.jpeg?Expires=1779196885&Signature=4pALaKM71WYPbfD~V9Y1PkFd7zftQhO6D8YV9GiYqmrlu~PcI6hHdVJc0h0DcBJCljtpr-CZZjZl26vJPNmbKn8aC6c79faJGisVAOZ1Gy69Z8FoSLJiCWogUZ2~nLoMs7BZfN0~IVpqxoMgFvpJHEH12zMU~RoLQTD0Zk~ytww~JJDHDAchCY0SUEg7vEiFGBQz7mNk8EaXzwEwkoQGEGhfhOzqFOZPjuV7KlAcu7Aw4AVwIyqgCeJPjToJsRYW5KqU8EHOpMg387Cb5Uh4jYCB3DqIvrw79LXJNW-kv9pAMxn~iPOy~0sn5tJcM6vb0wZnyk4-0zF80qyi2NIz-w__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)

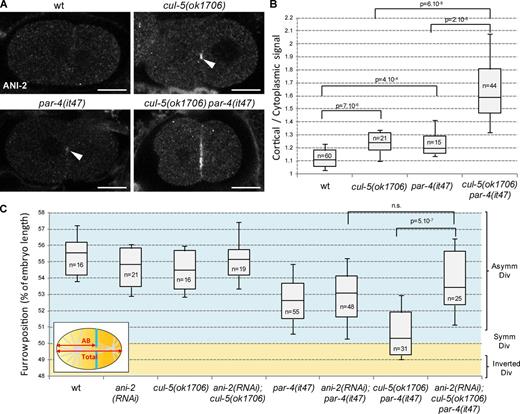
![Figure 4. Loss of PAR-4 and ANI-1 leads to strong furrow mispositioning. (A and B) ani-1(RNAi); par-4(it47) embryos show strong furrow mispositioning. DIC images (A) and quantification of furrow position (B) in dividing one-cell embryos of the indicated genotypes. Furrow position was measured when the furrow reached its most anterior position. Orange dashed lines correspond to the embryo center and arrowheads to furrow position. See also Fig. S2 (A and D) and Video 3. (C) Position of the spindle center at the onset of cytokinesis in one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene). (D and E) Furrow and spindle positions are strongly uncoupled in ani-1(RNAi); par-4(it47) embryos. Superposed confocal sections and DIC images (D) and quantification of furrow/spindle coupling (E) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Experiment was performed at 20°C. See also Fig. S2 (B and C) and Video 4. Bars, 10 µm. P-values from Student’s t test.](https://cdn.rupress.org/rup/content_public/journal/jcb/210/7/10.1083_jcb.201503006/7/m_jcb_201503006_fig4.jpeg?Expires=1779196885&Signature=001-qJIoN7KyuNXxD4mjhhPxd2iK4ZLyXdaswZhGoY9ogLf79WOz9ImIo3~8jcvnhoH9xoi5vO4ankKPO3dOtiPQEIZoORV~m0bAto2JkJfNZQB73vnXuR4FQfrlqu~gfo4ZRlaggswnMQXf36fWzgVT02NSA6lxDZYBD3VwamfgXwFQ6UJrenSUbd7gPLmLbB9efNs8bkHY31VPnbML6iZaf2PGESKaI~a80~YjPUkYMAuYeeVrbki4yrK7ywxBGT2tiZLhEqUn8quMUYuUhnjwY7BiN1NFtFoVij4Zm8pubiH9aP73wJXHI8hPoA~K08x4Ld2c9bKCvtJDdRiUfg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
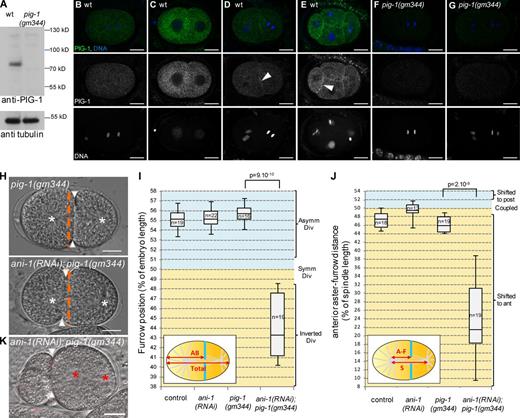
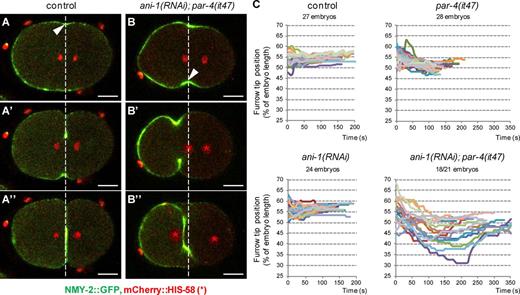

![Figure 8. Myosin up-regulation leads to furrow mispositioning. (A–D) Furrow and spindle positions are coupled when myosin is inhibited in ani-1(RNAi); par-4(it47) embryos (A and C) and uncoupled when myosin is hyperactivated upon rga-3/4(RNAi) (B and D). Superposed confocal sections and DIC images (A and B) and quantification of furrow/spindle coupling (C and D) in dividing one-cell embryos of the indicated genotypes (all strains also express an α-tub::YFP transgene [green]). Furrow position was measured when the furrow reached its most anterior position. Green dashed lines correspond to the spindle center and arrowheads to furrow position. Bars, 10 µm. P-values from Student’s t test. See also Videos 9 and 10. (E) A novel pathway regulating furrow positioning: PAR-4 and CUL-5 redundantly prevent ANI-2 accumulation at the cortex; ANI-2 in turns inhibits ANI-1. PAR-4 also regulates an anillin-independent pathway that involves the PIG-1/MELK kinase. Together, these two pathways prevent the cortical accumulation of activated myosin, thereby ensuring that myosin does not shift the furrow toward the anterior. (F) A model to explain furrow mispositioning in ani-1; par-4 and ani-1; pig-1 embryos. In wild-type as well as ani-1; par-4 or ani-1; pig-1 embryos, centralspindlin and astral microtubules induce furrowing in the vicinity of the spindle midzone. In wild-type embryos, PAR-4, PIG-1, and ANI-1 keep myosin accumulation and activity at the anterior cortex at a low level during cytokinesis, thereby preventing furrow displacement. In the absence of ANI-1 and PAR-4 or PIG-1, myosin accumulation at the anterior cortex increases during cytokinesis and excess of myosin activity drives the furrow toward the anterior pole of the embryo.](https://cdn.rupress.org/rup/content_public/journal/jcb/210/7/10.1083_jcb.201503006/7/m_jcb_201503006_fig8.jpeg?Expires=1779196885&Signature=DjO6~N6k8PjXgj26phYFTy35tkMOivfsLciKFl72F1wqaYNeAScY~8lTzO4wKU-V2u5I1E2SO-SDrNd8WRYyE3mqVKTSBogx~dSGj9xLWZMMRafp-ouRkN~Dp-wMEZTZhZaOS1kmVzs3yjnIUdZnoUoZqkbYuQ-xCz~jXxbgsfB9oYpja-1JKrNtAf6T-ZOuaEQfwRWGBAPpRsnEW5Da7TNNVcpEn15IVG-YREQMAnUWHOGGO9ybQCEXIuydkTSVhUWWgpV6QLKgVnWlPePjOsBq3huxgmuFln9dJHmD45VCQH0nW8QPprlWMaZRkBkgbC06WpCFdBf66Xut2T23eA__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)


