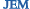PTEN deficiency increases collagen deposition and scarring (brown) in injured lungs.
Fibroblasts help repair damaged tissue by proliferating and producing collagen. But too much collagen can create scars and impair tissue function, as seen in patients with a chronic lung disease called idiopathic pulmonary fibrosis (IPF). Collagen normally controls its own levels by binding to integrins on the fibroblast surface, which triggers a negative feedback loop that stops the cell from proliferating further. In patients with IPF, according to Xia et al., this feedback loop falls apart.
The defective cog in this inhibitory loop was a tumor suppressor phosphatase called PTEN, which normally halts the cell cycle by inactivating PI3K/Akt signaling. In fibroblasts from IPF patients, however, collagen-induced stop signals failed to activate PTEN and inhibit proliferation. In response to a chemical irritant, mice with reduced levels of PTEN developed unusually severe lung injury with excess collagen deposition.
Unlike certain tumors, which have low PTEN protein levels, fibroblasts had normal PTEN levels, but the protein failed to relocate to the cell membrane, where it gets activated. The defect that leads to this faulty localization is not yet known.
Other studies have suggested that collagen-producing cells in the lungs of patients with IPF are not good fibroblasts gone bad, but rather epithelial cells that differentiate into fibroblast-like cells in response to chronic exposure to TGF-β—an inflammatory cytokine that is prevalent in the lungs of IPF patients.