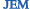Autoimmune colitis (top) caused by homeostatically proliferating CD8+ T cells is resolved in mice injected with anti–IL-6 antibody (bottom).
Infusion of naive T cells into an environment where there are few native T cells—such as in patients undergoing cancer therapy—spurs two kinds of proliferation. T cells activated by non-self-peptide/MHC complexes proliferate rapidly, whereas those activated by self-peptide/MHC in the presence of cytokines such as IL-7 and IL-15 proliferate slowly. CD4+ T cells that proliferate by these methods have previously been shown to drive the inflammation seen in several autoimmune diseases.
Tajima et al. now find a new variation in homeostatic proliferation of CD8+ T cells that also induces autoimmunity. In gut lymph nodes of T cell–deficient mice, injected CD8+ T cells proliferated rapidly in response to IL-6. This inflammatory cytokine was found at high levels in these lymph nodes, perhaps due to their proximity to the bacterium-laden gut.
The mice developed a thicker gut epithelium and lost weight—signs of an autoimmune disease called colitis. T cell–injected mice treated with an IL-6–blocking antibody or with bacterium-depleting drugs, however, remained healthy.
The proliferating T cells secreted several inflammatory cytokines, including IL-17. The IL-17 alone seemed to be responsible for the colitis, as mice injected with T cells that were unable to produce IL-17 remained healthy. If these IL-17–producing CD8+ T cells are found in patients suffering from diseases such as colitis, the anti–IL-6 antibody might be a good therapeutic option.