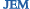The destruction (top) of TRAF2 (green) via SOCS2 (red) is blocked in mice treated with proteasome inhibitors (bottom).
Aspirin's power was initially attributed to its inhibition of proinflammatory lipids called prostaglandins—a discovery that won the Nobel Prize in 1982. Later, aspirin was also shown to beef up levels of lipids called lipoxins, which help resolve inflammation by blocking NF-κB activation and the recruitment of inflammatory cells.
Lipoxins were recently found to activate SOCS2, a protein that blocks signals from growth hormone receptors by targeting downstream signaling proteins to the proteasome. To determine whether SOCS2 also blocks inflammatory signals, Machado et al. fished for its binding partners among molecules that transmit innate immune receptor signals.
They now find that SOCS2 binds TRAF2 and TRAF6—adaptor proteins that are required for cytokine production by activated dendritic cells (DCs)—and seems to target them to the proteasome. Treating mice with aspirin decreased DC levels of cytokines and TRAF2 and TRAF6—effects that were mitigated by proteasome inhibitors. The same effects were not found in DCs from SOCS2-deficient mice.
Mice treated with a proteasome inhibitor after parasitic infection beat the bug but died from inflammatory damage. The deadly inflammation probably resulted from prolonged signaling via TRAF2 and TRAF6, as inflammation in SOCS2-deficient mice was severe even without the inhibitor. Infected mice that were left untreated resolved the inflammation and survived the infection.
Because proteasome inhibitors also block the degradation of IκB—the molecule that holds NF-κB in check—they are being developed as alternatives to aspirin, which can cause stomach problems and other side effects. But the current findings suggest that this strategy might be counterproductive.