Down syndrome (DS) is associated with chronic hyperactivation of interferon signaling and downstream Janus kinase (JAK) and STAT pathways, contributing to increased susceptibility to autoimmunity, autoinflammation, immunodeficiency, and immune dysregulation. JAK inhibitors represent a mechanistically rational therapeutic option, but systematic safety data in this vulnerable population are limited. This review evaluates the safety profile of JAK inhibitors in individuals with DS.
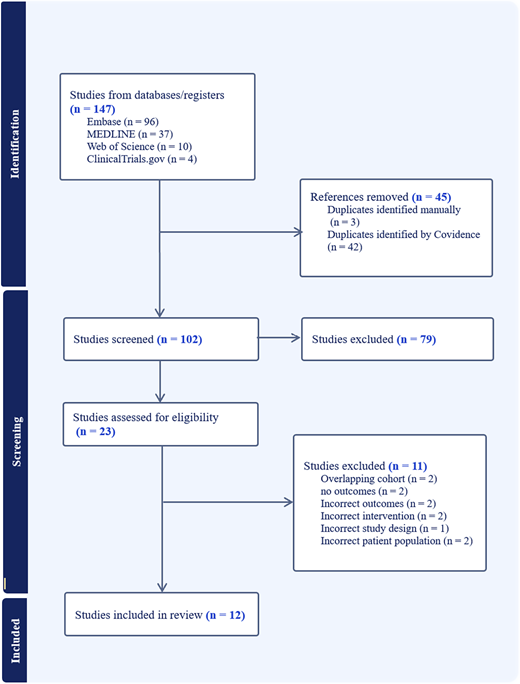
A systematic search of MEDLINE, Embase, Web of Science, and ClinicalTrials.gov identified 147 records. After deduplication, title and abstract screening, and full text review, 12 studies met the inclusion criteria. Eligible studies included human subjects with DS receiving a JAK inhibitor for any indication. Extracted variables included demographics, indication, agent, dose, duration, adverse events, infections, laboratory abnormalities, and treatment discontinuation. Studies sharing overlapping cohorts were examined to avoid double counting. The review protocol was registered in PROSPERO (CRD420251231172).
Twelve studies representing approximately 25 to 30 unique individuals with DS were included. JAK inhibitors used were tofacitinib, baricitinib, and ruxolitinib. Indications included DS-associated arthritis, alopecia areata, immune-mediated skin disease, DS regression disorder, and hemophagocytic lymphohistiocytosis. Treatment duration ranged from three months to more than two years. No deaths, venous thromboembolism, major cardiovascular events, or malignancies attributable to therapy were reported. Mild infections were the most common adverse events and generally resolved without treatment interruption. One child receiving baricitinib developed COVID-19 pneumonia but did not experience recurrence of hyperinflammatory disease. Laboratory abnormalities were infrequent, mild, and reversible. In the largest interventional dataset, no serious adverse events were attributed to tofacitinib during the sixteen-week primary analysis period. Long-term case reports similarly demonstrated stable monitoring without significant toxicity.
Current evidence suggests that short-term and intermediate-term use of JAK inhibitors in individuals with DS is associated with an acceptable safety profile. No increased signal for serious infection, thrombosis, malignancy, or cardiovascular events was observed. Larger prospective studies are required to define long-term safety and guide clinical use in this immunologically distinct population.