Increased risk of infections in patients with hematological malignancies (HM) is a growing concern. Immunoglobulin-replacement therapy (IgRT) is used to reduce infection risk in patients who develop secondary hypogammaglobulinemia (HGG). The effectiveness of IgRT in preventing infections, including COVID-19, has not been fully assessed. This study aims to assess the effectiveness of 10% human intravenous immunoglobulin caprylate-chromatography purified (IGIV-C10%) in reducing infection risk in HM patients with secondary HGG, including COVID-19 infections.
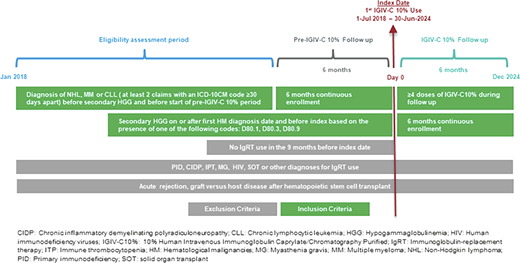
A retrospective study was conducted using PharMetrics Plus. The study included patients with non-Hodgkin lymphoma (NHL), multiple myeloma (MM), or chronic lymphocytic leukemia (CLL) who developed HGG and initiated treatment with IGIV-C10% between July 2018 and June 2024. The index date was the first IGIV-C10% dose. Figure 1 includes additional details on study design and eligibility. The proportion of patients with severe infections and any infection over the post-index (exposed) period was compared to that during the pre-index (unexposed) period (including and excluding COVID-19 infections). A sensitivity analysis based on the month-year of the index was performed.
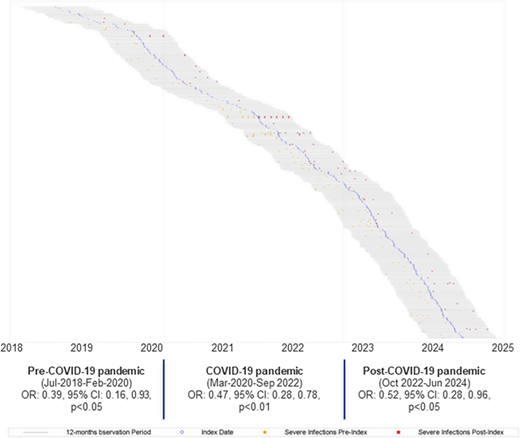
Overall, 348 patients (NHL: 130, MM: 154, CLL: 64) were included, with a mean (SD) age of 64.6 (10.9) years; 53.2% were male. Of these, 62 (17.8%), 123 (35.3%), and 163 (46.8%) patients were identified during the pre-COVID-19, COVID-19, and post-COVID-19 periods, respectively. Patients received 5.8 (1.2) IGIV-C10% doses, with a mean of 21.8 (17.5) grams-encounter. When excluding COVID-19 infections, the adjusted odds of severe infection and any infection were 53% (OR: 0.47; 0.33, 0.66; p < 0.001) and 66% (odds ratio [OR]: 0.34; 0.23–0.50; p < 0.001) lower during the IGIV-C10% period, respectively. The same trend was observed when including COVID-19 infections in the definitions of severe infections (OR: 0.41; 0.28, 0.60; p < 0.001) and any infections (OR: 0.36; 0.24, 0.54, p < 0.001). Similarly, the odds of severe infection were significantly reduced regardless of whether patients initiated IGIV-C10% therapy before, during, or after the COVID-19 pandemic (Figure 2).
Graphical representation of patients included in the study based on index day and presence of severe infections during the post-index and pre-index periods, and adjusted odds, 95% confidence intervals, and p-value of severe infections during the post-index (exposed to IGIV-C10%) compared to the pre-index (unexposed to IGIV-C10%).
Graphical representation of patients included in the study based on index day and presence of severe infections during the post-index and pre-index periods, and adjusted odds, 95% confidence intervals, and p-value of severe infections during the post-index (exposed to IGIV-C10%) compared to the pre-index (unexposed to IGIV-C10%).
Regular IGIV-C10% infusions are effective in reducing infections in HM patients with HGG. Similar effects were observed when including COVID-19 infections as an outcome and when effectiveness was assessed before, during, and after the COVID-19 pandemic.