Solid organ transplantation is an effective treatment for end-stage organ diseases, including renal, hepatic, cardiac, and pulmonary failure (1). Long-term graft survival depends on sustained immunosuppression; however, chronic exposure to immunosuppressive agents can impair humoral immunity and lead to secondary immune deficiencies (2). Secondary hypogammaglobulinemia, particularly with impaired specific antibody responses, increases vulnerability to recurrent or severe infections (3). Early recognition and coordinated management can significantly reduce morbidity and healthcare utilization.
A 67-year-old woman with a history of end-stage renal disease (ESRD) secondary to lupus nephritis, status post kidney transplant 13 years prior, was referred by the renal transplant team to a community immunologist for evaluation of recurrent infections, including Clostridioides difficile colitis, recurrent bacterial pneumonias, bronchitis, and sinusitis over the preceding year. Her immunosuppressive regimen—mycophenolate, tacrolimus, and low-dose prednisone—had been stable for 13 years with excellent graft function. She had no personal or family history of primary immunodeficiency.
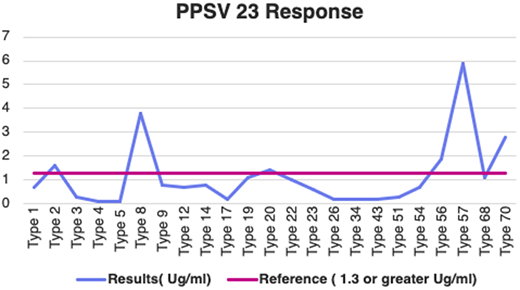
Immunologic evaluation revealed isolated hypogammaglobulinemia (IgG 563 mg/dL) with normal IgA, IgM, and IgE. Specific antibody testing demonstrated poor response to pneumococcal vaccines (Figure 1) but preserved responses to tetanus and diphtheria. Flow cytometry showed normal natural killer (NK), CD3, and CD8 T cell counts, mildly decreased CD4 counts (157 cells/uL), and reduced CD19 B cells (7 cells/uL), attributed to her chronic immunosuppressive regimen. Findings were consistent with secondary hypogammaglobulinemia related to prolonged immunosuppression.
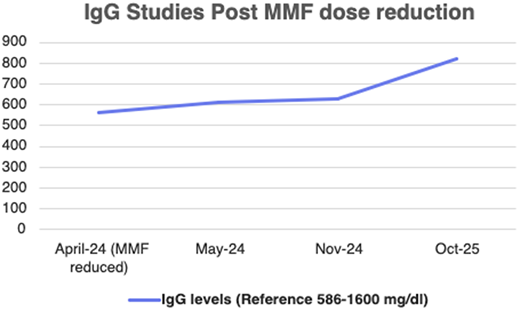
The immunologist discussed management options with the patient and the renal transplant team. These included immunoglobulin replacement therapy (intravenous immunoglobulin [IVIG]/subcutaneous immunoglobulin [SCIG]) and/or modification of immunosuppressive therapy. Through shared decision-making, the patient opted for a dose reduction of mycophenolate (to 180 mg BID) with close monitoring. On subsequent follow-ups in the immunology clinic, her IgG levels gradually improved to 821 mg/dl (Figure 2). There was a marked reduction in infections with no further hospitalizations and stable graft function.
Secondary hypogammaglobulinemia remains an important and under-recognized cause of specific antibody deficiency in solid organ transplant recipients. Early diagnosis and timely intervention are essential to preventing recurrent and potentially life-threatening infections. This case highlights a community immunologist-led, multidisciplinary approach playing a pivotal role in identifying and managing acquired immune deficiencies, ultimately strengthening the continuum of transplant immunology care.