Wiskott-Aldrich syndrome (WAS) is an X-linked immunodeficiency disorder frequently associated with systemic autoimmunity, including autoantibody-mediated cytopenias. WAS protein (WASp)–deficient B cells have increased B cell receptor (BCR) and Toll-like receptor (TLR) signaling, suggesting that these pathways might impact establishment of the mature, naive BCR repertoire. To directly investigate this possibility, we evaluated naive B cell specificity and composition in WASp-deficient mice and WAS subjects (n = 12). High-throughput sequencing and single-cell cloning analysis of the BCR repertoire revealed altered heavy chain usage and enrichment for low-affinity self-reactive specificities in murine marginal zone and human naive B cells. Although negative selection mechanisms including deletion, anergy, and receptor editing were relatively unperturbed, WASp-deficient transitional B cells showed enhanced proliferation in vivo mediated by antigen- and Myd88-dependent signals. Finally, using both BCR sequencing and cell surface analysis with a monoclonal antibody recognizing an intrinsically autoreactive heavy chain, we show enrichment in self-reactive cells specifically at the transitional to naive mature B cell stage in WAS subjects. Our combined data support a model wherein modest alterations in B cell–intrinsic, BCR, and TLR signals in WAS, and likely other autoimmune disorders, are sufficient to alter B cell tolerance via positive selection of self-reactive transitional B cells.
Development of the adaptive immune system requires selection of antigen receptors to establish a diverse but self-tolerant lymphocyte repertoire. Mechanisms to prevent selection of autoreactive B lymphocytes include clonal deletion, anergy, and receptor editing (Nemazee, 2006; Meffre and Wardemann, 2008). Alternatively, a growing body of literature also suggests that antigen-dependent positive selection of transitional B cells can occur via increased survival and/or clonal expansion (Hayakawa et al., 1999; Levine et al., 2000; Gaudin et al., 2004; Meyer-Bahlburg et al., 2008; Zikherman et al., 2012). These negative and positive selection mechanisms function in concert to shape the mature naive B cell repertoire.
Positive selection of transitional B cells is regulated by tonic B cell receptor (BCR) signaling (Stadanlick et al., 2008), signaling via the cytokine B cell–activating factor (BAFF; Stadanlick and Cancro, 2008), and T cell help via CD40L-CD40 signaling (Schwartz et al., 2014) to promote cell survival. Positive selection may help to select BCR specificities that maintain key homeostatic functions, including apoptotic cell clearance or conserved pathogen recognition (Grönwall and Silverman, 2014). Although positive selection can be beneficial for these important immune functions, enhanced positive selection of autoreactive BCRs, through incompletely defined mechanisms, is also predicted to occur in autoimmune-prone settings; this process is likely to lead to an enrichment in BCR specificities that may facilitate detrimental immune responses (Groom et al., 2002; Wang and Clarke, 2003; Eilat and Wabl, 2012).
In addition to BCR specificity, emerging data suggest a role for TLR signals in modulation of B cell selection. Previous data have shown that TLR signaling adapters, including MyD88, IRAK-4, and UNC93b, may operate in conjunction with the BCR to facilitate negative selection of autoreactive B cells (Isnardi et al., 2008). In contrast to promoting negative selection in immature B cells, dual signals mediated via the BCR and TLR pathways in mature B cells (Leadbetter et al., 2002; Groom et al., 2007; Silver et al., 2007; Rawlings et al., 2012) markedly enhance B cell activation and may directly initiate humoral autoimmunity. In this latter setting, loss in B cell tolerance occurs via generation of self-reactive, germinal center responses, leading ultimately to production of class-switched pathogenic autoantibodies (Jackson et al., 2015). Notably, although these combined data implicate TLR/MyD88 signals in both early and late B cell tolerance checkpoints, a potential role of BCR and/or TLR engagement in transitional B cell positive selection into the naive mature B cell compartment has not been defined.
Wiskott-Aldrich syndrome (WAS) is an X-linked immunodeficiency that results from mutations within the gene encoding the WAS protein (WASp), a key multiadapter protein linking a broad range of receptor signaling effectors to the actin cytoskeleton. This complex disorder is characterized by multiple alterations in hematopoietic cell surface receptor signal transduction, cell trafficking, and lineage- and developmental subset–specific homeostasis. Notably, up to 70% of WAS patients exhibit autoimmunity, including autoantibody-mediated cytopenias and organ-specific disease (Notarangelo and Ochs, 2003; Ochs and Thrasher, 2006; Bosticardo et al., 2009). In previous work, we have shown that WASp deficiency modestly enhances both BCR and TLR signaling in naive B cells (Becker-Herman et al., 2011). Furthermore, we and others have demonstrated that B cell–intrinsic WASp deficiency is sufficient to alter B cell tolerance and can promote production of class-switched autoantibodies and autoantibody-mediated autoimmune disease (Becker-Herman et al., 2011; Recher et al., 2012). This break in tolerance is associated with spontaneous GC formation and requires both BCR and TLR/MyD88 signaling (Becker-Herman et al., 2011; Jackson et al., 2014).
In this study, we hypothesized that increased BCR and TLR signaling in WASp-deficient B cells may also impact establishment of the mature, naive BCR repertoire. In partial support of this idea, previous studies have revealed evidence for skewing of heavy chain usage in both class-switched and bulk naive peripheral blood B cells isolated from WAS subjects (Castiello et al., 2014; O’Connell et al., 2014; Simon et al., 2014). Here, we present a detailed analysis of the impact of WASp deficiency on the selection of the naive B cell repertoire in mice and humans. Our combined data support a model wherein altered BCR and TLR signaling orchestrates increased positive selection of transitional B cells expressing low-affinity self-reactive BCRs, leading to their enrichment within the naive B cell compartment. As multiple human autoimmune disorders are associated with genetic changes potentially impacting B lineage signaling function, our findings have important implications with respect to understanding events that promote altered B cell selection in both WAS subjects and in other autoimmune-prone individuals.
RESULTS
WASp-deficient mice exhibit a skewed naive B cell repertoire enriched for low-affinity self-reactive BCRs
Increased λ–light chain (LC) usage in the mature B cell compartment of mice is correlated with increased autoreactivity (Doyle et al., 2006; Witsch and Bettelheim, 2008; Fournier et al., 2012). We therefore analyzed κ- versus λ-LC usage in splenic B cell subsets from young Was knockout (Was−/−) and control C57BL/6 (B6) mice. Although there were no differences in λ-LC usage in immature BM B cells (not depicted) or early transitional (T1) splenic B cells, we observed an increase in the percentage of λ-LC–expressing cells beginning in the late transitional (T2) subset and continuing within marginal zone (MZ) precursor (MZp) and mature naive (follicular mature [FM] and MZ) B cell subsets (Fig. 1 A). Consistent with a B cell–intrinsic process, equivalent findings were present in both Was−/− and B cell–specific WASp-deficient mice (Wasfl/flMb-1cre), where the Was gene is deleted early in BM B cell development via lineage-specific Cre expression mediated by the Mb-1 promoter (Recher et al., 2012).
Altered specificity of the naive B cell repertoire in WASp-deficient mice. (A) λ-LC usage in splenic B cell populations in 8–10-wk-old B6 (n = 6), Wasfl/fl × Mb-1cre (n = 5), and Was−/− (n = 7) mice assessed by flow cytometry. (B and C) Cloned WT and Was−/− MZ B cell mAb reactivities toward self-antigens (dsDNA, high [PC-4]- and low [PC-14]-affinity phosphorylcholine, MDA-LDL, and Sm-RNP) via ELISA depicted using a pie chart (blue = reactive clones identified based on threshold of 0.5 OD value; gray = nonreactive clones), with percentages of reactive clones and total number of clones tested noted. MZ B cells were FACS sorted and gated based on B220+CD23loCD1dhiCD24hiCD21hi surface marker expression from splenocytes pooled from five to six WT or Was−/− mice. (C) ELISA OD values of serial dilution curves of WT and Was−/− MZ mAbs (100 ng/µl). (D) Proportion of low affinity (OD of 0.5–1.5) and high affinity (OD of 1.5–3) in reactive antibody clones to individual self-antigens. (E) Relative binding affinity displayed as AUC of reactive antibodies. (F) Selection of Id (M167)+ B cells in peripheral B cell subsets in 10–12-wk-old WT M167 Tg (n = 9) and Was−/− M167 Tg (n = 9) mice. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Data are representative of at least two experiments.
Altered specificity of the naive B cell repertoire in WASp-deficient mice. (A) λ-LC usage in splenic B cell populations in 8–10-wk-old B6 (n = 6), Wasfl/fl × Mb-1cre (n = 5), and Was−/− (n = 7) mice assessed by flow cytometry. (B and C) Cloned WT and Was−/− MZ B cell mAb reactivities toward self-antigens (dsDNA, high [PC-4]- and low [PC-14]-affinity phosphorylcholine, MDA-LDL, and Sm-RNP) via ELISA depicted using a pie chart (blue = reactive clones identified based on threshold of 0.5 OD value; gray = nonreactive clones), with percentages of reactive clones and total number of clones tested noted. MZ B cells were FACS sorted and gated based on B220+CD23loCD1dhiCD24hiCD21hi surface marker expression from splenocytes pooled from five to six WT or Was−/− mice. (C) ELISA OD values of serial dilution curves of WT and Was−/− MZ mAbs (100 ng/µl). (D) Proportion of low affinity (OD of 0.5–1.5) and high affinity (OD of 1.5–3) in reactive antibody clones to individual self-antigens. (E) Relative binding affinity displayed as AUC of reactive antibodies. (F) Selection of Id (M167)+ B cells in peripheral B cell subsets in 10–12-wk-old WT M167 Tg (n = 9) and Was−/− M167 Tg (n = 9) mice. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Data are representative of at least two experiments.
Based on these findings and previous work characterizing autoantibody production in Was−/− mice, we expanded our analysis to determine whether WASp deficiency impacts the spectrum of BCR specificities within the mature, naive repertoire. We focused our analysis on MZ B cells as this compartment exhibited the greatest enrichment in λ-LC–expressing B cells, cells predicted to be enriched for polyreactive specificities (Li et al., 2002; Zhou et al., 2011). Using established methods (Wardemann et al., 2003; Schwartz et al., 2014), BCRs were cloned from single MZ B cells sorted from WT and Was−/− mice producing 74 WT and 64 Was−/− MZ recombinant mAbs. As anticipated for naive B cells, BCR clones lacked evidence for somatic hypermutation (SHM; not depicted). Antibody specificity was evaluated with ELISA assays for a series of self-antigens, including dsDNA, high- and low-affinity phosphorylcholine (PC-4 and PC-12), malondialdehyde (MDA)-LDL, and Sm-RNP (Fig. 1 B). Antibodies were defined as reactive if the observed OD was greater than a threshold value set at 0.5 OD. The proportion of mAbs reactive with RNA antigens was increased in Was−/− (32% reactive) versus WT (17%; Fig. 1 B). Furthermore, although we observed no significant differences in the percentage of reactive clones to other self-antigens, a much larger proportion of self-reactive Was−/− MZ clones exhibited low-affinity responses (OD level of 0.5–1.5) than in WT (Fig. 1 D). Consistent with this observation, calculation of the area under the curve (AUC) for clones reactive to each self-antigen revealed a trend toward decreased AUC for Was−/− clones (Fig. 1, C–E). Thus, WASp deficiency is associated with increased λ-LC usage in transitional and mature B cells and an increase in both RNA-reactive BCRs and other lower-affinity autoreactive clones within the MZ B cell compartment.
Next, we evaluated whether WASp deficiency impacted selection of B cells expressing a high-affinity self-reactive BCR. We used the M167 transgenic (Tg) model to assess positive selection of PC-specific idiotype (Id)+ late-transitional and MZ B cells (Kenny et al., 1991; Meyer-Bahlburg et al., 2008). Strikingly, Was−/− M167 Tg mice showed significantly reduced percentages of M167 Id+ B cells compared with WT mice. This change was evident in T2 B cells and retained in MZp and MZ cells, findings consistent with reduced selection for this high-affinity self-reactive BCR (Fig. 1 F). Of note, serum low-affinity PC-specific IgM and IgG were markedly higher in Was−/− × M167 Tg mice compared with WT × M167 Tg mice (not depicted), indicating that although high-affinity BCRs are not selected, there is enhanced production of low-affinity PC-specific autoantibodies in the absence of WASp. This trend toward increased selection to lower-affinity autoantibodies is further supported by the increase of low-affinity (PC-14) autoantibodies in the MZ compartment in Was−/− mice.
High-throughput BCR sequencing reveals an altered healthy control (HC) repertoire in Was−/− MZ B cells
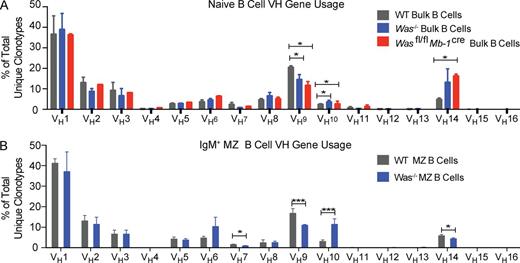
To expand the scope of our repertoire analysis, we sequenced BCR heavy chain genes from peripheral B cell subsets. We used 5′-rapid amplification of cDNA ends (RACE) followed by 454-pyrosequncing of amplicons generated using IgD and IgM gene-specific primers (Fig. 2, A and B). To limit any potential PCR amplification–induced bias, data analysis was restricted only to assessment of unique clonotypes. Over 80,000 unique sequences were identified from three independent experiments (each performed using pooled high-purity sorted B cell populations derived from at least five animals/genotype), resulting in 4,000–20,000 unique clonotypic sequences for each subset examined (Table S1). Analysis of the total splenic B cell repertoire revealed altered VH gene family usage in Was−/− mice, with a significant decrease in VH9 and an increase in both VH10 and VH14 usage (Fig. 2 A). Importantly, we confirmed that these changes were caused by B cell–intrinsic WASp deficiency as sequencing data in Wasfl/fl × Mb-1cre mice recapitulated these VH family alterations. Alternatively, analysis of Was−/− MZ B cells revealed a marked increase in VH10 expression as well as decreased VH7, VH9, and VH14 family usage (Fig. 2 B). Detailed analysis of mean HCDR3 lengths and JH usage did not reveal any statistically significant differences between WT and Was−/− B cell repertoires (not depicted). Together, these data demonstrate significant skewing of VH family gene usage in Was−/− naive mature B cells, including, in particular, a marked enrichment in VH10 and VH14 usage in MZ and bulk splenic B cell compartments, respectively, VH families commonly associated with natural autoantibodies in mice (Brigido et al., 1993; Matejuk et al., 2009; Jiang et al., 2011; Maranhão et al., 2013).
High-throughput BCR heavy chain sequencing of splenic B cell subsets from WT, Was−/−, and Wasfl/fl × Mb-1cre mice. B cell populations were sorted (total of ∼0.5–10 × 106 pooled cells/subset) from B6 WT, Was−/−, and Wasfl/fl × Mb-1cre mice using a minimum of five to six mice/genotype per experiment (three experiments total). RNA was isolated, sequenced, and analyzed using a 5′-RACE 454 platform (see Materials and methods). Data represent a mean of three experiments (15 mice per genotype). (A) Heavy chain variable (VH) gene family usage in bulk WT and Was−/− B cells (∼10 × 106 cells/sample). (B) VH gene family usage in sorted WT and Was−/− B220+CD21hiCD24hiCD23loCD1dhi MZ B cells (∼0.5–106 cells/sample). Additional sequence information is available in Table S1. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; ***, P < 0.001. Data are representative of at least three experiments.
High-throughput BCR heavy chain sequencing of splenic B cell subsets from WT, Was−/−, and Wasfl/fl × Mb-1cre mice. B cell populations were sorted (total of ∼0.5–10 × 106 pooled cells/subset) from B6 WT, Was−/−, and Wasfl/fl × Mb-1cre mice using a minimum of five to six mice/genotype per experiment (three experiments total). RNA was isolated, sequenced, and analyzed using a 5′-RACE 454 platform (see Materials and methods). Data represent a mean of three experiments (15 mice per genotype). (A) Heavy chain variable (VH) gene family usage in bulk WT and Was−/− B cells (∼10 × 106 cells/sample). (B) VH gene family usage in sorted WT and Was−/− B220+CD21hiCD24hiCD23loCD1dhi MZ B cells (∼0.5–106 cells/sample). Additional sequence information is available in Table S1. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; ***, P < 0.001. Data are representative of at least three experiments.
WASp deficiency does not perturb negative selection mechanisms
We next used Tg models to determine whether the observed changes in the Was−/− naive BCR repertoire (identified via clonal analysis and sequencing experiments) result from impaired negative selection. We evaluated both clonal deletion and anergy of WASp-deficient B cells using the hen egg lysozyme (HEL), anti-HEL BCR (MD4) double Tg mouse model, where Tg B cells express a high-affinity BCR specific for the foreign antigen, HEL (Goodnow et al., 1988; Cyster et al., 1996). Mixed BM chimeras were generated using WT or Was−/− MD4 Tg mice as donors. To emulate high- versus low-affinity interactions with self-ligand, BM was transplanted into irradiated recipient mice expressing either membrane-bound HEL (mHEL) or soluble HEL (sHEL), respectively, or into controls lacking self-antigen (B6; Fig. 3, A–E). Both Was−/− and WT MD4 Tg B cells were deleted in mHEL recipient mice, and HEL-specific serum antibodies were eliminated, indicating that in this monoclonal-setting clonal deletion is unperturbed in Was−/− B cells (Fig. 3, A–C). Similarly, we observed no defects in anergy induction in Was−/− B cells in sHEL-expressing recipient mice with similar numbers of BM and splenic B cells (not depicted), equivalent surface MD4 down-regulation (Fig. 3, D and E), and elimination of HEL-specific antibodies (Fig. 3 F). In addition to deletion or anergy of autoreactive B cells, tolerance is also achieved via induction of antibody LC receptor editing. To evaluate whether this process is intact in Was−/− B cells, we used a quantitative assay for recombining sequence (RS) rearrangement. RS rearrangement occurs via recombination of a noncoding gene segment in the κ-LC locus (Panigrahi et al., 2008). RS rearrangement levels were essentially identical in WT and Was−/− cells at each stage of BM B cell development (fraction D and κ-expressing fractions E and F) as well as in splenic B cell subsets (κ-expressing T1, T2, and FM B cells; Fig. 3 G). Collectively, these results indicate Was−/− mice manifest each of the key mechanisms responsible for negative selection of autoreactive immature B cells, suggesting that alterations in these events are unlikely to be primarily responsible for the observed changes in the BCR repertoire.
Was−/− B cells exhibit intact negative selection. (A–C) The HEL-Ig/mHel double Tg (DTg) chimeric transplant model was used to determine whether self-reactive Was−/− B cells undergo deletion in the BM. Recipients were analyzed at 6 wk after transplant (n = 6–7 per group; data are representative of two independent experiments). (A and B) BM B cells were analyzed by FACS. (C) HEL-specific serum antibody levels. (D–F) The HEL-Ig/sHel DTg system was used to determine whether self-reactive Was−/− B cells undergo efficient induction of anergy (n = 6–7 per group; data are representative of two independent experiments). (D and E) BM B cells were analyzed by FACS. (F) HEL-specific serum antibody levels. (G) Quantitative assay for RS rearrangement was used to estimate κ-LC rearrangement in BM and spleen B cell subsets of 6–8-wk-old WT or Was−/− mice. Results are normalized to β-actin and presented as fold difference relative to WT κ+ FM B cells (n = 7 in each group except for Was−/− fractions E [n = 6] and F [n = 5]). Error bars show SEM. Statistical analysis was performed using the Student’s t test: ***, P < 0.001. Data are representative of at least two independent experiments.
Was−/− B cells exhibit intact negative selection. (A–C) The HEL-Ig/mHel double Tg (DTg) chimeric transplant model was used to determine whether self-reactive Was−/− B cells undergo deletion in the BM. Recipients were analyzed at 6 wk after transplant (n = 6–7 per group; data are representative of two independent experiments). (A and B) BM B cells were analyzed by FACS. (C) HEL-specific serum antibody levels. (D–F) The HEL-Ig/sHel DTg system was used to determine whether self-reactive Was−/− B cells undergo efficient induction of anergy (n = 6–7 per group; data are representative of two independent experiments). (D and E) BM B cells were analyzed by FACS. (F) HEL-specific serum antibody levels. (G) Quantitative assay for RS rearrangement was used to estimate κ-LC rearrangement in BM and spleen B cell subsets of 6–8-wk-old WT or Was−/− mice. Results are normalized to β-actin and presented as fold difference relative to WT κ+ FM B cells (n = 7 in each group except for Was−/− fractions E [n = 6] and F [n = 5]). Error bars show SEM. Statistical analysis was performed using the Student’s t test: ***, P < 0.001. Data are representative of at least two independent experiments.
WASp deficiency promotes antigen-mediated proliferation of transitional B cells
Another hypothesis for altered repertoire is the expansion of positively selected clones. We first examined proliferation kinetics in transitional B and other B cell subsets as one potential surrogate measure of altered positive selection. After 24 h of in vivo BrdU labeling in Was−/−, Wasfl/flMb-1cre, or WT mice, we estimated the percentage of cycling cells within BM and splenic B cell subsets. Although there was no significant difference in BrdU incorporation within BM B cell subsets, we observed a significant increase in the percentage of BrdU+ transitional B cells in both Was−/− and Wasfl/flMb-1cre mice compared with WT control animals (Fig. 4 A). To further evaluate cycling, we labeled B cell subsets with DAPI and Pyronin Y and analyzed the percentage of B cells in G0 versus G2/S phase using flow cytometry (Fig. 4 B). Transitional, MZp, and MZ subsets in Was−/− mice contained significantly higher proportions of cycling cells (Fig. 4 B). Finally, we also observed evidence for enhanced cycling in Was−/− B cell subsets using Ki67 labeling (Fig. 5, A and B; and not depicted). Importantly, enhanced cycling was a BAFF-independent phenotype. Although Was−/− mice exhibit elevated serum BAFF, Wasfl/flMb-1cre mice have normal BAFF levels yet manifest similar increased rates of peripheral B cell proliferation (Fig. 4 C). Direct measurement of splenic BAFF protein and mRNA revealed no significant difference in WT versus Was−/− genotypes (not depicted), additionally supporting the idea that local BAFF levels were unlikely to impact this cycling phenotype. Thus, using multiple approaches we identified a marked increase in the proportion of cycling transitional B cells in Was−/− and/or Wasfl/flMb-1cre mice.
Was−/− B cells exhibit high levels of antigen-dependent clonal expansion. (A) 8–10-wk-old WT (n = 7), Was−/− (n = 9), and Wasfl/fl × Mb-1Cre (n = 6) mice were treated with BrdU in vivo for 24 h. BM and splenic B cell subsets were analyzed for BrdU incorporation via FACS. Data are representative of one of three experiments (WT, n = 3; Was−/−, n = 3; Wasfl/fl × Mb-1Cre, n = 3). (B) Cell cycle analysis of splenic B cell subsets via DAPI labeling in 8–10-wk-old WT (n = 7), Was−/− (n = 9) and Wasfl/fl × Mb-1Cre (n = 6) mice. (C) Serum BAFF levels in WT (n = 11), Was−/− (n = 9), and Wasfl/fl × Mb-1cre (n = 6) mice. (D) Representative data showing GFP staining of splenic T1 (left) and T2 (right) B cells in WT and Was−/− Nur77 Tg mice. (E and F) Percentage of GFPhi and GFPlo T2 B cells (E) and MFI of GFP in B cell subsets (F) in WT and Was−/− Nur77 Tg mice (n = 8/each). (G) Percentage of BrdU+ T2 GFPhi and GFPlo cells in WT (n = 7) and Was−/− Nur77 Tg (n = 6) mice. (G and H) 8–10-wk-old WT M167 Tg (n = 5) and Was−/− M167 Tg mice (n = 5) were treated with BrdU in vivo for 24 h. (H) Representative contour FACS plot showing BrdU incorporation in WT (top) versus Was−/− (bottom) M167 (Id)+ T2 B cells. (I) Percentage of M167 Id+Ki-67+ and Id−Ki-67+ splenic T2 B cells in WT (n = 5) versus Was−/− M167 (n = 6) Tg mice. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Data are representative of at least two experiments.
Was−/− B cells exhibit high levels of antigen-dependent clonal expansion. (A) 8–10-wk-old WT (n = 7), Was−/− (n = 9), and Wasfl/fl × Mb-1Cre (n = 6) mice were treated with BrdU in vivo for 24 h. BM and splenic B cell subsets were analyzed for BrdU incorporation via FACS. Data are representative of one of three experiments (WT, n = 3; Was−/−, n = 3; Wasfl/fl × Mb-1Cre, n = 3). (B) Cell cycle analysis of splenic B cell subsets via DAPI labeling in 8–10-wk-old WT (n = 7), Was−/− (n = 9) and Wasfl/fl × Mb-1Cre (n = 6) mice. (C) Serum BAFF levels in WT (n = 11), Was−/− (n = 9), and Wasfl/fl × Mb-1cre (n = 6) mice. (D) Representative data showing GFP staining of splenic T1 (left) and T2 (right) B cells in WT and Was−/− Nur77 Tg mice. (E and F) Percentage of GFPhi and GFPlo T2 B cells (E) and MFI of GFP in B cell subsets (F) in WT and Was−/− Nur77 Tg mice (n = 8/each). (G) Percentage of BrdU+ T2 GFPhi and GFPlo cells in WT (n = 7) and Was−/− Nur77 Tg (n = 6) mice. (G and H) 8–10-wk-old WT M167 Tg (n = 5) and Was−/− M167 Tg mice (n = 5) were treated with BrdU in vivo for 24 h. (H) Representative contour FACS plot showing BrdU incorporation in WT (top) versus Was−/− (bottom) M167 (Id)+ T2 B cells. (I) Percentage of M167 Id+Ki-67+ and Id−Ki-67+ splenic T2 B cells in WT (n = 5) versus Was−/− M167 (n = 6) Tg mice. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Data are representative of at least two experiments.
Antigen-specific selection of Was−/− transitional B cells requires Myd88 signals. (A) Representative FACS analysis of Ki67 staining of splenic T1 (B220+CD24hiCD21lo) and T2 (B220+CD24hiCD21mid) B cells in WT, Was−/−, and Was−/−Myd88−/− mice. (B) Percentage of Ki67+ B cells in WT (n = 12), Was−/− (n = 13), and Was−/−Myd88−/− (n = 12) mice. (C) VH gene family usage in sorted WT, Was−/−, and Was−/−Myd88−/− MZ B cells. Error bars show SEM. Statistical analysis was performed using the Student’s t test: **, P < 0.01. Data are representative of at least two experiments.
Antigen-specific selection of Was−/− transitional B cells requires Myd88 signals. (A) Representative FACS analysis of Ki67 staining of splenic T1 (B220+CD24hiCD21lo) and T2 (B220+CD24hiCD21mid) B cells in WT, Was−/−, and Was−/−Myd88−/− mice. (B) Percentage of Ki67+ B cells in WT (n = 12), Was−/− (n = 13), and Was−/−Myd88−/− (n = 12) mice. (C) VH gene family usage in sorted WT, Was−/−, and Was−/−Myd88−/− MZ B cells. Error bars show SEM. Statistical analysis was performed using the Student’s t test: **, P < 0.01. Data are representative of at least two experiments.
To begin to dissect the signals promoting increased transitional B cell proliferation in Was−/− mice, we used the Nur77-GFP Tg model, where GFP expression driven by the Nur77 promoter permits assessment of BCR-mediated signaling (Zikherman et al., 2012). In this model, WT T1 B cells exhibit little or no GFP expression, whereas a proportion of T2 B cells become GFPhi, consistent with self-antigen–mediated BCR signaling initially occurring within the late transitional compartment (Fig. 4 D, right vs. left panels). Strikingly, Was−/− Nur77 Tg mice showed roughly a twofold increase in the relative proportion of T2 GFPhi B cells (Fig. 4, D and E). Furthermore, consistent with increased BCR signaling, Was−/− GFP+ T2 B cells exhibited increased GFP mean fluorescence intensity (MFI) compared with control T2 B cells (Fig. 4 F). Next, we combined analyses using in vivo BrdU labeling with assessment using the Nur77-GFP reporter. Consistent with the idea that BCR engagement promotes the cycling of T2 B cells, the majority of T2 BrdU+ cells were located within the GFPhi population (Fig. 4 G).
To determine whether self-reactive T2 B cells underwent altered clonal expansion in a WASp-deficient setting, we measured cell cycling using dual assessment of BrdU incorporation and Ki67 expression using the M167 Tg self-reactive BCR model. Interestingly, although the overall percentage of cycling T2 B cells was higher in WASp-deficient mice, Was−/− T2 M167 Id+ B cells exhibited reduced cycling compared with WT M167 Id+ cells (Fig. 4, H and I). This preferential expansion of M167− Id− T2 B cells in Was−/− mice suggests that a larger proportion of lower-affinity clones are able enter the mature, naive repertoire.
Collectively, these findings suggest that the expanded, cycling T2 B cell population in Was−/− mice has engaged with self-antigen and that BCR engagement likely promotes and/or sustains cell cycling. Furthermore, these data suggest that WASp deficiency alters the breadth of self-reactive BCRs capable of competing for survival, limiting the competitive advantage of higher-affinity self-reactive clones, by permitting cells with lower-affinity self-reactive BCRs to more effectively compete and persist within the mature compartment.
Myd88 signals contribute to enhanced selection and the altered naive repertoire in Was−/− mice
Our previous work has shown that B cell–intrinsic MyD88 signals are required to initiate germinal center responses and promote the generation of pathogenic autoantibodies in Was−/− B cell chimeric mice (Becker-Herman et al., 2011; Jackson et al., 2014). We therefore hypothesized that MyD88 signals might also impact naive B cell selection in WASp-deficient animals. We first determined whether MyD88 signaling facilitated transitional B cell proliferation. Using flow cytometry, we measured Ki67+ T1 and T2 B cells in splenic B cell subsets derived from WT, Was−/−, and Was−/−Myd88−/− mice (Fig. 5 A). Strikingly, MyD88 deficiency markedly reduced the proliferation of WASp-deficient T1 and T2 B cells (Fig. 5, A and B). To further elucidate the impact of MyD88 signaling on the developing repertoire, Was−/−Myd88−/− MZ B cells were sorted and used for deep sequencing to evaluate BCR heavy chain gene usage. Although most VH families were not impacted, MyD88 deficiency specifically abrogated enrichment of VH10 and VH14 family BCRs in the Was−/− MZ and bulk splenic B cell compartments, respectively (Fig. 5 C and not depicted). Importantly, previous studies have correlated BCR VH10 and VH14 expression with nucleic acid reactivity (Brigido et al., 1993; Whitcomb et al., 1999; Jiang et al., 2011; Maranhão et al., 2013). Thus, our findings suggest that in the setting of WASp deficiency, MyD88 signals promote the selection of BCRs whose specificity correlates with anti-DNA and/or anti-RNA specificities, leading to the observed expansion of VH10 and VH14 family gene usage. Collectively, these data indicate that MyD88 signals contribute to the altered naive BCR repertoire in Was−/− mice.
Human WAS subjects also exhibit an altered naive B cell repertoire
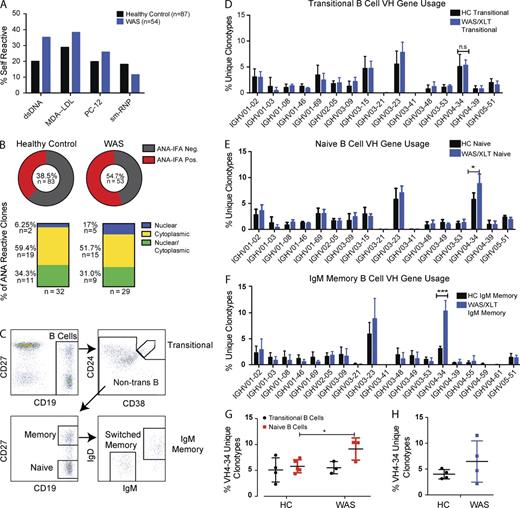
To determine whether BCR repertoire changes observed in the Was−/− murine model parallel findings in human subjects with WAS, we cloned BCRs from single mature naive B cells isolated by cell sorting from the peripheral blood of one pediatric WAS and two pediatric and one adult HC subjects (Fig. 6 C). Recombinant antibodies (54 WAS and 87 controls) were screened for reactivity to dsDNA, MDA-LDL, PC-12, and sm-RNP using ELISA assays. Compared with control BCRs, WAS BCRs were enriched for reactivity toward dsDNA, MDA-LDL, and PC-12 (Fig. 6 A) and also demonstrated increased staining for nuclear antigens using a Hep2 cell–based indirect fluorescence antibody (IFA) assay (Fig. 6 B). These initial data suggest that human WASp deficiency alters the naive B cell repertoire, leading to enrichment for B cells with self-reactive specificities.
WAS/XLT subjects exhibit an altered naive B cell repertoire enriched for self-reactive specificities. (A and B) BCRs were cloned from peripheral blood naive B cells from a 10-mo-old WAS subject (n = 54 mAbs) and two pediatric and one adult HC subjects (n = 87 antibodies). Percentages of mAb clones reactive to self-antigens are shown. Data are representative of one of two experiments. (B, top) Pie charts display frequency of ANA-IFA (antinuclear antibodies detected by IFA)–reactive clones. (bottom) Staining pattern of each IFA-reactive clone, defined as nuclear, cytoplasmic, or polyreactive (both nuclear and cytoplasmic), displayed according to overall percentages. (C) Human B cell subset gating and sorting strategy. (D–G) Sorted subsets from five HC and three WAS pediatric subjects were analyzed by Illumina high-throughput sequencing of the BCR heavy chain and combined to show mean VH family gene usage. (D) VH family usage of sorted transitional B cells (CD19+CD27+CD24hiCD38hi) displayed according to percentage of total unique clonotypes within the most abundant VH families; see Table S4 for full VH family usage. (E) VH family usage of naive B cells (CD19+CD27−). (F) VH family usage of IgM memory B cells (CD19+CD27+IgG−). (G) Percentage of VH4-34 within total unique, clonotypic sequences in naive B cell compartment. (H) Percentage of VH4-34 sequences in naive B cells in four HC (ages 23–29 yr) and four WAS adult subjects (ages 18–28 yr). Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; ***, P < 0.001. Data are representative of at least two experiments.
WAS/XLT subjects exhibit an altered naive B cell repertoire enriched for self-reactive specificities. (A and B) BCRs were cloned from peripheral blood naive B cells from a 10-mo-old WAS subject (n = 54 mAbs) and two pediatric and one adult HC subjects (n = 87 antibodies). Percentages of mAb clones reactive to self-antigens are shown. Data are representative of one of two experiments. (B, top) Pie charts display frequency of ANA-IFA (antinuclear antibodies detected by IFA)–reactive clones. (bottom) Staining pattern of each IFA-reactive clone, defined as nuclear, cytoplasmic, or polyreactive (both nuclear and cytoplasmic), displayed according to overall percentages. (C) Human B cell subset gating and sorting strategy. (D–G) Sorted subsets from five HC and three WAS pediatric subjects were analyzed by Illumina high-throughput sequencing of the BCR heavy chain and combined to show mean VH family gene usage. (D) VH family usage of sorted transitional B cells (CD19+CD27+CD24hiCD38hi) displayed according to percentage of total unique clonotypes within the most abundant VH families; see Table S4 for full VH family usage. (E) VH family usage of naive B cells (CD19+CD27−). (F) VH family usage of IgM memory B cells (CD19+CD27+IgG−). (G) Percentage of VH4-34 within total unique, clonotypic sequences in naive B cell compartment. (H) Percentage of VH4-34 sequences in naive B cells in four HC (ages 23–29 yr) and four WAS adult subjects (ages 18–28 yr). Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; ***, P < 0.001. Data are representative of at least two experiments.
To expand on these preliminary findings and to obtain a more in-depth assessment of the complete B cell repertoire in a larger number of subjects, we evaluated the transitional and naive B cell repertoire using high-throughput sequencing. Highly purified B cell subsets were isolated by flow sorting from pediatric WAS subjects (n = 3) and HCs (n = 4; Fig. 6 C). Genomic DNA was extracted from each sample and analyzed for heavy chain gene usage using Illumina-based deep sequencing. Comparison of the heavy chain V gene usage in transitional B cells revealed no statistically significant differences in WAS versus HC samples. In contrast, naive WAS B cells exhibited increased usage of the VH4-34 gene family (Fig. 6, D and E). Notably, VH4-34–expressing BCRs have been previously characterized as highly enriched for self- and polyreactivity (Pugh-Bernard et al., 2001; Jenks et al., 2013; Richardson et al., 2013). Although we observed no enrichment for VH4-34 sequences at the transitional to naive B cell transition in HC samples (n = 8), WAS subjects (n = 9) exhibited an ∼1.5-fold increase in VH4-34–expressing BCRs (Fig. 6 E). Notably, an earlier study has shown that although VH4-34 BCRs are present in the naive compartment in healthy subjects, they are nearly absent in the isotype-switched memory compartment and secreted antibody repertoire of healthy individuals. In contrast, lupus patients show a significant enrichment of VH4-34 in the memory compartment and in serum antibodies consistent with loss of tolerance in the GC reaction (Pugh-Bernard et al., 2001). Therefore, we also assessed the memory compartment of WAS patients and observed a significant increase in VH4-34–expressing IgM memory cells (Fig. 6 F). Although VH4-34 BCRs were expanded in WAS, these naive cells lacked SHM and IgM memory cells exhibited reduced rates of SHM compared with control (not depicted), consistent with the observed T cell defect in WAS subjects and a previous study of the WAS memory compartment (Castiello et al., 2014). Additional analysis of HDCR3 length and JH usage of HC and WAS subjects did not reveal significant differences (not depicted). Increased usage of VH4-34 heavy chain within the naive repertoire was also present in nontransplanted adult WAS patients (n = 4), indicating that skewing is evident as early as 1–3 yr of age and comprises a developmental feature maintained into adulthood (Fig. 6, G and H).
VH4-34–expressing BCRs are positively selected in WAS
Based on these BCR sequencing findings, we sought to further define the selection events operating upon VH4-34 HC–expressing B cells in a larger number of subjects with altered WASp function. We assessed the B cell compartment phenotype and tracked the selection of B cells in six pediatric WAS or X-linked thrombocytopenia (XLT) subjects and five age-matched HC subjects (Fig. 7, A–C). Phenotypic analysis of WAS and HC subjects revealed no significant differences in either transitional B cell population size or surface IgM expression (not depicted). We used the anti-idiotypic mAb 9G4, specific for the VH4-34 heavy chain, to track the selection of B cells throughout peripheral B cell development (Pugh-Bernard et al., 2001). Analysis of the transitional compartment revealed no significant difference in the relative proportion of 9G4+ B cells across all subjects, and there was no change in the proportion of 9G4+ cells in the naive versus transitional compartment in HCs. In contrast, there was a significant enrichment of 9G4+ naive mature B cells in WAS/XLT subjects, with a mean ∼2.5-fold increase in the naive versus transitional compartment, consistent with our sequencing data (available for three of these six subjects). Consistent with these findings, we observed a similar trend in adult WAS subjects (Fig. 7 D). To determine whether WAS/XLT 9G4+ B cells exhibit differences in VH4-34 BCR surface density, we assessed the MFI of 9G4 staining. Interestingly, transitional and naive WAS B cells exhibited a trend for higher 9G4 expression compared with HC B cell subsets (Fig. 7 E). Furthermore, although the 9G4 MFI declined markedly between the transitional and naive B cell stage in HCs, this change was less pronounced for WAS/XLT naive B cells. These changes are consistent with the lower surface expression of IgM in HC 9G4+ B cells and the postulated anergic behavior of these autoreactive B cells (Cappione et al., 2005).
WAS/XLT subjects exhibit increased selection for 9G4+ B cells specifically within the mature naive compartment. (A) Representative FACS histograms quantifying the percentage of 9G4+ peripheral blood transitional (left) and naive B cells (right) in age-matched HC and WAS subjects. (B) 9G4+ percentages of B cell subsets in HC (n = 5) and WAS subjects (n = 6). (C) Relative fold change in percentage of 9G4+ transitional versus naive B cells in WAS (n = 6) and HC subjects (n = 5). (D) Percentage of 9G4+ transitional versus naive B cells in HC (n = 4) and WAS adult subjects (n = 4). (E) Cumulative data showing the MFI of 9G4 staining in B cell subsets in HC (n = 6) and WAS pediatric subjects (n = 8). (F) Serum 9G4–specific IgG levels were determined using plasma collected from pediatric WAS (n = 8) and HC (n = 9) subjects. Titers are displayed as relative absorbance values, OD (450 nm). Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01. Data are representative of at least two experiments.
WAS/XLT subjects exhibit increased selection for 9G4+ B cells specifically within the mature naive compartment. (A) Representative FACS histograms quantifying the percentage of 9G4+ peripheral blood transitional (left) and naive B cells (right) in age-matched HC and WAS subjects. (B) 9G4+ percentages of B cell subsets in HC (n = 5) and WAS subjects (n = 6). (C) Relative fold change in percentage of 9G4+ transitional versus naive B cells in WAS (n = 6) and HC subjects (n = 5). (D) Percentage of 9G4+ transitional versus naive B cells in HC (n = 4) and WAS adult subjects (n = 4). (E) Cumulative data showing the MFI of 9G4 staining in B cell subsets in HC (n = 6) and WAS pediatric subjects (n = 8). (F) Serum 9G4–specific IgG levels were determined using plasma collected from pediatric WAS (n = 8) and HC (n = 9) subjects. Titers are displayed as relative absorbance values, OD (450 nm). Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01. Data are representative of at least two experiments.
In addition to changes in 9G4+ representation in naive B cell repertoire, we observed a significant enrichment for 9G4+ B cells in the total memory compartment (Fig. 7 B). Consistent with our deep sequencing analysis in pediatric WAS subjects, this change was specific to the IgM memory population and not present in the switched memory B cells (Fig. 7 B). To determine whether these alterations in the proportion of 9G4+ B cells predicted enhanced selection and activation of VH4-34–expressing B cells, we measured serum 9G4+ IgG levels by ELISA. We identified significantly increased levels of 9G4-specific IgG in WAS subjects compared with HCs (Fig. 7 F), findings consistent with a loss of tolerance in germinal center responses that recruit B cells with these autoreactive BCR specificities.
Together, these observations demonstrate altered selection of self-reactive WAS/XLT B cells precisely at the transitional to naive B cell transition and suggest that VH4-34–specific self-antigen engagement likely promotes differential survival of WAS/XLT versus HC 9G4+ B cells. Enrichment of these autoreactive specificities into the naive compartment promotes increased selection and generation of autoreactive class-switched antibodies in WAS subjects.
DISCUSSION
Expression of a sufficiently broad, naive BCR repertoire is essential both for the response to pathogen challenge and to orchestrate B cell–mediated homeostatic functions. The naive BCR repertoire is established via the interplay of negative and positive selection mechanisms that operate within distinct developmental windows and microenvironments. Importantly, alterations in these events are predicted to increase the risk for autoimmune disease. In the current study, we used animal models and human subjects with the immunodeficiency disorder WAS to gain new insight regarding the pathways involved within transitional B cells that modulate peripheral B cell selection. Our experiments establish a key role for combined BCR and TLR signals in orchestrating peripheral tolerance in the context of WASp deficiency and have important broader implications with respect to human B cell tolerance.
Using a series of animal models, we show that WASp deficiency significantly alters murine peripheral B cell tolerance as demonstrated by (a) increased λ-LC expression in T2, FM, and MZ B cells; (b) altered MZ B cell specificity with enrichment for lower-affinity self-reactive BCRs; (c) alteration in IgH family gene usage in the MZ compartment with, most notably, a striking increase in MZ B cells expressing VH10 family genes, previously shown to be associated with enhanced binding affinity for nucleic acids; and (d) a heightened frequency of late transitional B cells undergoing homeostatic proliferation (as summarized in Table S5). Furthermore, our data indicate that this proliferative program is mediated via both (a) self-antigen–driven BCR engagement, as demonstrated using a Tg Nur77-GFP reporter; and (b) TLR/Myd88 signaling, as shown by reduced proliferation in WASp-deficient B cells concurrently lacking Myd88. Finally, using deep sequencing, we demonstrate a direct requirement for MyD88 signaling in driving the selection of VH10- and VH14-expressing BCRs into the MZ and splenic B cell compartment, respectively, in Was−/− mice. Importantly, although the majority of our data were derived using Was−/− mice, we performed a subset of key experiments using Wasfl/flMb-1cre mice and observed essentially identical findings, including altered IGHV family usage, enhanced transitional cell cycling, and increased λ-LC expression. Also of note, although serum BAFF is modestly increased in Was−/− mice, the Wasfl/flMb-1cre model exhibits normal levels. Thus, although potentially exaggerated by alterations in the BAFF signaling program, these changes occur independently of BAFF signaling. In combination, our murine model data support the conclusion that cell-intrinsic signaling changes in WASp-deficient transitional B cells play a dominant role in altering B cell tolerance, leading to a skewing of the naive BCR repertoire.
Predicated on our murine findings, we performed experiments using highly purified peripheral blood B cell subsets derived from subjects with WAS (or the related, but less severe disorder XLT) and age-matched controls. Using single-cell BCR cloning, we observed evidence for altered naive mature B cell specificity with enrichment for self-reactive BCRs. Based on this finding, we expanded our analysis using two alternative approaches that directly demonstrate altered tolerance in WAS subjects precisely at the late transitional to naive mature B cell transition as shown by (a) deep sequencing showing changes in VH family usage specifically within the mature, naive repertoire, including increased utilization of the self-reactive VH4-34 family; and (b) flow cytometry using the mAb 9G4, specific to VH4-34, revealing preferential expansion of this inherently autoreactive specificity within the naive compartment (as summarized in Table S5). Together, these data provide a definitive demonstration of a human genetic disorder that specifically alters peripheral B cell tolerance at the transitional to naive B cell stage.
Collectively, our dataset is most consistent with a model wherein WASp deficiency promotes antigen-mediated positive selection of transitional B cells expressing low-affinity autoreactive BCRs into the naive compartment. Although we cannot eliminate the possibility that WASp deficiency may subtly alter, or perhaps enhance, negative selection, we extensively tested the impact of WASp on negative selection using Tg models as well as analysis of receptor editing in a polyclonal setting. We observed no differences from WT controls, indicating that WASp deficiency does not impact either central or peripheral negative selection as assessed using these specific Tg models or within the sensitivity of the editing assay. Importantly, WASp deficiency promotes rather than limits BCR signaling activity, and thus, altered negative selection is unlikely to explain the enrichment for specific self-reactive VH families at the transitional to naive B cell stage in both mice and humans. In addition to key survival programs mediated via BAFF-R, tonic BCR, and CD40 signaling (Schiemann et al., 2001; Lesley et al., 2004; Stadanlick and Cancro, 2008; Schwartz et al., 2014), recent work suggests that antigen-driven BCR signaling is a signature feature of B cells entering the late transitional (T2) compartment (Zikherman et al., 2012). Consistent with this idea, we previously identified a subpopulation of T2 B cells in normal mice that undergo homeostatic expansion in response to antigen-mediated BCR signaling (Meyer-Bahlburg et al., 2008). Together, our new findings in Was−/− mice showing (a) heightened frequency of transitional B cells undergoing homeostatic proliferation and (b) an increased proportion of GFPhi T2 B cells in Nur77-GFP Tg reporter mice suggest that antigen-mediated selection is enhanced in WASp-deficient T2 B cells. Furthermore, the idea that WASp deficiency preferentially promotes selection of B cells expressing lower-affinity autoreactive BCRs is supported by our single-cell cloning analysis, as well as by data derived using the self-reactive, anti-PC heavy chain Tg model. In these latter experiments, WASp-deficient T2 M167 Id+ (high affinity) B cells exhibited reduced proliferation compared with control cells, whereas, conversely, proliferation of M167 Id− (lower affinity) Was−/− B cells is increased, consistent with a broadening of competitive specificities using the PC-reactive heavy chain. Finally, our analysis in WAS subjects correlates directly with our murine data, as demonstrated by the selective expansion of B cells with self-reactive VH genes precisely at the transitional B to naive transition. Lastly, although we observed significant reduction in 9G4 expression at this transition in HC subjects, the change in expression was much less pronounced in WAS subjects, consistent with the idea that self-antigen engagement of VH4-34–specific transitional B cells leads to enhanced survival in the absence of WASp.
Importantly, this study also demonstrates a novel role for TLR signaling in orchestrating peripheral tolerance and implicates dual TLR/BCR signaling in promoting selection of autoreactive BCRs into the naive compartment. Previous work has linked TLR/Myd88 signaling to negative selection of immature B cells (Isnardi et al., 2008) and has extensively characterized dual TLR/BCR signals in activation of mature B cells and in triggering autoreactive GC responses (Leadbetter et al., 2002; Silver et al., 2007; Nickerson et al., 2010; Jackson et al., 2014). However, the impact of TLR or dual TLR/BCR signals in shaping the naive BCR repertoire has not been investigated. Here, we show that Myd88 deficiency markedly reduced the frequency of cycling T2 B cells in Was−/− mice, implying that Myd88 signals help to promote T2 B cell activation. Additionally, Myd88 deficiency abrogated the selection of VH10-expressing BCRs into the MZ, directly implicating Myd88 signals in modulating the naive repertoire. Finally, although we cannot formally link our observations to dual BCR/TLR signaling, this model is supported by both (a) the previously described nucleic acid reactivity of the selecting BCRs in both mouse and humans and (b) the finding that ablation of Myd88 eliminated VH10 enrichment. Surprisingly, investigation of individual TLRs (TLR 4, 7, and 9) revealed partial effects but suggests that no dominant TLR is responsible for the T2 proliferative response (unpublished data), implying that a complex interplay of antigen specificity and TLR coengagement likely orchestrates this dynamic process.
Previous studies have partially characterized the B cell repertoire in WAS subjects, including cloning and sequencing of limited numbers of sequences generated using VH3- and VH4-specific heavy chain primers from sorted B cell subsets (Castiello et al., 2014; Simon et al., 2014) and next generation sequencing of total CD19+ peripheral blood B cells (O’Connell et al., 2014). Collectively, these studies have revealed that WAS subjects exhibit altered VH gene family usage (specifically VH3- and VH4-expressing BCRs), skewed CDR3 lengths, a reduction in SHM in class-switched antibodies, and a potential enrichment for memory cells expressing self-reactive clones including VH4-34 and VH3-30 (associated with antiplatelet specificities; Castiello et al., 2014; O’Connell et al., 2014; Simon et al., 2014). Although these studies reveal an altered memory B cell compartment likely impacted by the T cell functional deficits in WAS, we tested whether WASp deficiency might directly impact the transitional and naive B cell repertoires. Based on single-cell studies of naive mature BCR specificity that suggested enhanced self-reactivity, we used deep sequencing of genomic DNA from three pediatric and four adult WAS subjects (and age-matched HCs) to obtain a quantitative assessment of the naive repertoire. Although we observed alterations in VH family gene usage within in the mature, naive repertoire in WAS subjects, there were little or no differences in the transitional compartment. The most dramatic change observed was enrichment in VH4-34 usage, a VH family with germline, intrinsic self-reactivity that is independent of both somatic mutation or associated LCs and that exhibits affinity toward multiple self-antigens including RBC carbohydrate antigens associated with cold-agglutinins, dsDNA, and CD45/B220 isoforms expressed on immature B cells (Cappione et al., 2004; Jenks et al., 2013; Richardson et al., 2013). This finding is clinically relevant as autoimmune RBC cytopenias, B cell lymphopenia, and increased circulating dsDNA autoantibodies are each frequent features in WAS (Notarangelo and Ochs, 2003). Importantly, the enrichment in VH4-34 usage paralleled our findings showing VH10 and VH14 enrichment in the murine model, where selection is directly impacted by self-antigen and TLR-dependent signals. Finally, we used a well-characterized mAb, 9G4, to directly track selection of B cells expressing these autoreactive specificities (Richardson et al., 2013). Our data clearly demonstrate preferential expansion of 9G4+ cells in the naive repertoire of WAS but not HC subjects. Notably, our human repertoire findings are not likely related to disease-associated inflammatory events as identical results were obtained in XLT subjects with no disease manifestations beyond thrombocytopenia. Interestingly, HC mature, naive B cells exhibited decreased surface 9G4 staining, suggesting that BCR down-regulation is a mechanism whereby VH4-34–expressing cells might be tolerized during normal development. In contrast, WASp-deficient B cells exhibited higher 9G4 MFI, consistent with expansion of this population, presumably via enhanced BCR and/or TLR signals. Finally, consistent with previous observations (Castiello et al., 2014; Simon et al., 2014), we saw additional enrichment for VH4-34 expression specifically within the IgM memory compartment in WAS subjects as well as significantly elevated serum 9G4+ IgG titers. Consistent with this finding, and similar to patients with systemic lupus erythematosus, a previous study has identified elevated levels of VH4-34 (previously referred to as VH4-21) IgM autoantibodies in WAS subjects (Grillot-Courvalin et al., 1992).
Together, these multiple lines of evidence support a scenario in which enhanced VH4-34 positive selection in WAS leads to enrichment of self-reactive B cells within the naive B cell compartment, and subsequently, as supported by our earlier work in murine models (Becker-Herman et al., 2011; Jackson et al., 2014), enhanced dual BCR/TLR signals orchestrate spontaneous activation of these self-reactive clones, leading to GC formation and production of VH4-34 IgG autoantibodies. This model is likely broadly relevant to other human autoimmune disorders including systemic lupus erythematosus, where waves of VH4-34 autoantibody–expressing antibody–secreting cells are generated from activated 9G4+ naive B cells, accounting for a major proportion of disease-relevant autoantibodies produced during lupus flares (Tipton et al., 2015). Additionally, enhanced BCR/TLR signals may allow for autoreactive B cells to better compete into the naive repertoire in other immunodeficiency disorders and conditions associated with significant B cell lymphopenia, leading to preferential selection for self-reactive specificities, including receptors using VH4-34 heavy chains.
In summary, this work provides new evidence supporting a model wherein modest alterations in BCR or TLR signal strength can alter the mature BCR repertoire and increase the risk for subsequent B cell–mediated autoimmunity. Based on these experiments, we anticipate that 9G4 mAb analysis may provide a biomarker for tracking B cell tolerance in subjects with WAS after transplantation or gene therapy, particularly in individuals with mixed chimerism where the risk for autoimmunity is increased. Finally, although WAS represents a rare genetic condition, there is mounting evidence that other human autoimmune risk alleles identified, for example, by genome-wide association studies may impact these key signaling cascades, implying relevance to many more common immune disorders (Dai et al., 2013; Jackson et al., 2015).
MATERIALS AND METHODS
Mice.
B6, Was−/−, Was−/−Myd88−/−, Wasfl/flMb-1cre, Was−/− MD4 Tg, MD4 Tg, M167 Tg, Was−/− M167 Tg, Nur77-GFP Tg, Was−/− Nur77-GFP Tg, sHEL, and mHEL mice were bred and maintained in the specific pathogen–free animal facility of Seattle Children’s Research Institute and handled according to Institutional Animal Care and Use Committee–approved protocols. M167 H chain mice (M167H Tg mice, line U243-4) were provided by J. Kenny and A. Lustig (National Institute on Aging, Bethesda, MD) and established as an M167H Tg/Tg homozygous breeding colony in the animal facility of the Albert Einstein College of Medicine (New York, NY) by S. Porcelli. Wasfl/flMb-1cre were provided by L.D. Notarangelo and A.J. Thrasher. Mb-1cre mice were provided by M. Reth (Max Planck Institute of Immunobiology, Freiburg, Germany).
Human WAS and HC sample preparation.
PBMCs were obtained after getting informed consent using a Seattle Children’s Research Institute Internal Review Board–approved research protocol. PBMCs were isolated from pediatric and adult WAS subjects or from HC subjects using Ficoll-Paque density centrifugation (followed by red blood cell lysis in 15 mM NH4Cl, 1 mM KHCO3, and 10 µM EDTA). For sequencing and flow cytometry assays, single-cell PBMC suspensions were enriched for B cells using CD19+ B Cell Isolation II negative selection kit (Miltenyi Biotec). Human WAS and HC subject information including age, WAS mutation, disease symptoms, and total cell numbers collected are described in Tables S2 and S3.
Reagents and antibodies.
Anti–murine antibodies used in this study include AA4.1, CD24 (M1/69), CD21 (7G6), B220 (RA3-6B2), and IgD (11-26C.2A) from BD; BP1 (FG35.4) and CD23 (B3B4) from Invitrogen; IgM (1B4B1), κ (187.1), λ (JC5-1), and SA-HRP conjugated from SouthernBiotech; CD19 (ID3) and IgMa (DS-1) from BioLegend; and Cy5 anti–rabbit polyclonal IgG from Jackson ImmunoResearch Laboratories, Inc. Alexa Fluor 647 anti-M167 (28–6-20) rat IgG2a was provided by S. Porcelli (Albert Einstein College of Medicine, Bronx, NY). Anti–human antibodies used in this study include CD19 (HIB19) and IgM (MHM-88) from BioLegend; CD10 (HI10a), CD24 (ML5), and IgD (IAG-2) from BD; CD27 (323) from eBioscience; and CD38 (HIT2) from BD. Anti–human rat monoclonal, FITC-conjugated 9G4 antibody was provided by I. Sanz.
Flow cytometry and cell sorting.
As previously described (Meyer-Bahlburg and Rawlings, 2008; Schwartz et al., 2014), single-cell suspensions from BM, peripheral blood, and spleen were incubated with fluorescently labeled antibodies, and data were collected on an LSRII (BD) and analyzed using FlowJo software (Tree Star). Cell sorting was performed using an Aria II (BD); sort purities were >90% in all experiments. Murine naive B cell subsets were gated as in (Schwartz et al., 2014) using B220, CD21, CD23, CD24, and CD1d surface markers. Human B cell subsets were gated as shown in Fig. 6 B.
BM transplantations.
For Hel-Ig experiments, Hel-Ig Tg mice were crossed to Was−/− mice to generate HEL-Ig Was−/− mice. BM was harvested from HEL-Ig Was−/− or HEL-Ig WT mice, and 5 × 106 cells in PBS were injected i.v. into lethally (1,050 cGy) irradiated mHEL or sHEL recipient mice.
Vκ-RS quantitative PCR.
Genomic DNA was isolated from sorted B cells using the Gentra PureGene kit (QIAGEN). Quantitative PCR was performed as described previously (Panigrahi et al., 2008). The amount of Vκ-RS product in each sample was normalized to the β-actin product and compared with the normalized target value in WT FM B220+ CT (DDCT method).
ELISA and HEp-2 IFA tests.
Antibodies were tested for reactivity to MDA-LDL (20P-MD L-105; Academy Bio-Medical), PC-4, PC-14, dsDNA (Sigma-Aldrich), and sm-RNP (ATRO1-10; Arotech Diagnostics Limited) at 100 µg/ml by ELISA with cloned monoclonal IgG antibodies at 10 µg/ml and serially diluted 1:5, 1:25, and 1:125. For ELISAs, 96-well Immuno plates (Thermo Fisher Scientific) were precoated (100 µg/ml) overnight at 4°C with dsDNA, PC-4, PC-14, sm-RNP, or MDA-LDL. After blocking with 0.5% BSA/PBS, recombinant antibodies were added, and plates were incubated with mouse anti–human IgG-HRP (SouthernBiotech; 1:2,000 dilution). IFA slides (BD) were stained according to the manufacturer’s instructions and were imaged following the same protocol described in Schwartz et al. (2014). Serum BAFF levels were measured using the BAFF/BlySS Quantikine ELISA kit (R&D Systems). Serum 9G4+ IgG levels were measured by coating ELISA plates with purified 9G4 IgG antibody (10 µg/ml) provided by I. Sanz. 9G4 IgG titers were detected using an HRP-anti–human IgG (JDC-10) secondary antibody.
High-throughput BCR sequencing.
Total RNA was obtained from purified murine B cells, cDNA was synthesized using a 5′-RACE kit (Ambion), and BCR heavy chain genes were amplified using a triple-nested primer strategy (as previously described in Schwartz et al. [2014]). Total RNA was obtained from purified B cells (0.5–10 × 106 cells) using RNeasy reagents (QIAGEN). Heavy chain cDNAs from each sample were synthesized using a 5′-RACE kit (Ambion) according to the manufacturer’s protocol. For RT and PCR, Transcriptor High Fidelity (Roche) and Phusion Hot Start (New England Biolabs) were used, respectively. A triple-nested primer strategy was used to amplify heavy chain IgM and IgD genes. An outer IgM and IgD constant region–specific primer was used for the RT reaction, a middle constant region IgM and IgD primer was used for the first round of PCR, and an innermost constant region IgM and IgD primer (adjacent to the J segment of the variable region) was used for the second PCR round. 454 adapters were included in the primers during second-round PCR, and a barcode strategy was used to run multiple samples simultaneously. Amplicon libraries were used for high-throughput pyrosequencing on a GS Junior (MYcroarray). Barcoded sequence data were separated using Geneious software, and IMGT/HighVQuest was used for alignment to germline IgH VDJ regions. IgAT software was used to generate descriptive statistics and calculation of CDR3 characteristics. Total number of sequences generated for murine sequencing can be found in Table S1. Sequencing files for the murine BCR repertoire experiments described can be found using the BioProject accession no. PRJNA294425.
For high-throughput sequencing of human WAS and HC subjects, genomic DNA was extracted from sorted transitional, naive, and both IgM and switched memory B cell populations. Ig heavy chain sequences were independently amplified using multiplex PCR with optimized primer sets. After high-throughput sequencing using Adaptive Biotechnologies’ ImmunoSEQ Illumina-based sequencing platform, sequences are then clustered into distinct clonotypes to determine overall frequencies. Clonotypes are identified based on their CDR3 (complementarity determining region 3) sequences. Clones at >5% of the total frequency within each sample were identified as index trackable sequences. V, (D), and J genes were identified for each clonotype. Total number of sequences generated and analyzed for human WAS and HC subjects can be found in Table S3. Mean VH genes in both human WAS and HC subjects can be found in Table S4. Sequencing files for the human B cell repertoire experiments described above can be found at http://www.adaptivebiotech.com/pub/Kolhatkar-2015-JEM (open to the public but free account setup required).
Single-cell BCR cloning.
Single murine MZ B cells (B220hiCD24hiCD23loCD1dhi) were FACS sorted from five to six 10–12-wk-old WT and Was−/− mice into 96-well plates. BCRs were cloned from the cDNA of single cells and used to generate mAbs using methods previously described in Tiller et al. (2009) and Schwartz et al. (2014). Human naive B cells were sorted from HC (one adult and two pediatric subjects) and WAS subjects (one pediatric subject) into 96-well plates. cDNA was extracted from single cells and used to generate mAbs using methods previously described in Tiller et al. (2008).
Cell cycle analysis using in vivo BrdU labeling.
For in vivo labeling of cycling cells, mice were given water with 1 mg BrdU and 20 g sucrose for 24 h. Both spleens and BM were collected, and cells were surface stained, fixed, and treated with DNase for 1 h at 37°C and then stained for intracellular BrdU and Ki67 immediately before FACS analysis (as previously described in Schwartz et al. [2014]).
Statistical analysis.
Unpaired, two-tailed Student’s t tests were applied to determine the statistical significance of the differences between groups. The p-values were considered significant when P < 0.05 (*), P < 0.01 (**), and P < 0.001 (***).
Online supplemental material.
Additional information regarding both human and murine sequencing datasets (cell numbers, total number of sequences, and total number of clonotypic sequences) is listed in Tables S1 and S3. WAS and HC subject clinical information (age, WAS mutation, and clinical presentation) is listed in detail in Table S2. Complete mean VH gene sequencing data for sorted naive B cells isolated from pediatric WAS and HC subjects and displayed in Fig. 6 are provided in Table S4. Summary of experimental assays in this study in both murine models of WASp deficiency and WAS subjects is provided in Table S5.
Acknowledgments
The authors would like to thank Drs. Suzanne Skoda Smith, Maite de la Morena (University of Texas Southwestern, Dallas, TX), Rob Sokolic (National Institutes of Health [NIH], Bethesda, MD), and Elizabeth Garabedian (NIH) for helping to acquire WAS subject samples, as well as Dr. Michael Reth for generously providing Mb-1cre mice.
This work was supported by the National Heart, Lung, and Blood Institute, National Institute of Diabetes and Digestive and Kidney Diseases, and National Institute of Allergy and Infectious Diseases of the NIH under award numbers R01HL075453, R01AI084457, R01AI071163, and DP3DK097672 (all to D.J. Rawlings); 5P01HL059561 (to L.D. Notarangelo); and UO1 DK070430 (to E.T. Luning Prak). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. Additional support was provided by the Wiskott-Aldrich Foundation and Benaroya Family Gift Fund (to D.J. Rawlings), an ACR REF/Ephraim P. Engleman Endowed Resident Research Preceptorship (to E.J. Allenspach), and a Cancer Research Institute Pre-doctoral Training Grant (to N.S. Kolhatkar and M.A. Schwartz).
The authors declare no competing financial interests.
References
- AUC
area under the curve
- B6
C57BL/6
- BAFF
B cell–activating factor
- BCR
B cell receptor
- FM
follicular mature
- HC
healthy control
- HEL
hen egg lysozyme
- Id
idiotype
- IFA
indirect fluorescence antibody
- LC
light chain
- MDA
malondialdehyde
- MFI
mean fluorescence intensity
- mHEL
membrane-bound HEL
- MZ
marginal zone
- MZp
MZ precursor
- RACE
rapid amplification of cDNA ends
- RS
recombining sequence
- sHEL
soluble HEL
- SHM
somatic hypermutation
- Tg
transgenic
- WAS
Wiskott-Aldrich syndrome
- WASp
WAS protein
- XLT
X-linked thrombocytopenia
Author notes
S. Becker-Herman’s present address is the Dept. of Immunology, The Weizmann Institute of Science, Rehovot 76100, Israel.
F. Candotti’s present address is the Division of Immunology and Allergy, University Hospital of Lausanne (CHUV), CH-1011 Lausanne, Switzerland.


![Figure 1. Altered specificity of the naive B cell repertoire in WASp-deficient mice. (A) λ-LC usage in splenic B cell populations in 8–10-wk-old B6 (n = 6), Wasfl/fl × Mb-1cre (n = 5), and Was−/− (n = 7) mice assessed by flow cytometry. (B and C) Cloned WT and Was−/− MZ B cell mAb reactivities toward self-antigens (dsDNA, high [PC-4]- and low [PC-14]-affinity phosphorylcholine, MDA-LDL, and Sm-RNP) via ELISA depicted using a pie chart (blue = reactive clones identified based on threshold of 0.5 OD value; gray = nonreactive clones), with percentages of reactive clones and total number of clones tested noted. MZ B cells were FACS sorted and gated based on B220+CD23loCD1dhiCD24hiCD21hi surface marker expression from splenocytes pooled from five to six WT or Was−/− mice. (C) ELISA OD values of serial dilution curves of WT and Was−/− MZ mAbs (100 ng/µl). (D) Proportion of low affinity (OD of 0.5–1.5) and high affinity (OD of 1.5–3) in reactive antibody clones to individual self-antigens. (E) Relative binding affinity displayed as AUC of reactive antibodies. (F) Selection of Id (M167)+ B cells in peripheral B cell subsets in 10–12-wk-old WT M167 Tg (n = 9) and Was−/− M167 Tg (n = 9) mice. Error bars show SEM. Statistical analysis was performed using the Student’s t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Data are representative of at least two experiments.](https://cdn.rupress.org/rup/content_public/journal/jem/212/10/10.1084_jem.20150585/5/m_jem_20150585_fig1.jpeg?Expires=1781609526&Signature=YbzHc5~11lkNWL5WGt-tzDC~PibO55aZraDP-ttzlsXEQDPwKwCfsj7s-RbgSi-PWtJGhs0FHPyJyEwfugaUxxN8zDlBv7bm1hhoaZ8C2UlEy-rne3jxnQplDSdWb8RpErf4r-rzVTB0I-05m2DX-~CHZqGi9g3~GS3NVol8iZ~xgsnLsT6p4xnhm0xYoQUKZijOmEztQ2asu~G0zrEkbnXME0iXSwD6m-9DJTiKNqc2Wy~9YL9gnHpJS~9EPaZVlWoBm~JYUwcX~udfC4amGyaO3eRH0WwUhvKUv86l8hn0O6FzqEgH5rP22jkGdaPdefoYNvmHIR9I3~bTrXh3Ww__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 3. Was−/− B cells exhibit intact negative selection. (A–C) The HEL-Ig/mHel double Tg (DTg) chimeric transplant model was used to determine whether self-reactive Was−/− B cells undergo deletion in the BM. Recipients were analyzed at 6 wk after transplant (n = 6–7 per group; data are representative of two independent experiments). (A and B) BM B cells were analyzed by FACS. (C) HEL-specific serum antibody levels. (D–F) The HEL-Ig/sHel DTg system was used to determine whether self-reactive Was−/− B cells undergo efficient induction of anergy (n = 6–7 per group; data are representative of two independent experiments). (D and E) BM B cells were analyzed by FACS. (F) HEL-specific serum antibody levels. (G) Quantitative assay for RS rearrangement was used to estimate κ-LC rearrangement in BM and spleen B cell subsets of 6–8-wk-old WT or Was−/− mice. Results are normalized to β-actin and presented as fold difference relative to WT κ+ FM B cells (n = 7 in each group except for Was−/− fractions E [n = 6] and F [n = 5]). Error bars show SEM. Statistical analysis was performed using the Student’s t test: ***, P < 0.001. Data are representative of at least two independent experiments.](https://cdn.rupress.org/rup/content_public/journal/jem/212/10/10.1084_jem.20150585/5/m_jem_20150585_fig3.jpeg?Expires=1781609526&Signature=gBwcnmoB9ck9vf1sWwH-pq4vb-P4SZu7L1Lqn3p6qbOPr7oUKQ6-PeL0u6OS-CeCdtrok4JxoOAp0JjsZKkD5fjAEPE-4ueAotEOZE3WJMnOvMOeVLxh~wElv61qVBZi1DO6okcTCe-kPr5s6rgt3fAJQukqZ5-g~BA5rAUEBeCUtFwofUARe86pQ98J0RJ9KpQ1XoAjIFdgtaYOWQb9Yka93K~eaHqy2w16qtS647thqaqXr0QNGMgX~QkURI7q-Cd~QurVMIQN3LCLWwjYSvyTcIJehW1twDKHa67DzjcRNJgK5bQjVlfImxQ2qYSudvdLOXkqitTzHuzIfhcdOQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)