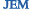Old mice that fight off a second strain of the flu (left column) have more NP epitope-specific T cells than do mice that stay infected (right column).
The CD8+ T cell pool is finite and in flux throughout an individual's lifetime. Each infection strengthens the numbers of T cells specific to that infecting microbe. In time, the T cell pool thus becomes skewed: the large number antigen-specific clones decreases the number and diversity of remaining naive T cells.
Yager et al. now show that this age-related shrinking of the naive T cell pool results in a loss of the ability to respond to epitopes for which there were a low number of precursors to start with. In young mice, CD8+ T cells specific for a particular influenza nucleoprotein (NP) epitope were at least ten times less frequent than those specific for other flu epitopes. This NP-specific naive T cell population was greatly reduced in most old mice. Age-related decay of the thymus, which maintains the naive T cell repertoire, may be at least partly to blame: young mice with a surgically removed thymus experienced similar declines in their naive T cell pool.
The resulting inability of the old mice to mount a strong immune response against the NP epitope weakened their ability to protect themselves against different flu strains. Older flu-infected mice that had the fewest NP-specific T cells were least adept at fighting off a second infection with a different flu strain, even though these mice had plenty of T cells directed against other flu epitopes. Why non–NP-specific T cells fail to protect the mice is unclear.
The findings suggest that current efforts to boost flu-fighting power in the elderly with vaccines carrying multiple T cell epitopes might misfire, as the naive T cells that would respond to these vaccines are missing. As the preferential loss of low frequency naive T cells is probably not unique to the flu, prolonging thymic function and vaccinating against as many microbes as possible before the thymus deteriorates too far might be better strategies.